Lisinopril/hydrochlorothiazide
| Combination of | |
|---|---|
| Lisinopril | ACE inhibitor |
| Hydrochlorothiazide | Thiazide diuretic |
| Names | |
| Trade names | Zestoretic, Prinzide, others |
| Clinical data | |
| Pregnancy category |
|
| Routes of use | By mouth |
| Defined daily dose | not established[1] |
| External links | |
| AHFS/Drugs.com | Multum Consumer Information |
| MedlinePlus | a601070 |
| Legal | |
| Legal status |
|
Lisinopril/hydrochlorothiazide, sold under the trade name Zestoretic among others, is a combination of the medications lisinopril, an ACE inhibitor, and hydrochlorothiazide, a diuretic.[2] It is used to treat high blood pressure.[2] Typically, it becomes an option once a person is doing well on the individual components.[3] It is taken by mouth.[2]
Common side effects include dizziness, headache, cough, and feeling tired.[4] Severe side effects may include angioedema and low blood pressure.[4] Use during pregnancy may harm the baby.[4]
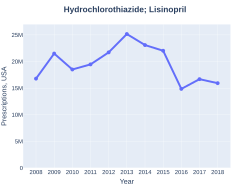
The combination was approved for medical use in the United States in 1989.[2] It is on the World Health Organization's List of Essential Medicines.[5] It is available as a generic medication.[3] In the United States the wholesale cost per dose is less than US$0.05 as of 2018[update].[6] In the United Kingdom it costs the NHS about GB£0.40 per dose as of 2017[update].[3] In 2017, it was the 44th most commonly prescribed medication in the United States, with more than 16 million prescriptions.[7][8]
Dosage
The defined daily dose is not established.[1]
Society and culture
Cost
In the United States the wholesale cost per dose is less than US$0.05 as of 2018[update].[6]In the United Kingdom it costs the NHS about GB£0.40 per dose as of 2017[update].[3] In 2017, it was the 44th most commonly prescribed medication in the United States, with more than 16 million prescriptions.[7][8]
-
Lisinopril/hydrochlorothiazide prescriptions (US)
-
Lisinopril/hydrochlorothiazide costs (US)
References
- ↑ 1.0 1.1 "WHOCC - ATC/DDD Index". www.whocc.no. Archived from the original on 1 July 2021. Retrieved 9 September 2020.
- ↑ 2.0 2.1 2.2 2.3 "Hydrochlorothiazide and lisinopril Uses, Side Effects & Warnings". Drugs.com. Cerner Multum. Archived from the original on 28 June 2019. Retrieved 26 December 2018.
- ↑ 3.0 3.1 3.2 3.3 British national formulary : BNF 74 (74 ed.). British Medical Association. 2017. p. 166. ISBN 978-0857112989.
- ↑ 4.0 4.1 4.2 "Lisinopril and Hydrochlorothiazide - FDA prescribing information, side effects and uses". Drugs.com. Solco Healthcare LLC. Archived from the original on 18 April 2019. Retrieved 26 December 2018.
- ↑ World Health Organization (2019). World Health Organization model list of essential medicines: 21st list 2019. Geneva: World Health Organization. hdl:10665/325771. WHO/MVP/EMP/IAU/2019.06. License: CC BY-NC-SA 3.0 IGO.
- ↑ 6.0 6.1 "NADAC as of 2018-12-19". Centers for Medicare and Medicaid Services. Archived from the original on 19 December 2018. Retrieved 22 December 2018.
- ↑ 7.0 7.1 "The Top 300 of 2020". ClinCalc. Archived from the original on 12 February 2021. Retrieved 11 April 2020.
- ↑ 8.0 8.1 "Hydrochlorothiazide; Lisinopril - Drug Usage Statistics". ClinCalc. Archived from the original on 11 April 2020. Retrieved 11 April 2020.
External links
| Identifiers: |
|---|
- "Hydrochlorothiazide mixture with Lisinopril". Drug Information Portal. U.S. National Library of Medicine. Archived from the original on 12 April 2020. Retrieved 12 April 2020.
- Pages using duplicate arguments in template calls
- Use dmy dates from September 2019
- Articles with invalid date parameter in template
- Drugs with non-standard legal status
- Chemical articles with unknown parameter in Infobox drug
- Chemical articles without CAS registry number
- Articles without EBI source
- Chemical pages without ChemSpiderID
- Chemical pages without DrugBank identifier
- Articles without KEGG source
- Articles without InChI source
- Articles without UNII source
- Drugs missing an ATC code
- Drugs that are a combination of chemicals
- Articles containing potentially dated statements from 2018
- All articles containing potentially dated statements
- Articles containing potentially dated statements from 2017
- Antihypertensive agents
- Combination drugs
- AstraZeneca brands
- RTT
- World Health Organization essential medicines
- All stub articles
- Antihypertensive agent stubs