Anastrozole
 | |
 | |
| Names | |
|---|---|
| Trade names | Arimidex, Aremed, others |
| Other names | Anastrazole; anastrozol; ICI-D1033; ZD-1033 |
| |
| Clinical data | |
| Drug class | Aromatase inhibitor; Antiestrogen |
| Main uses | Breast cancer[2] |
| Side effects | Hot flashes, altered mood, joint pain, nausea[3][2] |
| Pregnancy category |
|
| Routes of use | By mouth (tablets) |
| Defined daily dose | 1 mg[4] |
| External links | |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a696018 |
| Legal | |
| License data |
|
| Legal status | |
| Pharmacokinetics | |
| Bioavailability | Unknown (but well-absorbed in animals)[5] |
| Protein binding | 40%[6][7] |
| Metabolism | Liver (~85%) (N-dealkylation, hydroxylation, glucuronidation)[6][5][7] |
| Elimination half-life | 40–50 hours[6][5][7] |
| Excretion | Urine (11%)[6][5][7] |
| Chemical and physical data | |
| Formula | C17H19N5 |
| Molar mass | 293.374 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Anastrozole, sold under the brand name Arimidex among others, is a medication used in addition to other treatments for breast cancer.[2][3] Specifically it is used for hormone receptor-positive breast cancer.[3] It has also been used to prevent breast cancer in those at high risk.[3] It is taken by mouth.[3]
Common side effects of anastrozole include hot flashes, altered mood, joint pain, and nausea.[3][2] Severe side effects include an increased risk of heart disease and osteoporosis.[3] Use during pregnancy may harm the baby.[3] Anastrozole is in the aromatase-inhibiting family of medications.[3] It works by blocking the production of estrogens in the body, and hence has antiestrogenic effects.[3]
Anastrozole was patented in 1987 and was approved for medical use in 1995.[8][9] It is on the World Health Organization's List of Essential Medicines as an alternative to letrozole.[10] It is available as a generic medication.[3] The wholesale cost in the developing world is about US$1.92–30.60 a month.[11] In the United States the wholesale cost is about $3.81 per month.[12] In 2017, it was the 258th most commonly prescribed medication in the United States, with more than one million prescriptions.[12][13]
Medical uses
Breast cancer
Anastrozole is used in the treatment and prevention of breast cancer in women.[3] The Arimidex, Tamoxifen, Alone or in Combination (ATAC) trial was of localized breast cancer and women received either anastrozole, the selective estrogen receptor modulator tamoxifen, or both for five years, followed by five years of follow-up.[14] After more than 5 years the group that received anastrozole had better results than the tamoxifen group.[14] The trial suggested that anastrozole is the preferred medical therapy for postmenopausal women with localized estrogen receptor-positive breast cancer.[14]
Early puberty
Anastrozole is used at a dosage of 0.5 to 1 mg/day in combination with the antiandrogen bicalutamide in the treatment of peripheral precocious puberty, for instance due to familial male-limited precocious puberty (testotoxicosis) and McCune–Albright syndrome, in boys.[15][16][17][18][19][20][21][22][23][24]
Dosage
The defined daily dose is 1 milligram (by mouth).[4]
Contraindications
Contraindications of anastrozole include hypersensitivity to anastrozole or any other component of anastrozole formulations, pregnancy, and breastfeeding.[2] Hypersensitivity reactions to anastrozole including anaphylaxis, angioedema, and urticaria have been observed.[2]
Side effects
Common side effects of anastrozole (≥10% incidence) include hot flashes, asthenia, arthritis, pain, arthralgia, hypertension, depression, nausea and vomiting, rash, osteoporosis, bone fractures, back pain, insomnia, headache, bone pain, peripheral edema, coughing, dyspnea, pharyngitis, and lymphedema.[2] Serious but rare adverse effects (<0.1% incidence) include skin reactions such as lesions, ulcers, or blisters; allergic reactions with swelling of the face, lips, tongue, and/or throat that may cause difficulty swallowing or breathing; and abnormal liver function tests as well as hepatitis.[2]
Interactions
Anastrozole is thought to have clinically negligible inhibitory effects on the cytochrome P450 enzymes CYP1A2, CYP2A6, CYP2D6, CYP2C8, CYP2C9, and CYP2C19.[6][5][7][2] As a result, it is thought that drug interactions of anastrozole with cytochrome P450 substrates are unlikely.[7] No clinically significant drug interactions have been reported with anastrozole as of 2003.[6]
Anastrozole does not affect circulating levels of tamoxifen or its major metabolite N-desmethyltamoxifen.[6][5] However, tamoxifen has been found to decrease steady-state area-under-the-curve levels of anastrozole by 27%.[6][5] But estradiol levels were not significantly different in the group that received both anastrozole and tamoxifen compared to the anastrozole alone group, so the decrease in anastrozole levels is not thought to be clinically important.[7]
Pharmacology
Pharmacodynamics
Anastrozole works by reversibly binding to the aromatase enzyme, and through competitive inhibition blocks the conversion of androgens to estrogens in peripheral (extragonadal) tissues.[25] The medication has been found to achieve 96.7% to 97.3% inhibition of aromatase at a dosage of 1 mg/day and 98.1% inhibition of aromatase at a dosage of 10 mg/day in humans.[6][5] As such, 1 mg/day is considered to be the minimal dosage required to achieve maximal suppression of aromatase with anastrozole.[6] This decrease in aromatase activity results in an at least 85% decrease in estradiol levels in postmenopausal women.[6] Levels of corticosteroids and other adrenal steroids are unaffected by anastrozole.[6]
| Generation | Medication | Dosage | % inhibitiona | Classb | IC50c |
|---|---|---|---|---|---|
| First | Testolactone | 250 mg 4x/day p.o. | ? | Type I | ? |
| 100 mg 3x/week i.m. | ? | ||||
| Rogletimide | 200 mg 2x/day p.o. 400 mg 2x/day p.o. 800 mg 2x/day p.o. |
50.6% 63.5% 73.8% |
Type II | ? | |
| Aminoglutethimide | 250 mg mg 4x/day p.o. | 90.6% | Type II | 4,500 nM | |
| Second | Formestane | 125 mg 1x/day p.o. 125 mg 2x/day p.o. 250 mg 1x/day p.o. |
72.3% 70.0% 57.3% |
Type I | 30 nM |
| 250 mg 1x/2 weeks i.m. 500 mg 1x/2 weeks i.m. 500 mg 1x/1 week i.m. |
84.8% 91.9% 92.5% | ||||
| Fadrozole | 1 mg 1x/day p.o. 2 mg 2x/day p.o. |
82.4% 92.6% |
Type II | ? | |
| Third | Exemestane | 25 mg 1x/day p.o. | 97.9% | Type I | 15 nM |
| Anastrozole | 1 mg 1x/day p.o. 10 mg 1x/day p.o. |
96.7–97.3% 98.1% |
Type II | 10 nM | |
| Letrozole | 0.5 mg 1x/day p.o. 2.5 mg 1x/day p.o. |
98.4% 98.9%–>99.1% |
Type II | 2.5 nM | |
| Footnotes: a = In postmenopausal women. b = Type I: Steroidal, irreversible (substrate-binding site). Type II: Nonsteroidal, reversible (binding to and interference with the cytochrome P450 heme moiety). c = In breast cancer homogenates. Sources: See template. | |||||
Pharmacokinetics
The bioavailability of anastrozole in humans is unknown, but it was found to be well-absorbed in animals.[5][2] Absorption of anastrozole is linear over a dosage range of 1 to 20 mg/day in humans and does not change with repeated administration.[6][7][2] Food does not significantly influence the extent of absorption of anastrozole.[7][2] Peak levels of anastrozole occur a median 3 hours after administration, but with a wide range of 2 to 12 hours.[5][7] Steady-state levels of anastrozole are achieved within 7 to 10 days of continuous administration, with 3.5-fold accumulation.[6][5][7] However, maximal suppression of estradiol levels occurs within 3 or 4 days of therapy.[6]
Active efflux of anastrozole by P-glycoprotein at the blood–brain barrier has been found to limit the central nervous system penetration of anastrozole in rodents, whereas this was not the case with letrozole and vorozole.[26][27][28] As such, anastrozole may have peripheral selectivity in humans, although this has yet to be confirmed.[28] In any case, estradiol is synthesized peripherally and readily crosses the blood–brain barrier, so anastrozole would still expected to reduce estradiol levels in the central nervous system to a certain degree. The plasma protein binding of anastrozole is 40%.[6][7]
The metabolism of anastrozole is by N-dealkylation, hydroxylation, and glucuronidation.[6] Inhibition of aromatase is due to anastrozole itself rather than to metabolites, with the major circulating metabolite being inactive.[2] The elimination half-life of anastrozole is 40 to 50 hours (1.7 to 2.1 days).[6][5][7] This allows for convenient once-daily administration.[7] The medication is eliminated predominantly by metabolism in the liver (83 to 85%) but also by residual excretion by the kidneys unchanged (11%).[6][5][7] Anastrozole is excreted primarily in urine but also to a lesser extent in feces.[7]
Chemistry
Anastrozole is a nonsteroidal benzyltriazole.[6][7] It is also known as α,α,α',α'-tetramethyl-5-(1H-1,2,4-triazol-1-ylmethyl)-m-benzenediacetonitrile.[29] Anastrozole is structurally related to letrozole, fadrozole, and vorozole, with all being classified as azoles.[30][31][32][33]
History
Anastrozole was patented by Imperial Chemical Industries (ICI) in 1987 and was approved for medical use, specifically the treatment of breast cancer, in 1995.[8][9]
Society and culture
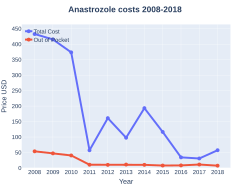
Cost
The wholesale cost in the developing world is about US$1.92–30.60 a month.[11] In the United States the wholesale cost is about $3.81 per month.[12]In 2017, it was the 258th most commonly prescribed medication in the United States, with more than one million prescriptions.[34][13]
-
Anastrozole costs (USA)
-
Anastrozole prescriptions (USA)
Available forms
Anastrozole is available in the form of 1 mg oral tablets.[2][35] No alternative forms or routes are available.[35] Anastrozole is available widely throughout the world.[29]
Names
Anastrozole is the generic name of the drug and its INN, USAN, BAN, and JAN.[29]
Anastrozole is primarily sold under the brand name Arimidex.[29] However, it is also marketed under a variety of other brand names throughout the world.[29]
Research
Anastrozole is surprisingly ineffective at treating gynecomastia, in contrast to selective estrogen receptor modulators like tamoxifen.[36][37]
Anastrozole was under development for the treatment of female infertility but did not complete development and hence was never approved for this indication.[38]
An anastrozole and levonorgestrel vaginal ring (developmental code name BAY 98-7196) was under development for use as a hormonal contraceptive and treatment for endometriosis, but development was discontinued in November 2018 and the formulation was never marketed.[39]
Anastrozole increases testosterone levels in males and has been studied as an alternative method of androgen replacement therapy in men with hypogonadism.[40][41] However, there are concerns about its long-term influence on bone mineral density in this patient population, as well as other adverse effects.[40]
References
- ↑ "anastrozole". Chemical Entities of Biological Interest (ChEBI). European Molecular Biology Laboratory. Archived from the original on 2011-09-22. Retrieved 2011-08-14.
- ↑ 2.00 2.01 2.02 2.03 2.04 2.05 2.06 2.07 2.08 2.09 2.10 2.11 2.12 2.13 "Highlights of Prescribing Information Anastrozole" (PDF). FDA. Archived (PDF) from the original on 31 July 2019. Retrieved 31 July 2019.
- ↑ 3.00 3.01 3.02 3.03 3.04 3.05 3.06 3.07 3.08 3.09 3.10 3.11 3.12 "Anastrozole". The American Society of Health-System Pharmacists. Archived from the original on 21 December 2016. Retrieved 8 December 2016.
- ↑ 4.0 4.1 "WHOCC - ATC/DDD Index". www.whocc.no. Archived from the original on 27 November 2020. Retrieved 18 September 2020.
- ↑ 5.00 5.01 5.02 5.03 5.04 5.05 5.06 5.07 5.08 5.09 5.10 5.11 5.12 Lønning P (2003). "Clinical pharmacokinetics of aromatase inhibitors and inactivators". Clinical Pharmacokinetics. 42 (7): 619–31. doi:10.2165/00003088-200342070-00002. PMID 12844324.
- ↑ 6.00 6.01 6.02 6.03 6.04 6.05 6.06 6.07 6.08 6.09 6.10 6.11 6.12 6.13 6.14 6.15 6.16 6.17 6.18 6.19 Lønning P, Pfister C, Martoni A, Zamagni C (August 2003). "Pharmacokinetics of third-generation aromatase inhibitors". Seminars in Oncology. 30 (4 Suppl 14): 23–32. doi:10.1016/S0093-7754(03)00305-1. PMID 14513434.
- ↑ 7.00 7.01 7.02 7.03 7.04 7.05 7.06 7.07 7.08 7.09 7.10 7.11 7.12 7.13 7.14 7.15 7.16 Sanford M, Plosker GL (2008). "Anastrozole: a review of its use in postmenopausal women with early-stage breast cancer". Drugs. 68 (9): 1319–40. doi:10.2165/00003495-200868090-00007. PMID 18547136.
- ↑ 8.0 8.1 Fischer, Janos; Ganellin, C. Robin (2006). Analogue-based Drug Discovery. John Wiley & Sons. p. 516. ISBN 9783527607495. Archived from the original on 2016-12-20.
- ↑ 9.0 9.1 Dukes M (1997). "The relevance of preclinical models to the treatment of postmenopausal breast cancer". Oncology. 54 Suppl 2 (2): 6–10. doi:10.1159/000227748. PMID 9394853.
- ↑ World Health Organization (2019). World Health Organization model list of essential medicines: 21st list 2019. Geneva: World Health Organization. hdl:10665/325771. WHO/MVP/EMP/IAU/2019.06. License: CC BY-NC-SA 3.0 IGO.
- ↑ 11.0 11.1 "Anastrozole". International Drug Price Indicator Guide. Archived from the original on 22 January 2018. Retrieved 8 December 2016.
- ↑ 12.0 12.1 12.2 "NADAC as of 2016-12-07 | Data.Medicaid.gov". Centers for Medicare and Medicaid Services. Archived from the original on 21 December 2016. Retrieved 18 December 2016. Cite error: Invalid
<ref>tag; name "ref" defined multiple times with different content - ↑ 13.0 13.1 "Anastrozole - Drug Usage Statistics". ClinCalc. Archived from the original on 5 July 2020. Retrieved 11 April 2020.
- ↑ 14.0 14.1 14.2 Howell A, Cuzick J, Baum M, Buzdar A, Dowsett M, Forbes JF, et al. (2005). "Results of the ATAC (Arimidex, Tamoxifen, Alone or in Combination) trial after completion of 5 years' adjuvant treatment for breast cancer". Lancet. 365 (9453): 60–2. doi:10.1016/S0140-6736(04)17666-6. PMID 15639680.[non-primary source needed]
- ↑ "Casodex® (bicalutamide) Tablets" (PDF). FDA. Archived (PDF) from the original on 30 July 2019.
- ↑ Schoelwer M, Eugster EA (2015). "Treatment of Peripheral Precocious Puberty". Puberty from Bench to Clinic. Endocrine Development. Vol. 29. pp. 230–9. doi:10.1159/000438895. ISBN 978-3-318-02788-4. PMC 5345994. PMID 26680582.
- ↑ Zacharin M (May 2019). "Disorders of Puberty: Pharmacotherapeutic Strategies for Management". Handbook of Experimental Pharmacology. doi:10.1007/164_2019_208. PMID 31144045.
- ↑ Kliegman, Robert M.; Stanton, Bonita; St. Geme, Joseph; Schor, Nina F (17 April 2015). Nelson Textbook of Pediatrics. Elsevier Health Sciences. pp. 2661–. ISBN 978-0-323-26352-8. Archived from the original on 27 July 2020. Retrieved 31 July 2019.
- ↑ Neyman A, Eugster EA (December 2017). "Treatment of Girls and Boys with McCune-Albright Syndrome with Precocious Puberty - Update 2017". Pediatric Endocrinology Reviews. 15 (2): 136–141. doi:10.17458/per.vol15.2017.nau.treatmentgirlsboys. PMC 5808444. PMID 29292624.
- ↑ Haddad, Nadine G.; Eugster, Erica A. (2012). "Peripheral Precocious Puberty: Interventions to Improve Growth". Handbook of Growth and Growth Monitoring in Health and Disease. pp. 1199–1212. doi:10.1007/978-1-4419-1795-9_71. ISBN 978-1-4419-1794-2.
- ↑ Haddad NG, Eugster EA (April 2019). "Peripheral precocious puberty including congenital adrenal hyperplasia: causes, consequences, management and outcomes". Best Practice & Research. Clinical Endocrinology & Metabolism. 33 (3): 101273. doi:10.1016/j.beem.2019.04.007. hdl:1805/19111. PMID 31027974.
- ↑ Misra, Madhusmita; Radovick, Sally (2018). "Precocious Puberty". Pediatric Endocrinology. pp. 589–615. doi:10.1007/978-3-319-73782-9_26. ISBN 978-3-319-73781-2.
- ↑ Mauras N (October 2011). "Strategies for maximizing growth in puberty in children with short stature". Pediatric Clinics of North America. 58 (5): 1167–79, x. doi:10.1016/j.pcl.2011.07.007. PMID 21981954.
- ↑ Fuqua JS (June 2013). "Treatment and outcomes of precocious puberty: an update". The Journal of Clinical Endocrinology and Metabolism. 98 (6): 2198–207. doi:10.1210/jc.2013-1024. PMID 23515450.
- ↑ Simpson ER (September 2003). "Sources of estrogen and their importance". The Journal of Steroid Biochemistry and Molecular Biology. 86 (3–5): 225–30. doi:10.1016/S0960-0760(03)00360-1. PMID 14623515.
- ↑ Costa R, Carneiro BA, Wainwright DA, Santa-Maria CA, Kumthekar P, Chae YK, et al. (January 2017). "Developmental therapeutics for patients with breast cancer and central nervous system metastasis: current landscape and future perspectives". Annals of Oncology. 28 (1): 44–56. doi:10.1093/annonc/mdw532. PMID 28177431.
- ↑ Russell N, Cheung A, Grossmann M (August 2017). "Estradiol for the mitigation of adverse effects of androgen deprivation therapy". Endocrine-Related Cancer. 24 (8): R297–R313. doi:10.1530/ERC-17-0153. PMID 28667081.
- ↑ 28.0 28.1 Miyajima M, Kusuhara H, Takahashi K, Takashima T, Hosoya T, Watanabe Y, Sugiyama Y (September 2013). "Investigation of the effect of active efflux at the blood-brain barrier on the distribution of nonsteroidal aromatase inhibitors in the central nervous system". Journal of Pharmaceutical Sciences. 102 (9): 3309–19. doi:10.1002/jps.23600. PMID 23712697.
- ↑ 29.0 29.1 29.2 29.3 29.4 "Anastrozole". Archived from the original on 2019-07-31. Retrieved 2019-07-31.
- ↑ Thurston, David E. (22 November 2006). Chemistry and Pharmacology of Anticancer Drugs. CRC Press. pp. 136–. ISBN 978-1-4200-0890-6. Archived from the original on 27 August 2021. Retrieved 31 July 2019.
- ↑ Roy, Kunal (28 February 2015). Quantitative Structure-Activity Relationships in Drug Design, Predictive Toxicology, and Risk Assessment. IGI Global. pp. 437–. ISBN 978-1-4666-8137-8. Archived from the original on 27 August 2021. Retrieved 31 July 2019.
- ↑ Smith, Dennis A.; Allerton, Charlotte; Kalgutkar, Amit S.; van de Waterbeemd, Han; Walker, Don K. (13 September 2012). Pharmacokinetics and Metabolism in Drug Design. John Wiley & Sons. pp. 197–. ISBN 978-3-527-64529-9. Archived from the original on 27 August 2021. Retrieved 31 July 2019.
- ↑ Environmental Health Perspectives: Supplements. U.S. Department of Health and Human Services, Public Health Service, National Institutes of Health, National Institute of Environmental Health Sciences. 1993. pp. 256–260. Archived from the original on 2021-08-27. Retrieved 2019-07-31.
- ↑ Cite error: Invalid
<ref>tag; no text was provided for refs namedref1 - ↑ 35.0 35.1 White, Rebecca; Bradnam, Vicky (11 March 2015). Handbook of Drug Administration via Enteral Feeding Tubes, 3rd edition. Pharmaceutical Press. pp. 108–. ISBN 978-0-85711-162-3. Archived from the original on 10 July 2020. Retrieved 31 July 2019.
- ↑ Fagerlund A, Cormio L, Palangi L, Lewin R, Santanelli di Pompeo F, Elander A, Selvaggi G (2015). "Gynecomastia in Patients with Prostate Cancer: A Systematic Review". PLOS One. 10 (8): e0136094. Bibcode:2015PLoSO..1036094F. doi:10.1371/journal.pone.0136094. PMC 4550398. PMID 26308532.
- ↑ Bedognetti D, Rubagotti A, Zoppoli G, Boccardo F (2010). "Gynaecomastia: the anastrozole paradox". Journal of Pediatric Endocrinology & Metabolism. 23 (1–2): 205–6. doi:10.1515/JPEM.2010.23.1-2.205. PMID 20432826.
- ↑ "Anastrozole - AstraZeneca". Adis Insight. Archived from the original on 2019-07-31. Retrieved 2019-07-31.
- ↑ "Anastrozole/levonorgestrel intravaginal ring - Bayer HealthCare". Adis Insight. Archived from the original on 2021-08-27. Retrieved 2019-07-31.
- ↑ 40.0 40.1 Serefoglu EC, Gokce A, Hellstrom WJ, Guay AT (2013). "Alternate Therapies for Testosterone Replacement". Androgen Deficiency and Testosterone Replacement. pp. 141–147. doi:10.1007/978-1-62703-179-0_11. ISBN 978-1-62703-178-3.
- ↑ Khera M, Adaikan G, Buvat J, Carrier S, El-Meliegy A, Hatzimouratidis K, et al. (December 2016). "Diagnosis and Treatment of Testosterone Deficiency: Recommendations From the Fourth International Consultation for Sexual Medicine (ICSM 2015)". The Journal of Sexual Medicine. 13 (12): 1787–1804. doi:10.1016/j.jsxm.2016.10.009. PMID 27914560.
External links
| External sites: | |
|---|---|
| Identifiers: |
|
- Pages using duplicate arguments in template calls
- Pages with reference errors
- CS1: long volume value
- All pages needing factual verification
- Wikipedia articles needing factual verification from June 2018
- Articles with invalid date parameter in template
- Chemical articles with unknown parameter in Infobox drug
- Chemical articles without CAS registry number
- Articles without EBI source
- Chemical pages without ChemSpiderID
- Chemical pages without DrugBank identifier
- Articles without KEGG source
- Articles without UNII source
- Drugs missing an ATC code
- 27-Hydroxylase inhibitors
- Aromatase inhibitors
- AstraZeneca brands
- Hormonal antineoplastic drugs
- Nitriles
- Peripherally selective drugs
- Triazoles
- World Anti-Doping Agency prohibited substances
- World Health Organization essential medicines (alternatives)
- RTT