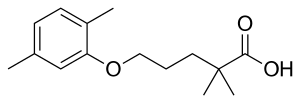
Gemfibrozil
 | |
| Names | |
|---|---|
| Trade names | Lopid, Jezil, others |
| |
| Clinical data | |
| Drug class | Fibrates[1] |
| Main uses | Abnormal blood lipid levels[1] |
| Side effects | Headache, dizziness, feeling tired, intestinal upset[1] |
| Pregnancy category |
|
| Routes of use | By mouth |
| Defined daily dose | 1.2 gram[2] |
| External links | |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a686002 |
| Legal | |
| License data |
|
| Legal status |
|
| Pharmacokinetics | |
| Bioavailability | Close to 100% |
| Protein binding | 95% |
| Metabolism | Hepatic (CYP3A4) |
| Elimination half-life | 1.5 hours |
| Excretion | Renal 94% Feces 6% |
| Chemical and physical data | |
| Formula | C15H22O3 |
| Molar mass | 250.338 g·mol−1 |
| 3D model (JSmol) | |
| Melting point | 61 to 63 °C (142 to 145 °F) |
| |
| |
Gemfibrozil, sold under the brand name Lopid among others, is a medication used to treat abnormal blood lipid levels.[1] Its is generally less preferred than statins.[1][3] Use is recommended together with dietary changes and exercise.[1] It is unclear if it changes the risk of heart disease.[1] It is taken by mouth.[1]
Common side effects include headache, dizziness, feeling tired, and intestinal upset.[1] Serious side effects may include angioedema, gallstones, liver problems, and muscle breakdown.[1] Use in pregnancy and breastfeeding is of unclear safety.[4] It belongs to the fibrates group of medications and works by decreasing the breakdown of lipids in fat cells.[1]
Gemfibrozil was patented in 1968 and came into medical use in 1982.[5] It is available as a generic medication.[3] A month supply in the United Kingdom costs the NHS about 30 £ as of 2019.[3] In the United States the wholesale cost of this amount is about 6 USD.[6] In 2017, it was the 138th most commonly prescribed medication in the United States, with more than four million prescriptions.[7][8]
Medical uses
- Hyperlipidemia (Type III)
- Hypertriglyceridemia (Type IV): Gemfibrozil, though not as effective as niacin (nicotinic acid, a form of Vitamin B3), is better tolerated.[citation needed]
- Reduce triglyceride levels [9]
- Reduce very low density lipoprotein (VLDL) levels
- Modest reduction of low density lipoprotein (LDL) levels
- Moderate increase in high density lipoprotein (HDL) levels
Dosage
The defined daily dose is 1.2 grams by mouth.[2]
Side effects
- GI distress
- Musculoskeletal pain
- Increased incidence of gallstone
- Hypokalemia (low blood potassium)
- Increased risk of cancer
Contraindications
- Gemfibrozil should not be given to people with:[citation needed]
- Liver dysfunction
- Gemfibrozil should be used with caution in these higher risk categories:[citation needed]
- Biliary tract disease
- Renal dysfunction
- Pregnant women
- Obese patients
Interactions
- Anticoagulants: Gemfibrozil potentiates the action of warfarin and indanedione anticoagulants.[citation needed]
- Statin drugs: Concomitant administration of fibrates (including gemfibrozil) with statin drugs increases the risk of muscle cramping, myopathy, and rhabdomyolysis.[10]
- Gemfibrozil inhibits the activation of the liver's Cytochrome P450 system and CYP2C8, reducing liver metabolism of many drugs, and prolonging their half lives and duration of action.
- Drugs metabolized by the Cytochrome P450 system include:
- Many antidepressants
- Many antipsychotics
- Many antiepileptics
- Theophylline and other methylxanthine drugs
- Several anesthetic agents
- Oral contraceptive pills
- Statins
- Warfarin
- Selexipag
- Drugs metabolized by the Cytochrome P450 system include:
Mechanism of actions

The exact mechanism of action of gemfibrozil is unknown; however, several theories exist regarding the very low density lipoprotein (VLDL) effect; it can inhibit lipolysis and decrease subsequent hepatic fatty acid uptake as well as inhibit hepatic secretion of VLDL; together these actions decrease serum VLDL levels and increase HDL-cholesterol; the mechanism behind HDL elevation is currently unknown.
Gemfibrozil increases the activity of extrahepatic lipoprotein lipase (LL), thereby increasing lipoprotein triglyceride lipolysis. It does so by activating peroxisome proliferator-activated receptor alpha (PPARα) 'transcription factor ligand', a receptor that is involved in metabolism of carbohydrates and fats, as well as adipose tissue differentiation. This increase in the synthesis of lipoprotein lipase thereby increases the clearance of triglycerides. Chylomicrons are degraded, VLDLs are converted to LDLs, and LDLs are converted to HDL. This is accompanied by a slight increase in secretion of lipids into the bile and ultimately the intestine. Gemfibrozil also inhibits the synthesis and increases the clearance of apolipoprotein B, a carrier molecule for VLDL.[12]
History
Gemfibrozil was selected from a series of related compounds synthesized in the laboratories of the American company Parke-Davis in the late 1970s. It came from research for compounds that lower plasma lipid levels in humans and in animals.[13]
Environmental data
Gemfibrozil has been detected in biosolids (the solids remaining after sewage treatment) at concentrations up to 2650 ng/g wet weight.[14] This indicates that it survives the wastewater treatment process. It is also detected as environmental persistent micropollutant in aquifers and in groundwaters in karstic areas.[15]
Society and culture
Cost
A month supply in the United Kingdom costs the NHS about 30 £ as of 2019.[3] In the United States the wholesale cost of this amount is about 6 USD.[6] In 2017, it was the 138th most commonly prescribed medication in the United States, with more than four million prescriptions.[7][8]
-
Gemfibrozil costs (US)
-
Gemfibrozil prescriptions (US)
References
- ↑ 1.00 1.01 1.02 1.03 1.04 1.05 1.06 1.07 1.08 1.09 1.10 "Gemfibrozil Monograph for Professionals". Drugs.com. American Society of Health-System Pharmacists. Archived from the original on 17 July 2020. Retrieved 3 March 2019.
- ↑ 2.0 2.1 "WHOCC - ATC/DDD Index". www.whocc.no. Archived from the original on 22 October 2020. Retrieved 5 September 2020.
- ↑ 3.0 3.1 3.2 3.3 British national formulary : BNF 76 (76 ed.). Pharmaceutical Press. 2018. pp. 198–199. ISBN 9780857113382.
- ↑ "Gemfibrozil Use During Pregnancy". Drugs.com. Archived from the original on 30 November 2020. Retrieved 3 March 2019.
- ↑ Fischer, Jnos; Ganellin, C. Robin (2006). Analogue-based Drug Discovery. John Wiley & Sons. p. 474. ISBN 9783527607495. Archived from the original on 2021-04-14. Retrieved 2020-07-01.
- ↑ 6.0 6.1 "NADAC as of 2019-02-27". Centers for Medicare and Medicaid Services. Archived from the original on 2019-03-06. Retrieved 3 March 2019.
- ↑ 7.0 7.1 "The Top 300 of 2020". ClinCalc. Archived from the original on 12 February 2021. Retrieved 11 April 2020.
- ↑ 8.0 8.1 "Gemfibrozil - Drug Usage Statistics". ClinCalc. Archived from the original on 13 April 2020. Retrieved 11 April 2020.
- ↑ "Gemfibrozil." WebMD.com Accessed 14 June 2014. http://www.webmd.com/drugs/drug-11423-gemfibrozil+oral.aspx Archived 2014-06-05 at the Wayback Machine
- ↑ "Medicines Complete". Medicines Complete. British National Formulary. Archived from the original on 29 May 2020. Retrieved 1 February 2020.
- ↑ Kim, Kyeongsoon; Kleinman, Hynda K.; Lee, Hahn-Jun; Pahan, Kalipada (17 June 2017). "Safety and potential efficacy of gemfibrozil as a supportive treatment for children with late infantile neuronal ceroid lipofuscinosis and other lipid storage disorders". Orphanet Journal of Rare Diseases. 12 (1): 113. doi:10.1186/s13023-017-0663-8. ISSN 1750-1172.
- ↑ "Gemfibrozil". Archived from the original on 2018-03-19. Retrieved 2018-03-19.
- ↑ Rodney G, Uhlendorf P, Maxwell RE (1976). "The hypolipidaemic effect of gemfibrozil (CI-719) in laboratory animals". Proceedings of the Royal Society of Medicine. 69 Suppl 2 (2_suppl): 6–10. doi:10.1177/00359157760690S203. PMC 1864017. PMID 828263.
- ↑ "Biosolids". 2014-04-23. Archived from the original on 2015-02-16. Retrieved 2015-08-25.
- ↑ Doummar J, Aoun M (August 2018). "Assessment of the origin and transport of four selected emerging micropollutants sucralose, Acesulfame-K, gemfibrozil, and iohexol in a karst spring during a multi-event spring response". Journal of Contaminant Hydrology. 215: 11–20. Bibcode:2018JCHyd.215...11D. doi:10.1016/j.jconhyd.2018.06.003. PMID 29983209.
Further reading
- Bellosta S, Paoletti R, Corsini A (June 2004). "Safety of statins: focus on clinical pharmacokinetics and drug interactions". Circulation. 109 (23 Suppl 1): III50-7. doi:10.1161/01.cir.0000131519.15067.1f. PMID 15198967.
- Quintanilla Rodriguez, B. S.; Correa, R. (2020). "Gemfibrozil". StatPearls. National Center for Biotechnology Information (NCBI). PMID 31424850. Bookshelf ID: NBK545266. Archived from the original on 2021-08-28. Retrieved 2020-05-15.
External links
| External sites: | |
|---|---|
| Identifiers: |
|
- "Lopid International Study" (PDF). European Medicines Agency. Archived from the original (PDF) on 2007-06-11.
- "Indian Health Service National Pharmacy and Therapeutics Committee Review of Statins, Fibrates, and Niacin" (PDF). Indian Health Service. San Diego: U.S. Department of Health and Human Services. February 12–13, 2009. Archived from the original (PDF) on June 22, 2014.
- Pages using duplicate arguments in template calls
- Webarchive template wayback links
- CS1: long volume value
- Drugs with non-standard legal status
- Chemical articles with unknown parameter in Infobox drug
- Chemical articles without CAS registry number
- Articles without EBI source
- Chemical pages without ChemSpiderID
- Chemical pages without DrugBank identifier
- Articles without KEGG source
- Articles without UNII source
- Drugs missing an ATC code
- Drugboxes which contain changes to watched fields
- All articles with unsourced statements
- Articles with unsourced statements from December 2012
- Articles with invalid date parameter in template
- RTT
- Fibrates
- Phenol ethers
- Carboxylic acids
- CYP2C8 inhibitors

