Ezetimibe
 | |
| Names | |
|---|---|
| Pronunciation | /ɛˈzɛtɪmɪb, -maɪb/ |
| Trade names | Zetia, Ezetrol, others |
| |
| Clinical data | |
| Drug class | Cholesterol absorption inhibitor |
| Pregnancy category |
|
| Routes of use | By mouth (tablets) |
| Defined daily dose | 10 mg[1] |
| External links | |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a603015 |
| Legal | |
| License data | |
| Legal status | |
| Pharmacokinetics | |
| Bioavailability | 35% to 65% |
| Protein binding | >90% |
| Metabolism | Intestinal wall, liver |
| Elimination half-life | 19 h to 30 h |
| Excretion | Renal 11%, faecal 78% |
| Chemical and physical data | |
| Formula | C24H21F2NO3 |
| Molar mass | 409.433 g·mol−1 |
| 3D model (JSmol) | |
| Melting point | 164 to 166 °C (327 to 331 °F) |
| |
| |
Ezetimibe is a medication used to treat high blood cholesterol and certain other lipid abnormalities.[2] Generally it is used together with dietary changes and a statin.[3] Alone, it is less preferred than a statin.[2] It is taken by mouth.[2] It is also available in the fixed combinations ezetimibe/simvastatin, ezetimibe/atorvastatin, and ezetimibe/rosuvastatin.[2]
Common side effects include upper respiratory tract infections, joint pain, diarrhea, and tiredness.[2] Serious side effects may include anaphylaxis, liver problems, depression, and muscle breakdown.[2][3] Use in pregnancy and breastfeeding is of unclear safety.[4] Ezetimibe works by decreasing cholesterol absorption in the intestines.[3]
Ezetimibe was approved for medical use in the United States in 2002.[2] It is available as a generic medication.[3] A month supply in the United Kingdom costs the NHS about £26.21 as of 2019[update].[3] In the United States the wholesale cost of this amount is about US$5.85.[5] In 2017, it was the 151st most commonly prescribed medication in the United States, with more than four million prescriptions.[6][7]
Medical uses
A 2018 review found that ezetimibe used as sole treatment slightly lowered plasma levels of lipoprotein(a), but the effect was not large enough to be important.[8] A 2015 review found that adding ezetimibe to statin treatment of high blood cholesterol had no effect on overall mortality or cardiovascular mortality, although it significantly reduced the risk of MI and stroke.[9] A 2015 trial found that adding ezetimibe to simvastatin had no effect on overall mortality but did lower the risk of heart attack or stroke in people with prior heart attack.[10][11] Several treatment guidelines recommend adding ezetimibe in select high risk persons in whom LDL goals cannot be achieved by maximally tolerated statin alone.[12][13][14][15][16]
Ezetimibe is indicated in the United States as an add-on to dietary measures to reduce levels of certain lipids in people with:[17]
- Primary hyperlipidemia, alone or with a statin
- Mixed hyperlipidemia, in combination with fenofibrate
- Homozygous familial hypercholesterolemia, in combination with specific statins
- Homozygous sitosterolemia
Ezetimibe improves the non-alcoholic fatty liver disease activity score but the available evidence indicates it does not improve outcomes of hepatic steatosis.[18]
Dosage
The defined daily dose is 10 mg by mouth.[1]
Side effects
Common side effects (≥1% of people) associated with ezetimibe therapy include headache and/or diarrhea (steatorrhea). Infrequent adverse effects (0.1–1% of patients) include myalgia and/or raised liver function test (ALT/AST) results. Rarely (<0.1% of patients), hypersensitivity reactions (rash, angioedema) or myopathy may occur.[17] Cases of muscle problems (myalgia and rhabdomyolysis) have been reported and are included as warnings on the label for ezetimibe.[17]
The two contraindications to taking ezetimibe are a previous allergic reaction to it, including symptoms of rash, angioedema, and anaphylaxis, and severe liver disease, especially when taken with a statin.[19]
Ezetimibe may have medication interactions with ciclosporin and with fibrates other than fenofibrate.[17]
Overdose
The incidence of overdose with ezetimibe is rare; subsequently, few data exist on the effects of overdose. However, an acute overdose of ezetimibe is expected to produce an exaggeration of its usual effects, leading to loose stools, abdominal pain, and fatigue.[20]
Pharmacology
Mechanism of action
Ezetimibe inhibits the absorption of cholesterol from the small intestine and decreases the amount of cholesterol normally available to liver cells. This leads them to absorb more cholesterol from circulation and thus causes lowering levels of circulating cholesterol. It blocks the critical mediator of cholesterol absorption, the Niemann-Pick C1-like 1 (NPC1L1) protein on the gastrointestinal tract epithelial cells, as well as in hepatocytes; it blocks aminopeptidase N and interrupts a caveolin 1–annexin A2 complex involved in trafficking cholesterol.[10]
Pharmacokinetics
Within 4–12 hours of the oral administration of a 10-mg dose to fasting adults, the attained mean ezetimibe peak plasma concentration (Cmax) was 3.4–5.5 ng/ml. Following oral administration, ezetimibe is absorbed and extensively conjugated to a phenolic glucuronide (active metabolite). Mean Cmax (45–71 ng/ml) of ezetimibe-glucuronide is attained within 1–2 hours. The concomitant administration of food (high-fat vs. nonfat meals) has no effect on the extent of absorption of ezetimibe. However, coadministration with a high-fat meal increases its Cmax by 38%. The absolute bioavailability cannot be determined, since ezetimibe is insoluble in aqueous media suitable for injection. Ezetimibe and its active metabolites are highly bound to human plasma proteins (90%).[17]
Ezetimibe is primarily metabolized in the liver and the small intestine via glucuronide conjugation with subsequent renal and biliary excretion.[21] Both the parent compound and its active metabolite are eliminated from plasma with a half-life around 22 hours, allowing for once-daily dosing. Ezetimibe lacks significant inhibitor or inducer effects on cytochrome P450 isoenzymes, which explains its limited number of drug interactions. No dose adjustment is needed in patients with chronic kidney disease or mild hepatic dysfunction (Child-Pugh score 5–6). Due to insufficient data, the manufacturer does not recommend ezetimibe for patients with moderate to severe hepatic impairment (Child-Pugh score 7–15). In patients with mild, moderate, or severe hepatic impairment, the mean AUC values for total ezetimibe are increased about 1.7-fold, 3-to-4-fold, and 5-to-6-fold, respectively, compared to healthy subjects.[17]
Society and culture
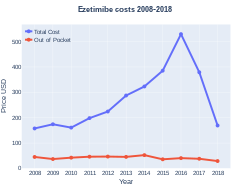
Cost
In the United States, as of 2019[update], it costs about US$0.21 per dose.[5] In 2015, it cost between US$4.84 and US$7.88.[22]
-
Ezetimibe costs (US)
-
Ezetimibe prescriptions (US)
References
- ↑ 1.0 1.1 "WHOCC - ATC/DDD Index". www.whocc.no. Archived from the original on 13 August 2020. Retrieved 9 September 2020.
- ↑ 2.0 2.1 2.2 2.3 2.4 2.5 2.6 "Ezetimibe Monograph for Professionals". Drugs.com. American Society of Health-System Pharmacists. Archived from the original on 17 June 2019. Retrieved 13 April 2019.
- ↑ 3.0 3.1 3.2 3.3 3.4 British national formulary : BNF 76 (76 ed.). Pharmaceutical Press. 2018. p. 196. ISBN 9780857113382.
- ↑ "Ezetimibe (Zetia) Use During Pregnancy". Drugs.com. Archived from the original on 13 April 2019. Retrieved 13 April 2019.
- ↑ 5.0 5.1 "NADAC as of 2019-02-27". Centers for Medicare and Medicaid Services. Archived from the original on 6 March 2019. Retrieved 3 March 2019. Cite error: Invalid
<ref>tag; name "NADAC2019" defined multiple times with different content - ↑ "The Top 300 of 2020". ClinCalc. Archived from the original on 12 February 2021. Retrieved 11 April 2020.
- ↑ "Ezetimibe - Drug Usage Statistics". ClinCalc. Archived from the original on 28 February 2020. Retrieved 11 April 2020.
- ↑ Awad K, Mikhailidis DP, Katsiki N, Muntner P, Banach M (March 2018). "Effect of Ezetimibe Monotherapy on Plasma Lipoprotein(a) Concentrations in Patients with Primary Hypercholesterolemia: A Systematic Review and Meta-Analysis of Randomized Controlled Trials". Drugs. 78 (4): 453–462. doi:10.1007/s40265-018-0870-1. PMID 29396832.
- ↑ Savarese G, De Ferrari GM, Rosano GM, Perrone-Filardi P (December 2015). "Safety and efficacy of ezetimibe: A meta-analysis". Int. J. Cardiol. 201: 247–52. doi:10.1016/j.ijcard.2015.08.103. PMID 26301648.
- ↑ 10.0 10.1 Phan BA, Dayspring TD, Toth PP (2012). "Ezetimibe therapy: mechanism of action and clinical update". Vasc Health Risk Manag. 8: 415–27. doi:10.2147/VHRM.S33664. PMC 3402055. PMID 22910633.
- ↑ Cannon, Christopher P. (18 June 2015). "Ezetimibe Added to Statin Therapy after Acute Coronary Syndromes". New England Journal of Medicine. 372 (25): 2387–2397. doi:10.1056/NEJMoa1410489. hdl:11573/1150638. PMID 26039521.
- ↑ Alenghat, Francis J.; Davis, Andrew M. (2019). "Management of Blood Cholesterol". JAMA. 321 (8): 800–801. doi:10.1001/jama.2019.0015. ISSN 0098-7484. PMC 6679800. PMID 30715135.
- ↑ Catapano AL, Reiner Z, De Backer G, et al. (July 2011). "ESC/EAS Guidelines for the management of dyslipidaemias The Task Force for the management of dyslipidaemias of the European Society of Cardiology (ESC) and the European Atherosclerosis Society (EAS)". Atherosclerosis. 217 (1): 3–46. doi:10.1016/j.atherosclerosis.2011.06.011. PMID 21882396.
- ↑ Teramoto T, Sasaki J, Ishibashi S, et al. (2013). "Executive summary of the Japan Atherosclerosis Society (JAS) guidelines for the diagnosis and prevention of atherosclerotic cardiovascular diseases in Japan -2012 version". J. Atheroscler. Thromb. 20 (6): 517–23. doi:10.5551/jat.15792. PMID 23665881.
- ↑ "Lipid modification: cardiovascular risk assessment and the modification of blood lipids for the primary and secondary prevention of cardiovascular disease | Guidance and guidelines | NICE". Archived from the original on 19 November 2014.
- ↑ Expert Dyslipidemia Panel of the International Atherosclerosis Society Panel members (2014). "An International Atherosclerosis Society Position Paper: global recommendations for the management of dyslipidemia--full report". J Clin Lipidol. 8 (1): 29–60. doi:10.1016/j.jacl.2013.12.005. PMID 24528685.
- ↑ 17.0 17.1 17.2 17.3 17.4 17.5 Zetia label, Rev 23 Archived 27 February 2017 at the Wayback Machine. Revised: January 2012
- ↑ Lee HY, Jun DW, Kim HJ, Oh H, Saeed WK, Ahn H, Cheung RC, Nguyen MH (2019). "Ezetimibe decreased nonalcoholic fatty liver disease activity score but not hepatic steatosis". Korean J. Intern. Med. 34 (2): 296–304. doi:10.3904/kjim.2017.194. PMC 6406097. PMID 29551054.
- ↑ U.S. National Library of Medicine, National Institutes of Health, U.S. Department of Health and Human Services. Page last updated: 27 October 2014 Medline Plus: Ezetimibe Archived 5 July 2016 at the Wayback Machine
- ↑ "Ezetimibe - National Library of Medicine HSDB Database". toxnet.nlm.nih.gov. National Library of Medicine. Archived from the original on 12 June 2018. Retrieved 29 May 2018.
- ↑ SJS Basha; SA Naveed; NK Tiwari; D Shashikumar; S Muzeeb; TR Kumar; NV Kumar; NP Rao; N Srinivas; M Ramesh; NR Srinivas (2007). "Mechanism of Drug-Drug Interactions Between Warfarin and Statins". Journal of Chromatography B. 853 (1): 88–96. doi:10.1016/j.jchromb.2007.02.053. PMID 17442643.
- ↑ Langreth, Robert (29 June 2016). "Decoding Big Pharma's Secret Drug Pricing Practices". Bloomberg. Archived from the original on 13 July 2016. Retrieved 15 July 2016.
External links
| External sites: | |
|---|---|
| Identifiers: |
|
- Pages using duplicate arguments in template calls
- Pages with reference errors
- Webarchive template wayback links
- Use dmy dates from February 2020
- Articles with invalid date parameter in template
- Chemical articles with unknown parameter in Infobox drug
- Chemical articles without CAS registry number
- Articles without EBI source
- Chemical pages without ChemSpiderID
- Chemical pages without DrugBank identifier
- Articles without KEGG source
- Articles without UNII source
- Drugs missing an ATC code
- Drugboxes which contain changes to watched fields
- Articles containing potentially dated statements from 2019
- All articles containing potentially dated statements
- Hypolipidemic agents
- Lactams
- Merck & Co. brands
- Organofluorides
- Phenols
- Schering-Plough brands
- RTT