Olmesartan
 | |
| Names | |
|---|---|
| Trade names | Benicar |
| Other names | Olmesartan medoxomil |
| |
| Clinical data | |
| Drug class | Angiotensin II receptor antagonist[1] |
| Main uses | High blood pressure, heart failure, diabetic kidney disease[1] |
| Side effects | Dizziness, headaches, diarrhea, back pain[1] |
| Pregnancy category |
|
| Routes of use | By mouth |
| Defined daily dose | 20 mg[2] |
| External links | |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a603006 |
| Legal | |
| License data |
|
| Legal status |
|
| Pharmacokinetics | |
| Bioavailability | 26% |
| Metabolism | Liver (cannot be removed by hemodialysis) |
| Elimination half-life | 13 hours |
| Excretion | Kidney 40%, biliary 60% |
| Chemical and physical data | |
| Formula | C29H30N6O6 |
| Molar mass | 558.595 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Olmesartan, sold under the trade name Benicar among others, is a medication used to treat high blood pressure, heart failure, and diabetic kidney disease.[1] It is a reasonable initial treatment for high blood pressure.[1] It is taken by mouth.[1] Versions are available as the combination olmesartan/hydrochlorothiazide and olmesartan/amlodipine.[1]
Common side effects include dizziness, headaches, diarrhea, and back pain.[1] Serious side effects may include kidney problems, low blood pressure, and angioedema.[1] Use in pregnancy may harm the baby and use when breastfeeding is not recommended.[3] It is an angiotensin II receptor antagonist and works by blocking the effects of angiotensin II.[1]
It was patented in 1991 and came into medical use in 2002.[4] It is available as a generic medication.[5] A month supply in the United Kingdom costs the NHS less than 2.50 £ as of 2019.[5] In the United States the wholesale cost of this amount is less than US$5.[6] In 2017, it was the 223rd most commonly prescribed medication in the United States, with more than two million prescriptions.[7][8]
Medical uses
Olmesartan is used for the treatment of hypertension. It may be used alone or in combination with other antihypertensive agents.[9] The U.S. Food and Drug Administration (FDA) has determined that the benefits of olmesartan continue to outweigh its potential risks when used for the treatment of patients with high blood pressure according to the drug label.[10]
Dosage
The defined daily dose is 20 mg by mouth.[2]
The usual recommended starting dose of olmesartan is 20 mg once daily. The dose may be increased to 40 mg after two weeks of therapy, if further reduction in blood pressure is desirable. Doses above 40 mg do not appear to have greater effect, and twice-daily dosing offers no advantage over the same total dose given once daily.[9] No adjustment of dosage is typically necessary for advanced age, renal impairment, or hepatic dysfunction. For patients with possible depletion of intravascular volume (e.g., patients treated with diuretics), olmesartan should be initiated with caution; consideration should be given to use of a lower starting dose in such cases.[9] If blood pressure is not controlled by olmesartan alone, a diuretic may be added. Olmesartan may be administered with other antihypertensive agents. Olmesartan may be administered with or without food.[9]
Contraindications
Contraindications for treatment with olmesartan include biliary obstruction. Another major contraindication is pregnancy; reports in the scientific literature reveal fetal malformations for pregnant women taking sartan-derived drugs.[11]
Side effects
The rate of side effects with olmesartan is similar to placebo; the only adverse effect that occurred in >1% of people treated with it and more frequently than placebo was dizziness (3% vs 1%). As with all drugs that act directly on the renin-angiotensin system, olmesartan is contraindicated in pregnancy and can cause injury and even death to the developing baby. In studies of angiotensin II receptor antagonists such as olmesartan, people with unilateral or bilateral renal artery stenosis, increases in serum creatinine or blood urea nitrogen have been reported. There has been no long-term use of olmesartan in people with unilateral or bilateral renal artery stenosis, but similar results may be expected.[12]
Rarely, olmesartan can cause severe gastrointestinal issues. The symptoms, which include nausea, vomiting, diarrhea, weight loss, and electrolyte abnormalities, are common among those who have celiac disease.[13] Recent studies suggested this form of sprue-like enteropathy could be caused by the inhibition of TGF-β, a polypeptide cytokine that maintains intestinal homeostasis. However, it is still unclear why this action was never observed with other ARBs.[14]
Chemistry
An ester prodrug, it is completely and rapidly hydrolyzed to its active acid form.[15]
Society and culture
Olmesartan and Sevikar HCT combined is marketed worldwide by Daiichi Sankyo, in India by Abbott Healthcare Pvt. Ltd. under the trade name WinBP, by Zydus Cadila under the trade name Olmy, by Ranbaxy Laboratories Ltd. under the trade name Olvance, Olsar by Unichem Laboratories and in Canada by Schering-Plough as Olmetec.
Several preparations containing olmesartan and other antihypertensives are available. Teva Pharmaceuticals produces a formulation containing olmesartan, amlodipine, and hydrochlorothiazide for once daily use.[16] Benicar HCT is the brand name of a medication containing olmesartan medoxomil with hydrochlorothiazide. Benitec H, another medication containing olmesartan medoxomil and hydrochlorothiazide, is marketed by GlaxoSmithKline in India.
Cost
A month supply in the United Kingdom costs the NHS less than 2.50 £ as of 2019.[5] In the United States the wholesale cost of this amount is less than US$5.[6] In 2017, it was the 223rd most commonly prescribed medication in the United States, with more than two million prescriptions.[7][8]
-
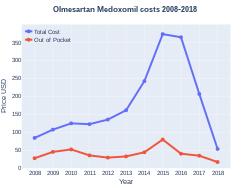
Olmesartan costs (US)
-
Olmesartan prescriptions (US)
History
It was patented in 1991 and came into medical use in 2002.[4]
References
- ↑ 1.0 1.1 1.2 1.3 1.4 1.5 1.6 1.7 1.8 1.9 "Olmesartan Medoxomil Monograph for Professionals". Drugs.com. American Society of Health-System Pharmacists. Archived from the original on 24 April 2019. Retrieved 3 March 2019.
- ↑ 2.0 2.1 "WHOCC - ATC/DDD Index". www.whocc.no. Archived from the original on 8 January 2021. Retrieved 7 September 2020.
- ↑ "Olmesartan Pregnancy and Breastfeeding Warnings". Drugs.com. Archived from the original on 30 November 2020. Retrieved 3 March 2019.
- ↑ 4.0 4.1 Fischer, Jnos; Ganellin, C. Robin (2006). Analogue-based Drug Discovery. John Wiley & Sons. p. 471. ISBN 9783527607495. Archived from the original on 2019-03-01. Retrieved 2019-03-01.
- ↑ 5.0 5.1 5.2 British national formulary : BNF 76 (76 ed.). Pharmaceutical Press. 2018. p. 177. ISBN 9780857113382.
- ↑ 6.0 6.1 "NADAC as of 2019-02-27". Centers for Medicare and Medicaid Services. Archived from the original on 2019-03-06. Retrieved 3 March 2019.
- ↑ 7.0 7.1 "The Top 300 of 2020". ClinCalc. Archived from the original on 12 February 2021. Retrieved 11 April 2020.
- ↑ 8.0 8.1 "Olmesartan Medoxomil - Drug Usage Statistics". ClinCalc. Archived from the original on 11 April 2020. Retrieved 11 April 2020.
- ↑ 9.0 9.1 9.2 9.3 "Benicar (olmesartan medoxomil)". RxList Inc. 5 July 2007. Archived from the original on 25 January 2021. Retrieved 22 July 2010.
- ↑ "FDA Alert: Benicar (olmesartan): Ongoing Safety Review". Drugs.com. Archived from the original on 2012-10-14. Retrieved 2013-06-27.
- ↑ Hünseler, C; Paneitz, A; Friedrich, D; Lindner, U; Oberthuer, A; Körber, F; Schmitt, K; Welzing, L; Müller, A; Herkenrath, P; Hoppe, B; Gortner, L; Roth, B; Kattner, E; Schaible, T (Jan 2011). "Angiotensin II receptor blocker induced fetopathy: 7 cases". Klin Padiatr. 223 (1): 10–4. doi:10.1055/s-0030-1269895.
- ↑ "BENICAR Prescribing Information" (PDF). Archived from the original (PDF) on 2010-12-13. Retrieved 2011-01-20.
- ↑ De Petris G, Caldero SG, Chen L, et al. (May 2014). "Histopathological changes in the gastrointestinal tract due to medications: an update for the surgical pathologist (part II of II)". Int. J. Surg. Pathol. 22 (3): 202–11. doi:10.1177/1066896913502230. PMID 24021900.
- ↑ Rubio-Tapia, Alberto; Herman, Margot L.; Ludvigsson, Jonas F.; Kelly, Darlene G.; Mangan, Thomas F.; Wu, Tsung-Teh; Murray, Joseph A. (2012-08-01). "Severe Spruelike Enteropathy Associated With Olmesartan". Mayo Clinic Proceedings. 87 (8): 732–738. doi:10.1016/j.mayocp.2012.06.003. ISSN 0025-6196. PMC 3538487. PMID 22728033.
- ↑ Aulakh, GK; Sodhi, RK; Singh, M (2 August 2007). "An update on non-peptide angiotensin receptor antagonists and related RAAS modulators". Life sciences. 81 (8): 615–39. doi:10.1016/j.lfs.2007.06.007. PMID 17692338.
- ↑ "OLMESARTAN MEDOXOMIL, AMLODIPINE AND HYDROCHLOROTHIAZIDE – olmesartan medoxomil, amlodipine and hydrochlorothiazide tablet, film coated". DailyMed. U.S. National Library of Medicine. Archived from the original on 2017-03-02. Retrieved 2017-03-01.
External links
| External sites: | |
|---|---|
| Identifiers: |
|
- Pages using duplicate arguments in template calls
- Drugs with non-standard legal status
- Chemical articles with unknown parameter in Infobox drug
- Chemical articles without CAS registry number
- Articles without EBI source
- Chemical pages without ChemSpiderID
- Chemical pages without DrugBank identifier
- Articles without KEGG source
- Articles without UNII source
- Drugs missing an ATC code
- Drugboxes which contain changes to verified fields
- Articles with hatnote templates targeting a nonexistent page
- Articles with changed EBI identifier
- Angiotensin II receptor antagonists
- Imidazoles
- Tetrazoles
- Carboxylate esters
- Tertiary alcohols
- Biphenyls
- 1995 in biotechnology
- Daiichi Sankyo
- Merck & Co. brands
- RTT