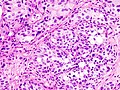
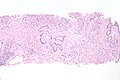
Urothelial carcinoma
| Urothelial carcinoma | |
|---|---|
| Other names: Transitional cell carcinoma (TCC) | |
 | |
| Histopathology of transitional carcinoma of the urinary bladder. Transurethral biopsy. Hematoxylin and eosin stain. | |
| Specialty | Oncology, urology |
| Symptoms | Blood in the urine, back pain, weight loss, pain with urination[1] |
| Complications | Spread to other parts of the body[1] |
| Types | Upper tract, urinary bladder[2] |
| Risk factors | Smoking, aromatic amines, arsenic, aristolochic acid, Lynch syndrome[3][4] |
| Diagnostic method | Cystoscopy and biopsy[5] |
| Treatment | Surgery, chemotherapy, laser surgery[1] |
| Prognosis | Good if found early[6] |
| Frequency | Common[3] |
Urothelial carcinoma, previously called transitional cell carcinoma, is a type of cancer that can occur anywhere along the urinary tract.[7][8] This includes the kidneys, ureters, bladder, prostate, and urethra.[7][9] Symptoms may include blood in the urine, back pain, weight loss, or pain with urination.[1]
Risk factors include smoking, aromatic amines, arsenic, aristolochic acid, Schistosoma haematobium, and genetic disorders such as Lynch syndrome.[3][4][2] They develop from the transitional epithelium.[3] The diagnosis is generally confirmed by cystoscopy and biopsy.[5]
Treatment often include surgery.[1] Other options may include chemotherapy or laser surgery.[1] If the cancer remains localized a cure if often possible.[6] Multiple areas of the urinary tract; however, may be affected and there is a risk of reoccurrence.[3] Spread may also occur to lymph nodes, lung, liver, or bone.[1]
Urothelial carcinomas make up about 12.5% of cancer cases.[3] Males are more commonly affected than females.[2] They are the most common cancer of the urinary tract,[10] representing more than 90% of bladder cancers and about 7% of kidney cancers.[6][3] They are the second most common reason for death from genitourinary cancers.[10]
Signs and symptoms
Symptoms of transitional cell carcinomas depend on the location and extent of the cancer.[citation needed]
Transitional cell carcinomas can also affect the prostate.[11][12]
Causes
Urothelial carcinoma is a prototypical example of a malignancy arising from environmental carcinogenic influences. By far the most important cause is cigarette smoking, which contributes to approximately one-half of the disease burden. Chemical exposure, such as those sustained by workers in the petroleum industry, the manufacture of paints and pigments (e.g., aniline dyes), and agrochemicals are known to predispose one to urothelial cancer. The risk is lowered by increased liquid consumption, presumably as a consequence of increased urine production and thus less dwell time on the urothelial surface. Conversely, risk is increased among long-haul truck drivers and others in whom long urine dwell-times are encountered. As with most epithelial cancers, physical irritation has been associated with increased risk of malignant transformation of the urothelium. Thus, urothelial carcinomas are more common in the context of chronic urinary stone disease, chronic catheterization (as in patients with paraplegia or multiple sclerosis), and chronic infections. Some particular examples are listed below:
- Certain drugs, such as cyclophosphamide, via the metabolites acrolein and phenacetin, may predispose to the development of transitional cell carcinomas (the latter especially with respect to the upper urinary tract).[13]
- Radiation exposure
- Somatic mutation, such as deletion of chromosome 9q, 9p, 11p, 17p, 13q, 14q and overexpression of RAS (oncogene) and epidermal growth factor receptor (EGFR).
Pathology
Transitional cell carcinomas are often multifocal, with 30–40% of patients having more than one tumor at diagnosis. The pattern of growth of transitional cell carcinomas can be papillary, sessile, or carcinoma in situ. The most common site of transitional cell carcinoma metastasis outside the pelvis is bone (35%); of these, 40 percent are in the spine.[14]
Diagnosis

Transitional refers to the histological subtype of the cancerous cells as seen under a microscope.
Classification
The 1973 WHO grading system for transitional cell carcinomas (papilloma, G1, G2 or G3) is most commonly used despite being superseded by the 2004 WHO[15] grading (papillary neoplasm of low malignant potential [PNLMP], low grade, and high grade papillary carcinoma).
-
Histopathology of urothelial carcinoma of the urinary bladder, showing a nested pattern of invasion. Transurethral biopsy. Hematoxylin and eosin.
-
Histopathology of urothelial carcinoma of the urinary bladder.
-
Histopathology of urothelial carcinoma of the urinary bladder.
-
Micrograph of urethral urothelial cell carcinoma. Hematoxylin and eosin stain.
Treatment
Localized/early
Transitional cell carcinomas can be very difficult to treat. Treatment for localized stage transitional cell carcinomas is surgical resection of the tumor, but recurrence is common. Some people are given mitomycin into the bladder either as a one-off dose in the immediate post-operative period (within 24 hrs) or a few weeks after the surgery as a six dose regimen.
Localized/early transitional cell carcinomas can also be treated with infusions of Bacille Calmette–Guérin into the bladder. These are given weekly for either 6 weeks (induction course) or 3 weeks (maintenance/booster dose). Side effects include a small chance of developing systemic tuberculosis or the patient becoming sensitized to BCG, causing severe intolerance and a possible reduction in bladder volume due to scarring.
In people with evidence of early muscular invasion, radical curative surgery in the form of a cysto-prostatectomy usually with lymph node sampling can also be performed. In such patients, a bowel loop is often used to create either a "neo-bladder" or an "ileal conduit" which act as a place for the storage of urine before it is evacuated from the body either via the urethra or a urostomy respectively.
Advanced/metastatic
First-line chemotherapy regimens for advanced or metastatic transitional cell carcinomas consists of gemcitabine and cisplatin) or a combination of methotrexate, vinblastine, adriamycin, and cisplatin.[16]
Taxanes or vinflunine have been used as second-line therapy (after progression on a platinum containing chemotherapy).[17]
Immunotherapy such as pembrolizumab is often used as second-line therapy for metastatic urothelial carcinoma that has progressed despite treatment with GC or MVAC.[18]
In May 2016 FDA granted accelerated approval to atezolizumab for locally advanced or metastatic urothelial carcinoma treatment after failure of cisplatin-based chemotherapy.[19] The confirmatory trial (to convert the accelerated approval into a full approval) failed to achieve its primary endpoint of overall survival.[20]
See also
References
- ↑ 1.0 1.1 1.2 1.3 1.4 1.5 1.6 "Transitional Cell Cancer of the Renal Pelvis and Ureter Treatment (PDQ®)–Patient Version - National Cancer Institute". www.cancer.gov. 20 February 2004. Archived from the original on 8 June 2019. Retrieved 26 January 2021.
- ↑ 2.0 2.1 2.2 Miyazaki, J; Nishiyama, H (October 2017). "Epidemiology of urothelial carcinoma". International journal of urology : official journal of the Japanese Urological Association. 24 (10): 730–734. doi:10.1111/iju.13376. PMID 28543959.
- ↑ 3.0 3.1 3.2 3.3 3.4 3.5 3.6 Wild, Christopher (2020). "5.15". World Cancer Report: Cancer Research for Cancer Prevention (pdf). Lyon: International Agency for Research. pp. 439–445. ISBN 978-92-832-0448-0. Archived from the original on 2021-01-26. Retrieved 2021-01-26.
- ↑ 4.0 4.1 Cancer, International Agency for Research on (2004). Pathology and Genetics of Tumours of the Urinary System and Male Genital Organs. IARC. p. 93. ISBN 978-92-832-2415-0. Archived from the original on 2021-08-27. Retrieved 2021-01-26.
- ↑ 5.0 5.1 Shariat, Shahrokh F.; Xylinas, Evanguelos (2014). Upper Tract Urothelial Carcinoma. Springer. p. 64. ISBN 978-1-4939-1501-9. Archived from the original on 2021-08-27. Retrieved 2021-01-26.
- ↑ 6.0 6.1 6.2 "Transitional Cell Cancer of the Renal Pelvis and Ureter Treatment (PDQ®)–Health Professional Version - National Cancer Institute". www.cancer.gov. 11 May 2020. Archived from the original on 25 January 2021. Retrieved 26 January 2021.
- ↑ 7.0 7.1 Lee, Eugene K.; Dickstein, Rian J.; Kamat, Ashish M. (June 2011). "Imaging of Urothelial Cancers: What the Urologist Needs to Know". American Journal of Roentgenology. 196 (6): 1249–1254. doi:10.2214/AJR.10.6232. PMID 21606285.
- ↑ Kaseb, H; Aeddula, NR (January 2020). "Bladder Cancer". PMID 30725608.
{{cite journal}}: Cite journal requires|journal=(help) - ↑ Palou, J; Baniel, J; Klotz, L; Wood, D; Cookson, M; Lerner, S; Horie, S; Schoenberg, M; Angulo, J; Bassi, P (January 2007). "Urothelial carcinoma of the prostate". Urology. 69 (1 Suppl): 50–61. doi:10.1016/j.urology.2006.05.059. PMID 17280908.
- ↑ 10.0 10.1 McDougal, W. Scott; Wein, Alan J.; Kavoussi, Louis R.; Novick, Andrew C.; Partin, Alan W.; Peters, Craig A.; Ramchandani, Parvati (2011). Campbell-Walsh Urology 10th Edition Review E-Book. Elsevier Health Sciences. p. 437. ISBN 978-1-4557-2317-1. Archived from the original on 2021-08-27. Retrieved 2021-01-26.
- ↑ Walsh DL, Chang SS (2009). "Dilemmas in the treatment of urothelial cancers of the prostate". Urologic Oncology. 27 (4): 352–7. doi:10.1016/j.urolonc.2007.12.010. PMID 18439852.
- ↑ Njinou Ngninkeu B, Lorge F, Moulin P, Jamart J, Van Cangh PJ (January 2003). "Transitional cell carcinoma involving the prostate: a clinicopathological retrospective study of 76 cases". The Journal of Urology. 169 (1): 149–52. doi:10.1097/01.ju.0000042810.43380.36. PMID 12478124.
- ↑ Colin P, Koenig P, Ouzzane A, Berthon N, Villers A, Biserte J, Roupret M (November 2009). "Environmental factors involved in carcinogenesis of urothelial cell carcinomas of the upper urinary tract". BJU International. 104 (10): 1436–40. doi:10.1111/j.1464-410X.2009.08838.x. PMID 19689473.
- ↑ Punyavoravut V, Nelson SD (August 1999). "Diffuse bony metastasis from transitional cell carcinoma of urinary bladder: a case report and review of literature". Journal of the Medical Association of Thailand. 82 (8): 839–43. PMID 10511795.
- ↑ Sauter G, Algaba F, Amin MB, Busch C, Cheville J, Gasser T, Grignon D, Hofstaedter F, Lopez-Beltran A, Epstein JI. Noninvasive urothelial neoplasias: WHO classification of noninvasive papillary urothelial tumors. In World Health Organization classification of tumors. Pathology and genetics of tumors of the urinary system and male genital organs. Eble JN, Epstein JI, Sesterhenn I (eds): Lyon, IARCC Press, p. 110, 2004
- ↑ von der Maase, H; Hansen, SW; Roberts, JT; Dogliotti, L; Oliver, T; Moore, MJ; Bodrogi, I; Albers, P; Knuth, A; Lippert, CM; Kerbrat, P; Sanchez Rovira, P; Wersall, P; Cleall, SP; Roychowdhury, DF; Tomlin, I; Visseren-Grul, CM; Conte, PF (September 2000). "Gemcitabine and cisplatin versus methotrexate, vinblastine, doxorubicin, and cisplatin in advanced or metastatic bladder cancer: results of a large, randomized, multinational, multicenter, phase III study". Journal of Clinical Oncology. 18 (17): 3068–77. doi:10.1200/jco.2000.18.17.3068. PMID 11001674.
- ↑ "Immunotherapy Proceeds to Change Bladder Cancer Treatment 2017". Archived from the original on 2017-12-01. Retrieved 2017-11-22.
- ↑ Syn, Nicholas L; Teng, Michele W L; Mok, Tony S K; Soo, Ross A (2017). "De-novo and acquired resistance to immune checkpoint targeting". The Lancet Oncology. 18 (12): e731–e741. doi:10.1016/s1470-2045(17)30607-1. PMID 29208439.
- ↑ "FDA approves new, targeted treatment for bladder cancer". FDA. 18 May 2016. Archived from the original on 19 May 2016. Retrieved 20 May 2016.
- ↑ "Failed confirmatory trial raises questions about atezolizumab for advanced urothelial cancer. June 2017". Archived from the original on 2019-02-18. Retrieved 2017-11-25.
External links
| Classification | |
|---|---|
| External resources |