Oxandrolone
 | |
 | |
| Names | |
|---|---|
| Trade names | Oxandrin, Anavar, others |
| Other names | Var; CB-8075; NSC-67068; SC-11585; Protivar; 17α-Methyl-2-oxa-4,5α-dihydrotestosterone; 17α-Methyl-2-oxa-DHT; 17α-Methyl-2-oxa-5α-androstan-17β-ol-3-one |
| |
| Clinical data | |
| Drug class | Androgen; anabolic steroid |
| Main uses | Promote weight gain, prevent protein breakdown from corticosteroids, bone pain due to osteoporosis[1][2] |
| Side effects | Liver problems, lipid abnormalities[1] |
| Pregnancy category |
|
| Routes of use | By mouth |
| External links | |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a604024 |
| Legal | |
| Legal status |
|
| Pharmacokinetics | |
| Bioavailability | 97%[3] |
| Protein binding | 94–97%[3] |
| Metabolism | Kidneys (primarily), liver[4][3] |
| Elimination half-life | Adults: 9.4–10.4 hours[3][5] Elderly: 13.3 hours[5] |
| Excretion | Urine: 28% (unchanged)[5] Feces: 3%[5] |
| Chemical and physical data | |
| Formula | C19H30O3 |
| Molar mass | 306.446 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Oxandrolone, sold under the brand names Oxandrin among others, is a manufactured androgen and anabolic steroid (AAS).[1] It is used to promote weight gain in those losing weight, prevent protein breakdown caused by long-term corticosteroids, and bone pain associated with osteoporosis.[1][2] It is taken by mouth.[1]
Common side effects include liver problems and lipid abnormalities.[1] Other side effects may include heart failure, misuse, acne, increased hair growth, and voice changes.[1] Use during pregnancy may harm the baby.[1] Oxandrolone activates the androgen receptor (AR), the biological target of testosterone.[6] It has greater anabolic than androgenic effects.[1]
Oxandrolone was approved for medical use in the United States in 1964.[1] It is not medically available in the United Kingdom.[6] In the United States 60 tablets of 10 mg costs about 284 USD as of 2020.[7] It has also been inappropriately used to improve physique and performance in sports.[1] It is a controlled substance in many countries, so nonmedical is generally illegal.[8][9][10]
Uses
Medical
Oxandrolone has been researched and prescribed as a treatment for a wide variety of conditions. It is FDA-approved for treating bone pain associated with osteoporosis, aiding weight gain following surgery or physical trauma, during chronic infection, or in the context of unexplained weight loss, and counteracting the catabolic effect of long-term corticosteroid therapy.[11][12]As of 2016[update], it is often prescribed off-label to quicken recovery from severe burns, aid the development of girls with Turner syndrome, and counteract HIV/AIDS-induced wasting. Oxandrolone improves both short-term and long-term outcomes in people recovering from severe burns and is well-established as a safe treatment for this indication.[2][13] It is also used in the treatment of idiopathic short stature, anemia, hereditary angioedema, alcoholic hepatitis, and hypogonadism.[14][15]
Medical research has established the effectiveness of oxandrolone in aiding the development of girls with Turner syndrome. Although oxandrolone has long been used to accelerate growth in children with idiopathic short stature, it is unlikely to increase adult height, and in some cases may even decrease it[citation needed]. Oxandrolone has, therefore, largely been replaced by growth hormone for this use.[16] Children with idiopathic short stature or Turner syndrome are given doses of oxandrolone far smaller than those given to people with burns to minimize the likelihood of virilization and premature maturation.[16][17]
Non-medical
Many bodybuilders and athletes use oxandrolone for its muscle-building effects. It is much more anabolic than androgenic, so women and those seeking less intense steroid regimens use it particularly often. Many also value oxandrolone's low hepatotoxicity relative to most other orally active AAS.
Contraindications
Like other AAS, oxandrolone may worsen hypercalcemia by increasing osteolytic bone resorption.[11] When taken by pregnant women, oxandrolone may have unintended effects such as masculinization on the fetus.[11]
Side effects
Women who are administered oxandrolone may experience virilization, irreversible development of masculine features such as voice deepening, hirsutism, menstruation abnormalities, male-pattern hair loss, and clitoral enlargement.[16][11][17] Oxandrolone may disrupt growth in children, reducing their adult height.[18][better source needed] Because of these side effects, doses given to women and children are minimized and people are usually monitored for virilization and growth abnormalities.[16][17] Like other androgens, oxandrolone can cause or worsen acne and priapism (unwanted or prolonged erections).[11][18] Oxandrolone can also reduce males' fertility, another side effect common among androgens.[18] In an attempt to compensate for the exogenous increase in androgens, the body may reduce testosterone production via testicular atrophy and inhibition of gonadotropic activity.[11]
Unlike some AAS, oxandrolone does not generally cause gynecomastia because it is not aromatized into estrogenic metabolites.[19] However, although no reports of gynecomastia were made in spite of widespread use, oxandrolone was reported in a publication in 1991 to have been associated with 33 cases of gynecomastia in adolescent boys treated with it for short stature.[20][21] The gynecomastia developed during oxandrolone therapy in 19 of the boys and after the therapy was completed in 14 of the boys, and 10 of the boys had transient gynecomastia, while 23 had persistent gynecomastia that necessitated mastectomy.[20][21] Though transient gynecomastia is a natural and common occurrence in pubertal boys, the gynecomastia associated with oxandrolone was of a late/delayed onset and was persistent in a high percentage of the cases.[20][21] As such, the researchers stated, "although oxandrolone cannot be implicated as stimulatory [in] gynecomastia", a possible relationship should be considered in clinicians using oxandrolone in adolescents for growth stimulation.[20][21]
Uniquely among 17α-alkylated AAS, oxandrolone shows little to no hepatotoxicity, even at high doses.[22] No cases of severe hepatotoxicity have been singularly attributed to oxandrolone.[22] However, elevated liver enzymes have been observed in some people, particularly with high doses and/or prolonged treatment, although they return to normal ranges following discontinuation.[22]
Interactions
Oxandrolone greatly increases warfarin's blood-thinning effect, sometimes dangerously so.[23] In April 2004, Savient Pharmaceuticals published a safety alert through the FDA warning healthcare professionals of this.[24] Oxandrolone can also inhibit the metabolism of oral hypoglycemic agents.[11] It may worsen edema when taken alongside corticosteroids or adrenocorticotropic hormone.[11]
Pharmacology
Pharmacodynamics
| Medication | Ratioa |
|---|---|
| Testosterone | ~1:1 |
| Androstanolone (DHT) | ~1:1 |
| Methyltestosterone | ~1:1 |
| Methandriol | ~1:1 |
| Fluoxymesterone | 1:1–1:15 |
| Metandienone | 1:1–1:8 |
| Drostanolone | 1:3–1:4 |
| Metenolone | 1:2–1:30 |
| Oxymetholone | 1:2–1:9 |
| Oxandrolone | 1:3–1:13 |
| Stanozolol | 1:1–1:30 |
| Nandrolone | 1:3–1:16 |
| Ethylestrenol | 1:2–1:19 |
| Norethandrolone | 1:1–1:20 |
| Notes: In rodents. Footnotes: a = Ratio of androgenic to anabolic activity. Sources: See template. | |
Like other AAS, oxandrolone is an agonist of the androgen receptor, similar to androgens such as testosterone and DHT. The relative binding affinity of oxandrolone for the androgen receptor is about 0.8% of that of metribolone.[25] Activation of the androgen receptor stimulates protein synthesis, which increases muscle growth, lean body mass, and bone mineral density.[13]
Compared to testosterone and many other AAS, oxandrolone is less androgenic relative to its strength as an anabolic.[26] Oxandrolone has about 322 to 633% of the anabolic potency and 24% of the androgenic potency of methyltestosterone. Similarly, oxandrolone has as much as 6 times the anabolic potency of testosterone and has significantly reduced androgenic potency in comparison. The reduced ratio of anabolic to androgenic activity of oxandrolone often motivates its medical use in children and women because less androgenic effect implies less risk of virilization. The bodybuilding community also considers this fact when choosing between AAS.
As oxandrolone is already 5α-reduced, it is not a substrate for 5α-reductase, hence is not potentiated in androgenic tissues such as the skin, hair follicles, and prostate gland. This is involved in its reduced ratio of anabolic to androgenic activity. Due to the substitution of one of the carbon atoms with an oxygen atom at the C2 position in the A ring, oxandrolone is resistant to inactivation by 3α-hydroxysteroid dehydrogenase in skeletal muscle. This is in contrast to DHT, and is thought to underlie the preserved anabolic potency with oxandrolone. Because it is 5α-reduced, oxandrolone is not a substrate for aromatase, hence cannot be aromatized into metabolites with estrogenic activity. Oxandrolone similarly possesses no progestogenic activity.
Oxandrolone is, uniquely, far less hepatotoxic than other 17α-alkylated AAS, which may be due to differences in metabolism.[22][4][5]
Pharmacokinetics
The oral bioavailability of oxandrolone is 97%.[3] Its plasma protein binding is 94 to 97%.[3] The drug is metabolized primarily by the kidneys and to a lesser extent by the liver.[4][3] Oxandrolone is the only AAS that is not primarily or extensively metabolized by the liver, and this is thought to be related to its diminished hepatotoxicity relative to other AAS.[4][5] Its elimination half-life is reported as 9.4 to 10.4 hours, but is extended to 13.3 hours in the elderly.[3][5] About 28% of an oral dose of oxandrolone is eliminated unchanged in the urine and 3% is excreted in the feces.[5]
Chemistry
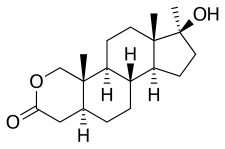
Oxandrolone is a synthetic androstane steroid and a 17α-alkylated derivative of DHT.[27][28] It is also known as 2-oxa-17α-methyl-5α-dihydrotestosterone (2-oxa-17α-methyl-DHT) or as 2-oxa-17α-methyl-5α-androstan-17β-ol-3-one, and is DHT with a methyl group at the C17α position and the C2 carbon replaced with an oxygen atom.[27][28] Closely related AAS include the marketed AAS mestanolone (17α-methyl-DHT), oxymetholone (2-hydroxymethylene-17α-methyl-DHT), and stanozolol (a 2,3-pyrazole A ring-fused derivative of 17α-methyl-DHT) and the never-marketed/designer AAS desoxymethyltestosterone (3-deketo-17α-methyl-δ2-DHT), methasterone (2α,17α-dimethyl-DHT), methyl-1-testosterone (17α-methyl-δ1-DHT), and methylstenbolone (2,17α-dimethyl-δ1-DHT).[27][28]
History
Oxandrolone was first made by Raphael Pappo and Christopher J. Jung while at Searle Laboratories (now part of Pfizer). The researchers first described the drug in 1962.[29][30] They were immediately interested in oxandrolone's very weak androgenic effects relative to its anabolic effects.[29] It was introduced as a pharmaceutical drug in the United States in 1964.
The drug was prescribed to promote muscle regrowth in disorders which cause involuntary weight loss, and is used as part of treatment for HIV/AIDS. It had also been shown to be partially successful in treating cases of osteoporosis. However, in part due to bad publicity from its illicit use by bodybuilders, production of Anavar was discontinued by Searle Laboratories in 1989. It was picked up by Bio-Technology General Corporation, which changed its name to Savient Pharmaceuticals, which following successful clinical trials in 1995, released it under the brand name Oxandrin. BTG subsequently won approvals for orphan drug status by the Food and Drug Administration for treating alcoholic hepatitis, Turner syndrome, and HIV-induced weight loss. It is also indicated as an offset to protein catabolism caused by long-term administration of corticosteroids.
Society and culture
Generic names
Oxandrolone is the generic name of the drug and its INN, USAN, USP, BAN, DCF, DCIT, and JAN, while ossandrolone is or was formerly the DCIT.[27][28][31][32][33]
Brand names
The original brand name of oxandrolone was Anavar, which was marketed in the United States and the Netherlands.[34] This product was eventually discontinued and replaced in the United States with a new product named Oxandrin, which is the sole remaining brand name for oxandrolone in the United States.[35] Oxandrolone has also been sold under the brand names Antitriol (Spain), Anatrophill (France), Lipidex (Brazil), Lonavar (Argentina, Australia, Italy), Protivar, and Vasorome (Japan), among others.[28][34][36] Additional brand names exist for products that are manufactured for the steroid black market.
Among those using oxandrolone for nonmedical purposes, it is often referred to colloquially as "Var", a shortened form of the brand name Anavar.[37][38]
Availability
United States
Oxandrolone is one of the few AAS that remain available for medical use in the United States.[35] The others (as of November 2017) are testosterone, testosterone cypionate, testosterone enanthate, testosterone undecanoate, methyltestosterone, fluoxymesterone, and oxymetholone.[35]
Other countries
Outside of the United States, the availability of oxandrolone is quite limited.[32] With the exception of Moldova, it is no longer available in Europe. Oxandrolone is available in some less-regulated markets in Asia such as Malaysia. It is also available in Mexico. Historically, oxandrolone has been marketed in Argentina, Australia, Brazil, France, Italy, Japan, and Spain, but it appears to no longer be available in these countries.[28][34][32]
Legal status
In the United States, oxandrolone is categorized as a Schedule III controlled substance under the Controlled Substances Act along with many other AAS.[8] It is a Schedule IV controlled substance in Canada,[9] and a Schedule 4 controlled drug in the United Kingdom.[10]
Doping in sports
Oxandrolone is sometimes used as a doping agent in sports. Cases of doping with oxandrolone by professional athletes have been reported.
References
- ↑ 1.00 1.01 1.02 1.03 1.04 1.05 1.06 1.07 1.08 1.09 1.10 "Oxandrolone Monograph for Professionals". Drugs.com. Archived from the original on 27 February 2021. Retrieved 14 December 2020.
- ↑ 2.0 2.1 2.2 Li H, Guo Y, Yang Z, Roy M, Guo Q (June 2016). "The efficacy and safety of oxandrolone treatment for patients with severe burns: A systematic review and meta-analysis". Burns. 42 (4): 717–27. doi:10.1016/j.burns.2015.08.023. PMID 26454425.
- ↑ 3.0 3.1 3.2 3.3 3.4 3.5 3.6 3.7 Ashraf Mozayani; Lionel Raymon (15 October 2003). Handbook of Drug Interactions: A Clinical and Forensic Guide. Springer Science & Business Media. pp. 513–. ISBN 978-1-59259-654-6. Archived from the original on 15 December 2019. Retrieved 6 November 2017.
- ↑ 4.0 4.1 4.2 4.3 R.A.S Hemat (2 March 2003). Andropathy. Urotext. pp. 108–. ISBN 978-1-903737-08-8. Archived from the original on 2021-07-25. Retrieved 2017-11-06.
- ↑ 5.0 5.1 5.2 5.3 5.4 5.5 5.6 5.7 Miller JT, Btaiche IF (February 2009). "Oxandrolone treatment in adults with severe thermal injury" (PDF). Pharmacotherapy. 29 (2): 213–26. doi:10.1592/phco.29.2.213. hdl:2027.42/90285. PMID 19170590. S2CID 25780591. Archived from the original on 2021-08-29. Retrieved 2019-08-30.
- ↑ 6.0 6.1 Kicman AT (June 2008). "Pharmacology of anabolic steroids". British Journal of Pharmacology. 154 (3): 502–21. doi:10.1038/bjp.2008.165. PMC 2439524. PMID 18500378.
- ↑ "Oxandrolone Prices, Coupons & Savings Tips". GoodRx. Archived from the original on 9 November 2016. Retrieved 14 December 2020.
- ↑ 8.0 8.1 "Controlled Substances Act". United States Food and Drug Administration. 11 June 2009. Archived from the original on 2 March 2017. Retrieved 17 June 2016.
- ↑ 9.0 9.1 Branch, Legislative Services. "Consolidated federal laws of Canada, Controlled Drugs and Substances Act". laws-lois.justice.gc.ca. Archived from the original on 2017-02-05. Retrieved 2017-01-14.
- ↑ 10.0 10.1 "List of most commonly encountered drugs currently controlled under the misuse of drugs legislation - GOV.UK". www.gov.uk. Archived from the original on 2019-12-08. Retrieved 2017-01-14.
- ↑ 11.0 11.1 11.2 11.3 11.4 11.5 11.6 11.7 "Oxandrolone Tablets, USP - Rx only" (PDF). Drugs@FDA. U.S. Food and Drug Administration. 1 December 2006. Archived (PDF) from the original on 26 August 2016. Retrieved 21 June 2016.
- ↑ "Oxandrin (oxandrolone tablets, USP)" (PDF). Drugs@FDA. BTG Pharmaceuticals, U.S. Food and Drug Administration. 21 April 2003. Archived (PDF) from the original on 1 March 2017. Retrieved 21 June 2016.
- ↑ 13.0 13.1 Rojas Y, Finnerty CC, Radhakrishnan RS, Herndon DN (December 2012). "Burns: an update on current pharmacotherapy". Expert Opinion on Pharmacotherapy. 13 (17): 2485–94. doi:10.1517/14656566.2012.738195. PMC 3576016. PMID 23121414.
- ↑ Bork K (August 2012). "Current management options for hereditary angioedema". Current Allergy and Asthma Reports. 12 (4): 273–80. doi:10.1007/s11882-012-0273-4. PMID 22729959. S2CID 207323793.
- ↑ Choi G, Runyon BA (May 2012). "Alcoholic hepatitis: a clinician's guide". Clinics in Liver Disease. 16 (2): 371–85. doi:10.1016/j.cld.2012.03.015. PMID 22541704.
- ↑ 16.0 16.1 16.2 16.3 Wit JM, Oostdijk W (June 2015). "Novel approaches to short stature therapy". Best Practice & Research. Clinical Endocrinology & Metabolism. 29 (3): 353–66. doi:10.1016/j.beem.2015.01.003. PMID 26051296.
- ↑ 17.0 17.1 17.2 Sas TC, Gault EJ, Bardsley MZ, Menke LA, Freriks K, Perry RJ, et al. (2014). "Safety and efficacy of oxandrolone in growth hormone-treated girls with Turner syndrome: evidence from recent studies and recommendations for use". Hormone Research in Paediatrics. 81 (5): 289–97. doi:10.1159/000358195. PMID 24776783.
- ↑ 18.0 18.1 18.2 "Oxandrolone". MedlinePlus. The American Society of Health-System Pharmacists, Inc. 15 May 2016. Archived from the original on 5 July 2016. Retrieved 21 June 2016.
- ↑ Corona G, Rastrelli G, Vignozzi L, Maggi M (June 2012). "Emerging medication for the treatment of male hypogonadism". Expert Opinion on Emerging Drugs. 17 (2): 239–59. doi:10.1517/14728214.2012.683411. PMID 22612692. S2CID 22068249.
- ↑ 20.0 20.1 20.2 20.3 Jeffrey K. Aronson (21 February 2009). Meyler's Side Effects of Endocrine and Metabolic Drugs. Elsevier. pp. 142–. ISBN 978-0-08-093292-7.
- ↑ 21.0 21.1 21.2 21.3 Moore, D.C.; Ruvalcaba, R.HA. (1991). "Late Onset Gynecomastia Associated with Oxandrolone Therapy in Adolescents with Short Stature". Journal of Pediatric Endocrinology and Metabolism. 4 (4). doi:10.1515/JPEM.1991.4.4.249. ISSN 2191-0251. S2CID 56669464.
- ↑ 22.0 22.1 22.2 22.3 Orr R, Fiatarone Singh M (2004). "The anabolic androgenic steroid oxandrolone in the treatment of wasting and catabolic disorders: review of efficacy and safety". Drugs. 64 (7): 725–50. doi:10.2165/00003495-200464070-00004. PMID 15025546. S2CID 32262454.
- ↑ Demling, Robert H. (September 2004). "Oxandrolone (Oxandrin) use and the interaction with warfarin" (PDF). U.S. Food and Drug Administration. Archived (PDF) from the original on 15 February 2017. Retrieved 20 June 2016.
- ↑ Ottery, Faith D. (20 April 2004). "Oxandrin (oxandrolone) Dear Healthcare Professional Letter Apr 2004". Safety Alerts for Human Medical Products. U.S. Food and Drug Administration. Archived from the original on 21 August 2016. Retrieved 20 June 2016.
- ↑ Dalton, James T.; Gao, Wenqing (2010). "Androgen Receptor". Nuclear Receptors. pp. 143–182. doi:10.1007/978-90-481-3303-1_6. ISBN 978-90-481-3302-4.
- ↑ Chrousos, George P. (2012). "The Gonadal Hormones & Inhibitors". In Katzung, Bertram G. (ed.). Basic & Clinical Pharmacology. New York London: McGraw-Hill Medical McGraw-Hill distributor. p. 735. ISBN 978-0071764018.
- ↑ 27.0 27.1 27.2 27.3 Elks J (14 November 2014). The Dictionary of Drugs: Chemical Data: Chemical Data, Structures and Bibliographies. Springer. pp. 911–. ISBN 978-1-4757-2085-3.
- ↑ 28.0 28.1 28.2 28.3 28.4 28.5 Index Nominum 2000: International Drug Directory. Taylor & Francis. 2000. pp. 767–. ISBN 978-3-88763-075-1.
- ↑ 29.0 29.1 Pappo, Raphael; Jung, Christopher J. (1962). "2-oxasteroids: A new class of biologically active compounds". Tetrahedron Letters. 3 (9): 365–371. doi:10.1016/S0040-4039(00)70883-5. ISSN 0040-4039.
- ↑ Fox M, Minot AS, Liddle GW (September 1962). "Oxandrolone: a potent anabolic steroid of novel chemical configuration". The Journal of Clinical Endocrinology and Metabolism. 22 (9): 921–4. doi:10.1210/jcem-22-9-921. PMID 13894381.
- ↑ Morton IK, Hall JM (6 December 2012). Concise Dictionary of Pharmacological Agents: Properties and Synonyms. Springer Science & Business Media. pp. 211–. ISBN 978-94-011-4439-1.
- ↑ 32.0 32.1 32.2 "Oxandrolone". Archived from the original on 2017-11-12. Retrieved 2017-11-12.
- ↑ World Health Organization (1982). International Nonproprietary Names (INN) for Pharmaceutical Substances: Cumulative List: Dénominations Communes Internationales (DCI) Pour Les Substances Pharmaceutiques: Liste Récapitulative. p. 225. ISBN 978-92-4-056013-0.
- ↑ 34.0 34.1 34.2 William Andrew Publishing (22 October 2013). Pharmaceutical Manufacturing Encyclopedia, 3rd Edition. Elsevier. pp. 2935–. ISBN 978-0-8155-1856-3.
- ↑ 35.0 35.1 35.2 "Drugs@FDA: FDA Approved Drug Products". United States Food and Drug Administration. Archived from the original on 16 November 2016. Retrieved 11 November 2017.
- ↑ Drugs of Abuse (PDF). United States Drug Enforcement Administration. 2011. p. 22. Archived (PDF) from the original on 2016-08-22. Retrieved 2016-06-19.
- ↑ Petros Levounis; Erin Zerbo; Rashi Aggarwal (3 May 2016). Pocket Guide to Addiction Assessment and Treatment. American Psychiatric Pub. pp. 69–. ISBN 978-1-61537-072-6.
External links
| Identifiers: |
|
|---|
- Oxandrin Homepage, savientpharma.com (via archive.org)
- Oxandrin Label Archived 2017-08-01 at the Wayback Machine, fda.gov (retrieved 23 October 2009)
- "Oxandrolone Side Effects, Interactions and Information". drugs.com. Archived from the original on 2018-02-10. Retrieved 2018-01-23.
- Pages using duplicate arguments in template calls
- Chemical articles with unknown parameter in Infobox drug
- Chemical articles without CAS registry number
- Articles without EBI source
- Chemical pages without ChemSpiderID
- Chemical pages without DrugBank identifier
- Articles without KEGG source
- Articles without UNII source
- Drugs missing an ATC code
- Articles containing potentially dated statements from 2016
- Articles with invalid date parameter in template
- All articles containing potentially dated statements
- All articles with unsourced statements
- Articles with unsourced statements from December 2019
- Articles with hatnote templates targeting a nonexistent page
- All articles lacking reliable references
- Articles lacking reliable references from June 2017
- Webarchive template wayback links
- Androgens and anabolic steroids
- Androstanes
- Appetite stimulants
- Hepatotoxins
- Lactones
- Tertiary alcohols
- World Anti-Doping Agency prohibited substances
- RTT