Valsartan/hydrochlorothiazide
| Combination of | |
|---|---|
| Valsartan | Angiotensin II receptor antagonist |
| Hydrochlorothiazide | Thiazide diuretic |
| Names | |
| Trade names | Co-vasotec, Diovan HCT, Co-Diovan, others |
| Clinical data | |
| Pregnancy category |
|
| Routes of use | By mouth |
| Defined daily dose | not established[1] |
| External links | |
| AHFS/Drugs.com | FDA Professional Drug Information |
| MedlinePlus | a611032 |
| Legal | |
| License data | |
| Legal status |
|
Valsartan/hydrochlorothiazide, sold under the trade name Co-Diovan among others, is an medication used to treat high blood pressure when valsartan is not sufficient.[2] It is a combination of valsartan, an angiotensin receptor blocker with hydrochlorothiazide, a diuretic.[3] It is taken by mouth.[3]
Common side effects include dizziness and headaches.[3] Serious side effects may include allergic reactions, electrolyte abnormalities, and glaucoma.[4] Use in pregnancy is not recommended.[4]
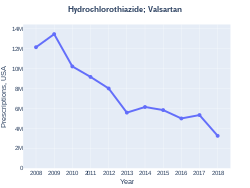
The combination was approved for medical use in the United States in 1998.[3] It is available as a generic medication.[2] A month supply in the United Kingdom costs the NHS about 1.85 as of 2019.[2] In the United States the wholesale cost of this amount is about US$4.40.[5] In 2017, it was the 133rd most commonly prescribed medication in the United States, with more than five million prescriptions.[6][7]
Dosage
The defined daily dose is not established[1]
Society and culture
Cost
A month supply in the United Kingdom costs the NHS about 1.85 as of 2019.[2] In the United States the wholesale cost of this amount is about US$4.40.[5] In 2017, it was the 133rd most commonly prescribed medication in the United States, with more than five million prescriptions.[7][6]
-
Hydrochlorothiazide/valsartan costs (US)
-
Hydrochlorothiazide/valsartan prescriptions (US)
References
- ↑ 1.0 1.1 "WHOCC - ATC/DDD Index". www.whocc.no. Archived from the original on 1 July 2021. Retrieved 10 September 2020.
- ↑ 2.0 2.1 2.2 2.3 British national formulary : BNF 76 (76 ed.). Pharmaceutical Press. 2018. p. 179. ISBN 9780857113382.
- ↑ 3.0 3.1 3.2 3.3 "Hydrochlorothiazide and valsartan Uses, Side Effects & Warnings". Drugs.com. Archived from the original on 8 February 2018. Retrieved 10 March 2019.
- ↑ 4.0 4.1 "Valsartan and Hydrochlorothiazide - FDA prescribing information, side effects and uses". Drugs.com. Archived from the original on 16 July 2017. Retrieved 10 March 2019.
- ↑ 5.0 5.1 "NADAC as of 2019-02-27". Centers for Medicare and Medicaid Services. Archived from the original on 2019-03-06. Retrieved 3 March 2019.
- ↑ 6.0 6.1 "The Top 300 of 2020". ClinCalc. Archived from the original on 18 March 2020. Retrieved 11 April 2020.
- ↑ 7.0 7.1 "Hydrochlorothiazide; Valsartan - Drug Usage Statistics". ClinCalc. Archived from the original on 11 April 2020. Retrieved 11 April 2020.
External links
| Identifiers: |
|---|
- "Hydrochlorothiazide mixture with valsartan". Drug Information Portal. U.S. National Library of Medicine. Archived from the original on 2020-02-13. Retrieved 2020-02-13.
- Pages using duplicate arguments in template calls
- Chemical articles with unknown parameter in Infobox drug
- Chemical articles without CAS registry number
- Articles without EBI source
- Chemical pages without ChemSpiderID
- Chemical pages without DrugBank identifier
- Articles without KEGG source
- Articles without InChI source
- Articles without UNII source
- Drugs missing an ATC code
- Drugs that are a combination of chemicals
- Angiotensin II receptor antagonists
- Combination drugs
- Novartis brands
- RTT
- All stub articles
- Cardiovascular system drug stubs