Interleukin 23
| IL12B | |||||||
|---|---|---|---|---|---|---|---|
 Crystal structure of IL-12B | |||||||
| Identifiers | |||||||
| Symbol | IL12B | ||||||
| Alt. symbols | CLMF2, NKSF2, p40 | ||||||
| NCBI gene | 3593 | ||||||
| HGNC | 5970 | ||||||
| OMIM | 161561 | ||||||
| PDB | 1F42 | ||||||
| RefSeq | NM_002187 | ||||||
| UniProt | P29460 | ||||||
| Other data | |||||||
| Locus | Chr. 5 q31.1-33.1 | ||||||
| |||||||
| interleukin 23, alpha subunit p19 | |||||||
|---|---|---|---|---|---|---|---|
| Identifiers | |||||||
| Symbol | IL23A | ||||||
| NCBI gene | 51561 | ||||||
| HGNC | 15488 | ||||||
| RefSeq | NM_016584 | ||||||
| UniProt | Q9NPF7 | ||||||
| Other data | |||||||
| Locus | Chr. 12 q13.13 | ||||||
| |||||||
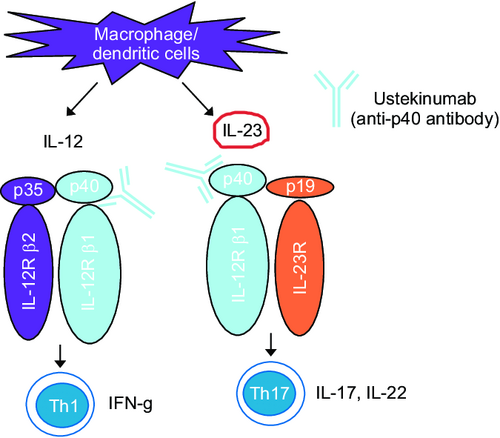
Interleukin 23 (IL-23) is a heterodimeric cytokine composed of an IL-12B (IL-12p40) subunit (which is shared with IL-12) and an IL-23A (IL-23p19) subunit.[1] IL-23 is part of the IL-12 family of cytokines.[2] The functional receptor for IL-23 (the IL-23 receptor) consists of a heterodimer between IL-12Rβ1 and IL-23R.[3]
Function
IL-23 is an inflammatory cytokine. It has been shown to be a key cytokine for T helper type 17 cell (Th17 cell) maintenance and expansion. Polarisation to a Th17 phenotype is triggered by IL-6 and TGF-β, which activate the Th17 transcription factor RORγt. IL-23 stabilises RORγt and thus enables Th17 cells to release their effector cytokines, such as IL-17, IL-21, IL-22 and GM-CSF, which mediate protection against extracellular fungi and bacteria and participate in barrier immunity.[4] Effects similar to those IL-23 has on Th17 cells were described for type 3 innate lymphoid cells, which actively secrete Th17 cytokines upon IL-23 stimulation.[5] Natural killer cells also express the IL-23 receptor. They respond with increased interferon-γ secretion and enhanced antibody-dependent cellular cytotoxicity. IL-23 also induces proliferation of CD4 memory T cells (but not naïve T cells).[6] Besides its proinflammatory effects, IL-23 promotes angiogenesis.[7]
IL-23 is mainly secreted by activated dendritic cells, macrophages or monocytes. Innate lymphoid cells and γδ T cells also produce IL-23.[2] B cells produce IL-23 through B cell antigen receptor signaling.[8] Secretion is stimulated by an antigen stimulus recognised by a pattern recognition receptor.[9] IL-23 imbalance and increase is associated with autoimmune diseases and cancer. It is thus a target for therapeutic research.[4] IL-23 expression by dendritic cells is further induced by thymic stromal lymphopoietin, a proallergic cytokine expressed by keratinocytes that is elevated in psoriatic lesions.[10] In the pathogenesis of psoriasis, dermal dendritic cells are stimulated to release IL-23 by nociceptive neurons.[11] IL-23 is also elevated during bacterial meningitis, leading to epithelial dysregulation and inflammation.[12]
Mycobacterium avium subspecies paratuberculosis-stimulated monocyte-derived macrophages are one of the contributors of IL-23, and thus cattle with Johne's disease have elevated IL-23.[13]
Prior to the discovery of IL-23, IL-12 had been proposed to represent a key mediator of inflammation in mouse models of inflammation.[14] However, many studies aimed at assessing the role of IL-12 by pharmacological blockade had targeted IL-12B, and were therefore not as specific as thought. Studies which blocked the function of IL-12A did not produce the same results as those targeting IL-12B, as would have been expected if both subunits formed part of IL-12 only.[15]
The discovery of an additional potential binding partner for IL-12B led to a reassessment of this role for IL-12. Studies in experimental autoimmune encephalomyelitis, a mouse model of multiple sclerosis, showed that IL-23 was responsible for the inflammation observed, not IL-12 as previously thought.[16] Subsequently, IL-23 was shown to facilitate development of inflammation in numerous other models of immune pathology where IL-12 had previously been implicated, including models of arthritis,[17] intestinal inflammation,[18][19][20] and psoriasis.[21] Low concentrations of IL-23 support lung tumor growth whereas high concentrations inhibit proliferation of lung cancer cells.[22] IL-23 and IL-23R were identified in serum from patients with non-small-cell lung cancer and have been proposed as prognostic serum markers.[23] IL-23 can also promote progression of cardiovascular diseases such as atherosclerosis, hypertension, aortic dissection, cardiac hypertrophy, myocardial infarction and acute cardiac injury[citation needed]. In brain, IL-23 is able to activate γδ T cells to increase their expression of IL-17, which contributes to the inflammatory response and thus plays a key role in secondary brain injury after spontaneous intracerebral hemorrhage.[24]
Signalling
The IL-23 heterodimer binds the receptor complex: the p19 subunit binds IL-23R while the p40 subunit binds IL-12RB1. Receptor binding leads to recruitment of Janus kinase 2 and Tyrosine kinase 2 kinases. Janus kinase 2 and Tyrosine kinase 2 transduce the signal and phosphorylate STAT3 and STAT4. STATs dimerise and activate transcription of target genes in nucleus. STAT3 is responsible for key Th17 development attributes such as RORγt expression and transcription of Th17 cytokines.[4]
Therapeutic targets
Monoclonal antibody drugs
IL-23 is one of the therapeutic targets to treat the inflammatory diseases.[26]
Ustekinumab, a monoclonal antibody directed against this cytokine, is used to treat certain autoimmune conditions.[27]
Guselkumab is another monoclonal antibody against IL-23. Blocking IL-23 can slow clinical manifestation of psoriasis, indirectly affecting Th17 immune response and production of IL-17.[28]
Ixekizumab, an IL-17A antagonist, has been reported to have faster onset of action in treatment of psoriasis than guselkumab, tildrakizumab or risankizumab, which are inhibitors of the p19 subunit of IL-23.[29]
However, risankizumab has been shown to have the best treatment results for psoriasis in comparison with other IL-23 inhibitors.[30]
Adnectin-2 binds to IL-23 and competes with IL-23–IL-23R binding.[26]
Discovery
IL-23 was first described by Robert Kastelein and colleagues at the DNAX research institute using a combination of computational, biochemical and cellular immunology approaches.[1]
References
- ↑ 1.0 1.1 Oppmann B, Lesley R, Blom B, Timans JC, Xu Y, Hunte B, et al. (November 2000). "Novel p19 protein engages IL-12p40 to form a cytokine, IL-23, with biological activities similar as well as distinct from IL-12". Immunity. 13 (5): 715–25. doi:10.1016/S1074-7613(00)00070-4. PMID 11114383.
- ↑ 2.0 2.1 Cauli A, Piga M, Floris A, Mathieu A (2015-10-01). "Current perspective on the role of the interleukin-23/interleukin-17 axis in inflammation and disease (chronic arthritis and psoriasis)". ImmunoTargets and Therapy. 4: 185–90. doi:10.2147/ITT.S62870. PMC 4918258. PMID 27471723.
- ↑ Parham C, Chirica M, Timans J, Vaisberg E, Travis M, Cheung J, et al. (June 2002). "A receptor for the heterodimeric cytokine IL-23 is composed of IL-12Rbeta1 and a novel cytokine receptor subunit, IL-23R". Journal of Immunology. 168 (11): 5699–708. doi:10.4049/jimmunol.168.11.5699. PMID 12023369.
- ↑ 4.0 4.1 4.2 Tang C, Chen S, Qian H, Huang W (February 2012). "Interleukin-23: as a drug target for autoimmune inflammatory diseases". Immunology. 135 (2): 112–24. doi:10.1111/j.1365-2567.2011.03522.x. PMC 3277713. PMID 22044352.
- ↑ Zeng B, Shi S, Ashworth G, Dong C, Liu J, Xing F (April 2019). "ILC3 function as a double-edged sword in inflammatory bowel diseases". Cell Death & Disease. 10 (4): 315. doi:10.1038/s41419-019-1540-2. PMC 6453898. PMID 30962426.
- ↑ Li Y, Wang H, Lu H, Hua S (2016). "Regulation of Memory T Cells by Interleukin-23". International Archives of Allergy and Immunology. 169 (3): 157–62. doi:10.1159/000445834. PMID 27100864. S2CID 24274565.
- ↑ Langowski JL, Zhang X, Wu L, Mattson JD, Chen T, Smith K, et al. (July 2006). "IL-23 promotes tumour incidence and growth". Nature. 442 (7101): 461–5. Bibcode:2006Natur.442..461L. doi:10.1038/nature04808. PMID 16688182. S2CID 4431794.
- ↑ Gagro A, Servis D, Cepika AM, Toellner KM, Grafton G, Taylor DR, et al. (May 2006). "Type I cytokine profiles of human naïve and memory B lymphocytes: a potential for memory cells to impact polarization". Immunology. 118 (1): 66–77. doi:10.1111/j.1365-2567.2006.02342.x. PMC 1782263. PMID 16630024.
- ↑ Re F, Strominger JL (October 2001). "Toll-like receptor 2 (TLR2) and TLR4 differentially activate human dendritic cells". The Journal of Biological Chemistry. 276 (40): 37692–9. doi:10.1074/jbc.M105927200. PMID 11477091.
- ↑ Volpe E, Pattarini L, Martinez-Cingolani C, Meller S, Donnadieu MH, Bogiatzi SI, et al. (August 2014). "Thymic stromal lymphopoietin links keratinocytes and dendritic cell-derived IL-23 in patients with psoriasis". The Journal of Allergy and Clinical Immunology. 134 (2): 373–81. doi:10.1016/j.jaci.2014.04.022. PMID 24910175.
- ↑ Riol-Blanco L, Ordovas-Montanes J, Perro M, Naval E, Thiriot A, Alvarez D, et al. (June 2014). "Nociceptive sensory neurons drive interleukin-23-mediated psoriasiform skin inflammation". Nature. 510 (7503): 157–61. Bibcode:2014Natur.510..157R. doi:10.1038/nature13199. PMC 4127885. PMID 24759321.
- ↑ Srinivasan L, Kilpatrick L, Shah SS, Abbasi S, Harris MC (2018-02-02). "Elevations of novel cytokines in bacterial meningitis in infants". PLOS ONE. 13 (2): e0181449. Bibcode:2018PLoSO..1381449S. doi:10.1371/journal.pone.0181449. PMC 5796685. PMID 29394248.
- ↑ DeKuiper JL, Cooperider HE, Lubben N, Ancel CM, Coussens PM (2020). "paratuberculosis Drives an Innate Th17-Like T Cell Response Regardless of the Presence of Antigen-Presenting Cells". Frontiers in Veterinary Science. 7: 108. doi:10.3389/fvets.2020.00108. PMC 7089878. PMID 32258066.
- ↑ Leonard JP, Waldburger KE, Goldman SJ (January 1995). "Prevention of experimental autoimmune encephalomyelitis by antibodies against interleukin 12". The Journal of Experimental Medicine. 181 (1): 381–6. doi:10.1084/jem.181.1.381. PMC 2191822. PMID 7528773.
- ↑ Becher B, Durell BG, Noelle RJ (August 2002). "Experimental autoimmune encephalitis and inflammation in the absence of interleukin-12". The Journal of Clinical Investigation. 110 (4): 493–7. doi:10.1172/JCI15751. PMC 150420. PMID 12189243.
- ↑ Cua DJ, Sherlock J, Chen Y, Murphy CA, Joyce B, Seymour B, et al. (February 2003). "Interleukin-23 rather than interleukin-12 is the critical cytokine for autoimmune inflammation of the brain". Nature. 421 (6924): 744–8. Bibcode:2003Natur.421..744C. doi:10.1038/nature01355. PMID 12610626. S2CID 4380302.
- ↑ Murphy CA, Langrish CL, Chen Y, Blumenschein W, McClanahan T, Kastelein RA, et al. (December 2003). "Divergent pro- and antiinflammatory roles for IL-23 and IL-12 in joint autoimmune inflammation". The Journal of Experimental Medicine. 198 (12): 1951–7. doi:10.1084/jem.20030896. PMC 2194162. PMID 14662908.
- ↑ Yen D, Cheung J, Scheerens H, Poulet F, McClanahan T, McKenzie B, et al. (May 2006). "IL-23 is essential for T cell-mediated colitis and promotes inflammation via IL-17 and IL-6". The Journal of Clinical Investigation. 116 (5): 1310–6. doi:10.1172/JCI21404. PMC 1451201. PMID 16670770.
- ↑ Kullberg MC, Jankovic D, Feng CG, Hue S, Gorelick PL, McKenzie BS, et al. (October 2006). "IL-23 plays a key role in Helicobacter hepaticus-induced T cell-dependent colitis". The Journal of Experimental Medicine. 203 (11): 2485–94. doi:10.1084/jem.20061082. PMC 2118119. PMID 17030948.
- ↑ Hue S, Ahern P, Buonocore S, Kullberg MC, Cua DJ, McKenzie BS, et al. (October 2006). "Interleukin-23 drives innate and T cell-mediated intestinal inflammation". The Journal of Experimental Medicine. 203 (11): 2473–83. doi:10.1084/jem.20061099. PMC 2118132. PMID 17030949.
- ↑ Chan JR, Blumenschein W, Murphy E, Diveu C, Wiekowski M, Abbondanzo S, et al. (November 2006). "IL-23 stimulates epidermal hyperplasia via TNF and IL-20R2-dependent mechanisms with implications for psoriasis pathogenesis". The Journal of Experimental Medicine. 203 (12): 2577–87. doi:10.1084/jem.20060244. PMC 2118145. PMID 17074928.
- ↑ Li J, Zhang L, Zhang J, Wei Y, Li K, Huang L, et al. (March 2013). "Interleukin 23 regulates proliferation of lung cancer cells in a concentration-dependent way in association with the interleukin-23 receptor". Carcinogenesis. 34 (3): 658–66. doi:10.1093/carcin/bgs384. PMID 23250909.
- ↑ Liu D, Xing S, Wang W, Huang X, Lin H, Chen Y, et al. (April 2020). "Prognostic value of serum soluble interleukin-23 receptor and related T-helper 17 cell cytokines in non-small cell lung carcinoma". Cancer Science. 111 (4): 1093–1102. doi:10.1111/cas.14343. PMC 7156824. PMID 32020720.
- ↑ Zhu H, Wang Z, Yu J, Yang X, He F, Liu Z, Che F, Chen X, Ren H, Hong M, Wang J (March 2019). "Role and mechanisms of cytokines in the secondary brain injury after intracerebral hemorrhage". Prog. Neurobiol. 178: 101610. doi:10.1016/j.pneurobio.2019.03.003. PMID 30923023. S2CID 85495400.
- ↑ Nakase, Hiroshi (15 January 2020). "Optimizing the Use of Current Treatments and Emerging Therapeutic Approaches to Achieve Therapeutic Success in Patients with Inflammatory Bowel Disease". Gut and Liver. 14 (1): 7–19. doi:10.5009/gnl18203.
- ↑ 26.0 26.1 Iacob RE, Krystek SR, Huang RY, Wei H, Tao L, Lin Z, et al. (April 2015). "Hydrogen/deuterium exchange mass spectrometry applied to IL-23 interaction characteristics: potential impact for therapeutics". Expert Review of Proteomics. 12 (2): 159–69. doi:10.1586/14789450.2015.1018897. PMC 4409866. PMID 25711416.
- ↑ Cingoz O (2009). "Ustekinumab". mAbs. 1 (3): 216–21. doi:10.4161/mabs.1.3.8593. PMC 2726595. PMID 20069753.
- ↑ Warren RB, Gooderham M, Burge R, Zhu B, Amato D, Liu KH, et al. (May 2020). "Comparison of cumulative clinical benefits of biologics for the treatment of psoriasis over 16 weeks: Results from a network meta-analysis". Journal of the American Academy of Dermatology. 82 (5): 1138–1149. doi:10.1016/j.jaad.2019.12.038. PMID 31884091.
- ↑ Gottlieb AB, Saure D, Wilhelm S, Dossenbach M, Schuster C, Smith SD, et al. (April 2020). "Indirect comparisons of ixekizumab versus three interleukin-23 p19 inhibitors in patients with moderate-to-severe plaque psoriasis - efficacy findings up to week 12". The Journal of Dermatological Treatment. 33 (1): 54–61. doi:10.1080/09546634.2020.1747592. PMID 32299269.
- ↑ Yasmeen N, Sawyer LM, Malottki K, Levin LÅ, Didriksen Apol E, Jemec GB (April 2020). "Targeted therapies for patients with moderate-to-severe psoriasis: a systematic review and network meta-analysis of PASI response at 1 year". The Journal of Dermatological Treatment. 33 (1): 204–218. doi:10.1080/09546634.2020.1743811. PMID 32202445.