Frontotemporal dementia
| Frontotemporal dementia | |
|---|---|
| Other names: Frontotemporal degeneration disease,[1] frontotemporal neurocognitive disorder[2] | |
 | |
| Brain MRI of a female of 65 years with frontotemporal dementia. Cortical and white matter atrophy of the frontal lobes is clear in all images. | |
| Specialty | Psychiatry, neurology |
| Symptoms | Personality changes, lack of emotions, repetitive behavior, loss of vocabulary, decreased ability to understand language[3][4] |
| Usual onset | 50s and 60s[5] |
| Types | Behavioral variant (bvFTD), semantic variant (svPPA), nonfluent variant (nfvPPA)[6][7] |
| Causes | Unknown[8] |
| Risk factors | Family history, head injury, thyroid disease[6][4] |
| Diagnostic method | Based on symptoms and MRI[5] |
| Differential diagnosis | Alzheimer, vascular dementia, Lewy body dementia, alcoholism[4] |
| Treatment | Supportive care[5] |
| Prognosis | Life expectancy 7.5 yrs[4] |
| Frequency | 2 per 10,000 (45 to 65 year old)[5] |
Frontotemporal dementia (FTD) encompasses several types of dementia involving the frontal and temporal lobes.[3][6] The primary symptoms generally involve behavior or language.[6] This may include personality changes, lack of emotions, repetitive behavior, loss of vocabulary, and decreased ability to understand language.[3][4] They generally start gradually and worsen over several years.[5] Memory and motor functions often remain relatively preserved.[3]
The cause is unknown.[8] Risk factors include family history, head injury, and thyroid disease.[6][4] The underlying mechanism involves loss of neurons, primarily spindle neurons.[4][9] There are three main subtypes: behavioral variant (bvFTD) and two types of primary progressive aphasia - semantic variant (svPPA) and nonfluent variant (nfvPPA).[6][7] Related disorders include progressive supranuclear palsy, corticobasal syndrome, and FTD with amyotrophic lateral sclerosis (FTD-ALS).[5]
There is no cure.[8] Management may require social supports, behavioral therapy, and speech therapy.[4] Other efforts to improve symptoms may include SSRIs, antipsychotics, and galantamine.[8] A number of treatments are under study.[4] The average life expectancy is 7.5 years after diagnosis.[4]
FTD newly affects per year about 2 per 100,000 people in their 40s, 3 per 100,000 in their 50s, and 9 per 100,000 in their 60s.[4] Before the age of 65, it is second only to Alzheimer's disease (AD) in frequency.[5] Those in their 40s to 60s are most commonly affected.[5] Males and females are affected equally frequently.[5] It was first described by Arnold Pick in 1892 and was originally called Pick's disease, a term now reserved for bvFTD.[10][8]
Signs and symptoms
Frontotemporal dementia (FTD) is an early-onset disorder that mostly occurs before the age of 65 but can begin earlier, and in 20%-25% of cases onset is later.[11][12] It is the most common early presenting dementia.[13] A gradual onset and progression of changes in behavior or language deficits are reported to have begun several years prior to presentation to a neurologist.[5]
FTD is traditionally difficult to diagnose owing to the diverse nature of the associated symptoms. Signs and symptoms are classified into three groups based on the affected functions of the frontal and temporal lobes:[8] These are behavioural variant frontotemporal dementia, semantic dementia, and progressive nonfluent aphasia. An overlap between symptoms can occur as the disease progresses and spreads through the brain regions.[12]
Other characteristics
In later stages of FTD, the clinical phenotypes may overlap.[14] People with FTD tend to struggle with binge eating and compulsive behaviors.[15] Binge eating habits are often associated with changes in food preferences (cravings for more sweets, carbohydrates), eating inedible objects and snatching food from others. Recent findings from structural MRI research have indicated that eating changes in FTD are associated with atrophy (wasting) in the right ventral insula, striatum, and orbitofrontal cortex.[15]
People with FTD show marked deficiencies in executive functioning and working memory.[16] Most become unable to perform skills that require complex planning or sequencing.[17] In addition to the characteristic cognitive dysfunction, a number of primitive reflexes known as frontal release signs are often able to be elicited. Usually the first of these frontal release signs to appear is the palmomental reflex which appears relatively early in the disease course whereas the palmar grasp reflex and rooting reflex appear late in the disease course.[citation needed]
In rare cases, FTD can occur in people with amyotrophic lateral sclerosis (ALS) a motor neuron disease. The prognosis for people with ALS is worse when combined with FTD, shortening survival by about a year.[18]
Genetics
A higher proportion of fontotemporal dementias seem to have a familial component than other neurodegenerative diseases such as Alzheimer's disease. More and more mutations and genetic variants are being identified all the time, needing constant updating of genetic influences.
- Tau-positive frontotemporal dementia and parkinsonism linked to chromosome 17 (FTDP-17) is caused by mutations in the MAPT gene on chromosome 17 that encodes the tau protein.[19] It has been determined that there is a direct relationship between the type of tau mutation and the neuropathology of gene mutations. The mutations at the splice junction of exon 10 of tau lead to the selective deposition of the repetitive tau in neurons and glia. The pathological phenotype associated with mutations elsewhere in tau is less predictable with both typical neurofibrillary tangles (consisting of both 3 repeat and 4 repeat tau) and Pick bodies (consisting of 3 repeat tau) having been described. The presence of tau deposits within glia is also variable in families with mutations outside of exon 10. This disease is now informally designated FTDP-17T. FTD shows a linkage to the region of the tau locus on chromosome 17, but it is believed that there are two loci leading to FTD within megabases of each other on chromosome 17.[20]
- FTD caused by FTLD-TDP43 has numerous genetic causes. Some cases are due to mutations in the GRN gene, also located on chromosome 17. Others are caused by VCP mutations, although these patients present with a complex picture of multisystem proteinopathy that can include amyotrophic lateral sclerosis, inclusion body myopathy, Paget's disease of bone, and FTD. The most recent addition to the list is a hexanucleotide repeat expansion in intron 1 of C9ORF72.[21] Only one or two cases have been reported describing TARDBP (the TDP-43 gene) mutations in a clinically pure FTD (FTD without MND).[citation needed]
- Several other genes have been linked to this condition. These include CYLD, OPTN, SQSTM1 and TBK1.[22] These genes have been implicated in the autophagy pathway.
- No genetic causes of FUS pathology in FTD have yet been reported.[citation needed]
Pathology
There are three main histological subtypes found at post-mortem: FTLD-tau, FTLD-TDP, and FTLD-FUS. In rare cases, patients with clinical FTD were found to have changes consistent with Alzheimer's disease on autopsy.[23] The most severe brain atrophy appears to be associated with behavioral variant FTD, and corticobasal degeneration.[24]
With regard to the genetic defects that have been found, repeat expansion in the C9orf72 gene is considered a major contribution to frontotemporal lobar degeneration, although defects in the GRN and MAPT genes are also associated with it.[25]
Diagnosis
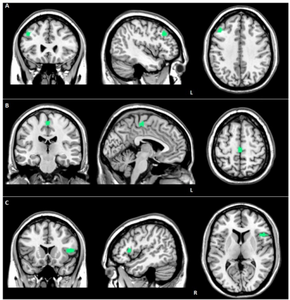
Structural MRI scans often reveal frontal lobe and/or anterior temporal lobe atrophy but in early cases the scan may seem normal. Atrophy can be either bilateral or asymmetric.[26] Registration of images at different points of time (e.g., one year apart) can show evidence of atrophy that otherwise (at individual time points) may be reported as normal. Many research groups have begun using techniques such as magnetic resonance spectroscopy, functional imaging and cortical thickness measurements in an attempt to offer an earlier diagnosis to the FTD patient. Fluorine-18-fluorodeoxyglucose positron emission tomography (FDG-PET) scans classically show frontal and/or anterior temporal hypometabolism, which helps differentiate the disease from Alzheimer's disease. The PET scan in Alzheimer's disease classically shows biparietal hypometabolism. Meta-analyses based on imaging methods have shown that frontotemporal dementia mainly affects a frontomedial network discussed in the context of social cognition or 'theory of mind'.[27] This is entirely in keeping with the notion that on the basis of cognitive neuropsychological evidence, the ventromedial prefrontal cortex is a major locus of dysfunction early on in the course of the behavioural variant of frontotemporal degeneration.[28] The language subtypes of frontotemporal lobar degeneration (semantic dementia and progressive nonfluent aphasia) can be regionally dissociated by imaging approaches in vivo.[29]
The confusion between Alzheimer's and FTD is justifiable due to the similarities between their initial symptoms. Patients do not have difficulty with movement and other motor tasks.[30] As FTD symptoms appear, it is difficult to differentiate between a diagnosis of Alzheimer's disease and FTD. There are distinct differences in the behavioral and emotional symptoms of the two dementias, notably, the blunting of emotions seen in FTD patients.[26] In the early stages of FTD, anxiety and depression are common, which may result in an ambiguous diagnosis. However, over time, these ambiguities fade away as this dementia progresses and defining symptoms of apathy, unique to FTD, start to appear.[citation needed]
Recent studies over several years have developed new criteria for the diagnosis of behavioral variant frontotemporal dementia (bvFTD).The confirmatory diagnosis is made by brain biopsy, but other tests can be used to help, such as MRI, EEG, CT, and physical examination and history.[31] Six distinct clinical features have been identified as symptoms of bvFTD.[32]
- Disinhibition
- Apathy/Inertia
- Loss of Sympathy/Empathy
- Perseverative/compulsive behaviors
- Hyperorality
- Dysexecutive neuropsychological profile
Of the six features, three must be present in a patient to diagnose one with possible bvFTD. Similar to standard FTD, the primary diagnosis stems from clinical trials that identify the associated symptoms, instead of imaging studies.[32] The above criteria are used to distinguish bvFTD from disorders such as Alzheimer's and other causes of dementia. In addition, the new criteria allow for a diagnostic hierarchy distinguished possible, probable, and definite bvFTD based on the number of symptoms present.[citation needed]
Neuropsychological tests
The progression of the degeneration caused by bvFTD may follow a predictable course. The degeneration begins in the orbitofrontal cortex and medial aspects such as ventromedial cortex. In later stages, it gradually expands its area to the dorsolateral cortex and the temporal lobe.[33] Thus, the detection of dysfunction of the orbitofrontal cortex and ventromedial cortex is important in the detection of early stage bvFTD. As stated above, a behavioural change may occur before the appearance of any atrophy in the brain in the course of the disease. Because of that, image scanning such as MRI can be insensitive to the early degeneration and it is difficult to detect early-stage bvFTD.[citation needed]
In neuropsychology, there is an increasing interest in using neuropsychological tests such as the Iowa gambling task or Faux Pas Recognition test as an alternative to imaging for the diagnosis of bvFTD.[34] Both the Iowa gambling task and the Faux Pas test are known to be sensitive to dysfunction of the orbitofrontal cortex.[citation needed]
Faux Pas Recognition test is intended to measure one’s ability to detect faux pas types of social blunders (accidentally make a statement or an action that offends others). It is suggested that people with orbitofrontal cortex dysfunction show a tendency to make social blunders due to a deficit in self-monitoring.[35] Self-monitoring is the ability of individuals to evaluate their behaviour to make sure that their behaviour is appropriate in particular situations. The impairment in self-monitoring leads to a lack of social emotion signals. The social emotions such as embarrassment are important in the way that they signal the individual to adapt social behaviour in an appropriate manner to maintain relationships with others. Though patients with damage to the OFC retain intact knowledge of social norms, they fail to apply it to actual behaviour because they fail to generate social emotions that promote adaptive social behaviour.[35]
The other test, the Iowa gambling task, is a psychological test intended to simulate real-life decision making. The underlying concept of this test is the somatic marker hypothesis. This hypothesis argues that when people have to make complex uncertain decisions, they employ both cognitive and emotional processes to assess the values of the choices available to them. Each time a person makes a decision, both physiological signals and evoked emotion (somatic marker) are associated with their outcomes and it accumulates as experience. People tend to choose the choice which might produce the outcome reinforced with positive stimuli, thus it biases decision-making towards certain behaviours while avoiding others.[36] It is thought that somatic marker is processed in orbitofrontal cortex.[citation needed]
The symptoms observed in bvFTD are caused by dysfunction of the orbitofrontal cortex, thus these two neuropsychological tests might be useful in detecting the early stage bvFTD. However, as self-monitoring and somatic marker processes are so complex, it likely involves other brain regions. Therefore, neuropsychological tests are sensitive to the dysfunction of orbitofrontal cortex, yet not specific to it. The weakness of these tests is that they do not necessarily show dysfunction of the orbitofrontal cortex.[citation needed]
In order to solve this problem, some researchers combined neuropsychological tests which detect the dysfunction of orbitofrontal cortex into one so that it increases its specificity to the degeneration of the frontal lobe in order to detect the early-stage bvFTD. They invented the Executive and Social Cognition Battery which comprises five neuropsychological tests.[34]
- Iowa gambling task
- Faux Pas test
- Hotel task
- Mind in the Eyes
- Multiple Errands Task
The result has shown that this combined test is more sensitive in detecting the deficits in early bvFTD.[34]
Subtypes
The main subtypes of frontotemporal dementia are behavioral variant FTD, semantic dementia, progressive nonfluent aphasia, and FTD associated with amyotrophic lateral sclerosis (FTD–ALS). Two distinct rare subtypes are neuronal intermediate filament inclusion disease, and basophilic inclusion body disease. Related disorders are corticobasal syndrome, and progressive supranuclear palsy.[5]
Behavioral variant frontotemporal dementia
Behavioral variant frontotemporal dementia (BvFTD) was previously known as Pick's disease, and is the most common of the FTD types.[10] BvFTD is diagnosed four times more than the PPA variants.[37] Behavior can change in BvFTD in either of two ways — it can change to being impulsive and disinhibited, acting in socially unacceptable ways; or it can change to being listless and apathetic.[38][14] About 12-13% of people with bvFTD develop motor neuron disease.[39]
The Pick bodies in behavioral variant FTD are spherical inclusion bodies found in the cytoplasm of affected cells. They consist of tau fibrils as a major component together with a number of other protein products including ubiquitin and tubulin.[40]
Semantic dementia
Semantic dementia (SD) is characterized by the loss of semantic understanding, resulting in impaired word comprehension. However, speech remains fluent and grammatical.[14]
Progressive nonfluent aphasia
Progressive nonfluent aphasia (PNFA) is characterized by progressive difficulties in speech production.[14]
Neuronal intermediate filament inclusion disease
Neuronal intermediate filament inclusion disease (NIFID) is a rare distinct variant. The inclusion bodies that are present in NIFID are cytoplasmic and made up of type IV intermediate filaments.[41][42] NIFID has an early age of onset between 23–56. Symptoms can include behavioural, and personality changes, memory and cognitive impairments, language difficulties, motor weakness, and extrapyramidal symptoms.[41] NIFID is one of the FTLD-FUS proteopathies.[42] Imaging commonly shows atrophy in the frontotemporal region, and in part of the striatum in the basal ganglia. Post-mortem studies show a marked reduction in the caudate nucleus of the striatum; frontal temporal gyri are narrowed with widened intervening sulci, and the lateral ventricles are enlarged.[41]
Basophilic inclusion body disease
Another rare FTD variant, also a FTLD-FUS proteopathy is basophilic inclusion body disease (BIBD).[43]
Management
Currently, there is no cure for FTD. Treatments are available to manage the behavioral symptoms. Disinhibition and compulsive behaviors can be controlled by selective serotonin reuptake inhibitors (SSRIs).[44][45] Although Alzheimer's and FTD share certain symptoms, they cannot be treated with the same pharmacological agents because the cholinergic systems are not affected in FTD.[26]
Because FTD often occurs in younger people (i.e. in their 40s or 50s), it can severely affect families. Patients often still have children living in the home.[citation needed]
Prognosis
Symptoms of frontotemporal dementia progress at a rapid, steady rate. Patients suffering from the disease can survive for 2–20 years. Eventually patients will need 24-hour care for daily function.[46]
CSF leaks are a known cause of reversible frontotemporal dementia.[47]
History
Frontotemporal dementia was first described by Pick in 1892.[48] In 1989, Snowden suggested the term “semantic dementia” to describe the patient with predominant left temporal atrophy and aphasia that Pick described. The first research criteria for FTD “Clinical and neuropathological criteria for frontotemporal dementia. The Lund and Manchester Groups,” was developed in 1994. The clinical diagnostic criteria were revised in the late 1990s, when the FTD spectrum was divided into a behavioral variant, a nonfluent aphasia variant and a semantic dementia variant.[37] The most recent revision of the clinical research criteria was by International Behavioural Variant FTD Criteria Consortium (FTDC) in 2011.[49]
Notable cases
Notable cases of bvFTD when mostly were referred to as Pick's disease.
- Don Cardwell (1935–2008), Major League Baseball pitcher[50]
- Jerry Corbetta (1947–2016), frontman, organist and keyboardist of American psychedelic rock band Sugarloaf[51]
- Ted Darling (1935–1996), Buffalo Sabres television announcer
- Robert W. Floyd (1936–2001), computer scientist[52]
- Colleen Howe (1933–2009), sports agent and hockey team manager, known as "Mrs. Hockey"[53]
- Ralph Klein (1942–2013), former premier of Alberta, Canada
- Kevin Moore (1958–2013), English footballer[54]
- Ernie Moss (born 1949), English footballer[55]
- Nic Potter (1951–2013), British bassist for Van der Graaf Generator[56]
- Christina Ramberg (1946–1995), American painter associated with the Chicago Imagists[57]
- Pat Moran (1947-2011), British record producer, singer and Mellotron player with progressive rock band Spring
- David Rumelhart (1942–2011), American cognitive psychologist[citation needed]
- Colin Savage, father of footballer Robbie Savage[58]
- Sir Nicholas Wall (1945–2017), English judge[59]
- Mark Wirtz (1943-2020), pop musician, composer and producer[60]
- Kazi Nazrul Islam (1899-1976), notable Bengali poet and National Poet of Bangladesh[61]
- Charmian Carr (1942-2016), who played Liesl, from the Sound of Music, born Charmian Anne Farnon.
See also
References
- ↑ Perez, L., "Ron Oberman, Senior Record Executive and Former Publicist to David Bowie, Dies at 76" Archived 2021-05-07 at the Wayback Machine, The Hollywood Reporter, November 24, 2019.
- ↑ Diagnostic and statistical manual of mental disorders : DSM-5 (5th ed.). Arlington, VA: American Psychiatric Association. 2013. pp. 614–618. ISBN 9780890425541.
- ↑ 3.0 3.1 3.2 3.3 "ICD-11 - Mortality and Morbidity Statistics". icd.who.int. Archived from the original on 2018-08-01. Retrieved 2020-10-17.
- ↑ 4.00 4.01 4.02 4.03 4.04 4.05 4.06 4.07 4.08 4.09 4.10 Khan, I; De Jesus, O (January 2021). "Frontotemporal Lobe Dementia". PMID 32644712.
{{cite journal}}: Cite journal requires|journal=(help) - ↑ 5.00 5.01 5.02 5.03 5.04 5.05 5.06 5.07 5.08 5.09 5.10 Finger, EC (April 2016). "Frontotemporal Dementias". Continuum (Minneapolis, Minn.). 22 (2 Dementia): 464–89. doi:10.1212/CON.0000000000000300. PMID 27042904.
- ↑ 6.0 6.1 6.2 6.3 6.4 6.5 Sivasathiaseelan, H; Marshall, CR; Agustus, JL; et al. (April 2019). "Frontotemporal Dementia: A Clinical Review". Seminars in Neurology. 39 (2): 251–263. doi:10.1055/s-0039-1683379. PMID 30925617. Archived from the original on 2021-08-28. Retrieved 2020-12-30.
- ↑ 7.0 7.1 "What is frontotemporal dementia". Dementia UK. Archived from the original on 17 April 2021. Retrieved 19 October 2020.
- ↑ 8.0 8.1 8.2 8.3 8.4 8.5 Cardarelli R, Kertesz A, Knebl JA (December 2010). "Frontotemporal dementia: a review for primary care physicians". Am Fam Physician. 82 (11): 1372–7. PMID 21121521. Archived from the original on 2021-03-04. Retrieved 2014-08-31.
- ↑ "Brain Cells for Socializing". Smithsonian. Archived from the original on 22 March 2021. Retrieved 30 October 2015.
- ↑ 10.0 10.1 "What are the Different Types of Frontotemporal Disorders?". National Institute on Aging. Archived from the original on 9 July 2021. Retrieved 1 November 2020.
- ↑ Hofmann JW, Seeley WW, Huang EJ, Kubota M (24 January 2019). "RNA Binding Proteins and the Pathogenesis of Frontotemporal Lobar Degeneration". Annual Review of Pathology. 14: 469–495. doi:10.1146/annurev-pathmechdis-012418-012955. PMC 6731550. PMID 30355151.
- ↑ 12.0 12.1 Rabinovici GD, Miller BL (May 2010). "Frontotemporal lobar degeneration: epidemiology, pathophysiology, diagnosis and management". CNS Drugs. 24 (5): 375–98. doi:10.2165/11533100-000000000-00000. PMC 2916644. PMID 20369906.
- ↑ "Focus on Frontotemporal Dementia (FTD) | National Institute of Neurological Disorders and Stroke". www.ninds.nih.gov. Archived from the original on 16 October 2021. Retrieved 3 March 2021.
- ↑ 14.0 14.1 14.2 14.3 Sleegers K, Cruts M, Van Broeckhoven C (2010). "Molecular Pathways of Frontotemporal Lobar Degeneration". Annual Review of Neuroscience. 33 (1): 71–88. doi:10.1146/annurev-neuro-060909-153144. PMID 20415586.
- ↑ 15.0 15.1 Piguet O (November 2011). "Eating disturbance in behavioural-variant frontotemporal dementia". J. Mol. Neurosci. 45 (3): 589–93. doi:10.1007/s12031-011-9547-x. PMID 21584651. S2CID 24125998.
- ↑ Neary D, Snowden J, Mann D (2005). "Frontotemporal dementia". Lancet Neurol. 4 (11): 771–80. doi:10.1016/s1474-4422(05)70223-4. PMID 16239184. S2CID 17310802.
- ↑ Kramer JH, Jurik J, Sha SJ, et al. (December 2003). "Distinctive neuropsychological patterns in frontotemporal dementia, semantic dementia, and Alzheimer disease". Cogn Behav Neurol. 16 (4): 211–8. doi:10.1097/00146965-200312000-00002. PMID 14665820. S2CID 46800951.
- ↑ Olney RK, Murphy J, Forshew D, et al. (December 2005). "The effects of executive and behavioral dysfunction on the course of ALS". Neurology. 65 (11): 1774–7. doi:10.1212/01.wnl.0000188759.87240.8b. PMID 16344521. S2CID 11672234.
- ↑ Luc Buée; André Delacourte (1999). "Comparative Biochemistry of Tau in Progressive Supranuclear Palsy, Corticobasal Degeneration, FTDP-17 and Pick's Disease". Brain Pathology. 9 (4): 681–693. doi:10.1111/j.1750-3639.1999.tb00550.x. PMID 10517507. S2CID 10711305.[permanent dead link]
- ↑ Hardy, John; Momeni, Parastoo; Traynor, Bryan J. (April 2006). "Frontal temporal dementia: dissecting the aetiology and pathogenesis" (PDF). Brain. 26. 129 (4): 830–831. doi:10.1093/brain/awl035. PMID 16543401. Archived from the original on August 28, 2021. Retrieved July 8, 2017.
- ↑ Convery R, Mead S, Rohrer JD (2018) Clinical, genetic and neuroimaging features of frontotemporal dementia. Neuropathol Appl Neurobiol
- ↑ Dobson-Stone C, Hallupp M, Shahheydari H, Ragagnin AMG, Chatterton Z, Carew-Jones F, Shepherd CE, Stefen H, Paric E, Fath T, Thompson EM, Blumbergs P, Short CL, Field CD, Panegyres PK, Hecker J, Nicholson G, Shaw AD, Fullerton JM, Luty AA, Schofield PR, Brooks WS, Rajan N, Bennett MF, Bahlo M, Landers JE, Piguet O, Hodges JR, Halliday GM, Topp SD, Smith BN, Shaw CE, McCann E, Fifita JA, Williams KL, Atkin JD, Blair IP, Kwok JB (2020). "CYLD is a causative gene for frontotemporal dementia - amyotrophic lateral sclerosis". Brain. 143 (3): 783–799. doi:10.1093/brain/awaa039. PMC 7089666. PMID 32185393.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ↑ Liscic RM, Storandt M, Cairns NJ, Morris JC (April 2007). "Clinical and psychometric distinction of frontotemporal and Alzheimer dementias". Arch. Neurol. 64 (4): 535–40. doi:10.1001/archneur.64.4.535. PMID 17420315.
- ↑ Jonathan D. Rohrer; Tammaryn Lashley; Jonathan M. Schott; Jane E. Warren; Simon Mead; Adrian M. Isaacs; Jonathan Beck; John Hardy; Rohan de Silva; Elizabeth Warrington; Claire Troakes; Safa Al-Sarraj; Andrew King; Barbara Borroni; Matthew J. Clarkson; Sebastien Ourselin; Janice L. Holton; Nick C. Fox; Tamas Revesz; Martin N. Rossor & Jason D. Warren (corresponding author) (September 2011). "Clinical and neuroanatomical signatures of tissue pathology in frontotemporal lobar degeneration". Brain. 134 (9): 2565–2581. doi:10.1093/brain/awr198. PMC 3170537. PMID 21908872.
- ↑ van der Zee, Julie; Van Broeckhoven, Christine (7 January 2014). "Dementia in 2013: Frontotemporal lobar degeneration—building on breakthroughs". Nature Reviews Neurology. 10 (2): 70–72. doi:10.1038/nrneurol.2013.270. PMID 24394289.
- ↑ 26.0 26.1 26.2 Snowden JS, Neary D, Mann DM (February 2002). "Frontotemporal dementia". Br J Psychiatry. 180 (2): 140–3. doi:10.1192/bjp.180.2.140. PMID 11823324.
- ↑ Schroeter ML, Raczka KK, Neumann J, von Cramon DY (2008). "Neural networks in frontotemporal dementia – A meta-analysis". Neurobiology of Aging. 29 (3): 418–426. doi:10.1016/j.neurobiolaging.2006.10.023. PMID 17140704. S2CID 9039002.
- ↑ Rahman S, Sahakian BJ, Hodges JR, Rogers RD, Robbins TW (August 1999). "Specific cognitive deficits in mild frontal variant frontotemporal dementia" (PDF). Brain. 122 (Pt 8): 1469–93. doi:10.1093/brain/122.8.1469. PMID 10430832. Archived (PDF) from the original on 2021-08-28. Retrieved 2017-07-09.
- ↑ Schroeter ML, Raczka KK, Neumann J, von Cramon DY (2007). "Towards a nosology for frontotemporal lobar degenerations – A meta-analysis involving 267 subjects". NeuroImage. 36 (3): 497–510. doi:10.1016/j.neuroimage.2007.03.024. PMID 17478101. S2CID 130161.
- ↑ Steinbart EJ, Smith CO, Poorkaj P, Bird TD (November 2001). "Impact of DNA testing for early-onset familial Alzheimer disease and frontotemporal dementia". Arch. Neurol. 58 (11): 1828–31. doi:10.1001/archneur.58.11.1828. PMID 11708991.
- ↑ "Pick Disease". Medline Plus. Archived from the original on 5 July 2017. Retrieved 29 June 2017.
- ↑ 32.0 32.1 Rascovsky K, Hodges JR, Knopman D, et al. (September 2011). "Sensitivity of revised diagnostic criteria for the behavioural variant of frontotemporal dementia". Brain. 134 (Pt 9): 2456–77. doi:10.1093/brain/awr179. PMC 3170532. PMID 21810890.
- ↑ Krueger, C.E.; Bird, A.C.; Growdon, M.E.; Jang, J.Y.; Miller, B.L. & Kramer, J.K. (2009). "Conflict in monitoring early frontotemporal dementia". Neurology. 73 (5): 349–55. doi:10.1212/wnl.0b013e3181b04b24. PMC 2725928. PMID 19652138.
- ↑ 34.0 34.1 34.2 Torralva, T; Roca, M; Gleichgerrcht, E; Bekinschtein, T & Manes, F (2009). "A neuropsychological battery to detect specific executive and social cognitive impairments in early frontotemporal dementia" (PDF). Brain. 132 (5): 1299–1309. doi:10.1093/brain/awp041. PMID 19336463. Archived (PDF) from the original on 2021-08-28. Retrieved 2017-07-09.
- ↑ 35.0 35.1 Beer, J.S.; John, O.P.; Scabini, D. & Knight, R.T. (2006). "Orbitofrontal cortex and Social behaviour: Integrating Self-monitoring and Emotion-cognition interactions". Journal of Cognitive Neuroscience. 18 (6): 871–888. CiteSeerX 10.1.1.527.3607. doi:10.1162/jocn.2006.18.6.871. PMID 16839295. S2CID 13590871.
- ↑ Damasio, A.R.; Everitt, B. J.; Bishop, D. (29 October 1996). "The Somatic marker hypothesis and the possible functions of the prefrontal cortex". Phil. Trans. R. Soc. Lond. B. 351 (1346): 1413–20. doi:10.1098/rstb.1996.0125. PMID 8941953. S2CID 1841280.
- ↑ 37.0 37.1 Olney, Nicholas T.; Spina, Salvatore; Miller, Bruce L. (May 2017). "Frontotemporal Dementia". Neurologic Clinics. 35 (2): 339–374. doi:10.1016/j.ncl.2017.01.008. ISSN 0733-8619. PMC 5472209. PMID 28410663.
- ↑ "Frontotemporal Dementia Information Page | National Institute of Neurological Disorders and Stroke". www.ninds.nih.gov. Archived from the original on 2020-12-16. Retrieved 2020-12-12.
- ↑ Bang, J; Spina, S; Miller, BL (24 October 2015). "Frontotemporal dementia". Lancet. 386 (10004): 1672–82. doi:10.1016/S0140-6736(15)00461-4. PMC 5970949. PMID 26595641.
- ↑ Gaillard, Frank. "Pick bodies | Radiology Reference Article | Radiopaedia.org". Radiopaedia. Archived from the original on 28 November 2020. Retrieved 12 March 2021.
- ↑ 41.0 41.1 41.2 Cairns, NJ; Grossman, M; Arnold, SE; et al. (26 October 2004). "Clinical and neuropathologic variation in neuronal intermediate filament inclusion disease". Neurology. 63 (8): 1376–84. doi:10.1212/01.wnl.0000139809.16817.dd. PMC 3516854. PMID 15505152.
- ↑ 42.0 42.1 Armstrong, RA; Gearing, M; Bigio, EH; et al. (November 2011). "Spatial patterns of FUS-immunoreactive neuronal cytoplasmic inclusions (NCI) in neuronal intermediate filament inclusion disease (NIFID)". Journal of Neural Transmission. 118 (11): 1651–7. doi:10.1007/s00702-011-0690-x. PMC 3199334. PMID 21792670.
- ↑ Ito, H (December 2014). "Basophilic inclusions and neuronal intermediate filament inclusions in amyotrophic lateral sclerosis and frontotemporal lobar degeneration". Neuropathology. 34 (6): 589–95. doi:10.1111/neup.12119. PMID 24673472. S2CID 19275295.
- ↑ Swartz JR, Miller BL, Lesser IM, Darby AL (May 1997). "Frontotemporal dementia: treatment response to serotonin selective reuptake inhibitors". J Clin Psychiatry. 58 (5): 212–6. doi:10.4088/jcp.v58n0506. PMID 9184615.
- ↑ "Medications for behavioral symptoms". ucsf.edu. Archived from the original on 21 September 2015. Retrieved 30 October 2015.
- ↑ Kertesz A (June 2004). "Frontotemporal dementia/Pick's disease". Arch. Neurol. 61 (6): 969–71. doi:10.1001/archneur.61.6.969. PMID 15210543.
- ↑ Samson, K. (2002). "Hypotension May Cause Frontotemporal Dementia". Neurology Today. 2 (9): 35–36. doi:10.1097/00132985-200209000-00013.
- ↑ PICK, A. (1892). "Uber die Beziehungen der senilen Hirnatrophie zur Aphasie". Prag Med Wochenschr. 17: 165–167. Archived from the original on 2021-02-04. Retrieved 2019-11-20.
- ↑ Rascovsky, Katya; Hodges, John R.; Knopman, David; Mendez, Mario F.; Kramer, Joel H.; Neuhaus, John; van Swieten, John C.; Seelaar, Harro; Dopper, Elise G. P.; Onyike, Chiadi U.; Hillis, Argye E. (September 2011). "Sensitivity of revised diagnostic criteria for the behavioural variant of frontotemporal dementia". Brain. 134 (9): 2456–2477. doi:10.1093/brain/awr179. ISSN 0006-8950. PMC 3170532. PMID 21810890.
- ↑ Goldstein, Richard. Don Cardwell, 72, Pitcher for 1969 Mets, Is Dead Archived 2018-12-21 at the Wayback Machine. The New York Times. January 16, 2008.
- ↑ "Sugarloaf frontman Jerry Corbetta dead at 68 – The Denver Post". 2016-09-20. Archived from the original on 2016-10-25. Retrieved 2016-10-29.
- ↑ Knuth, Donald E. (December 2003). "Robert W Floyd, In Memoriam". ACM SIGACT News. 34 (4): 3–13. doi:10.1145/954092.954488. S2CID 35605565. Archived from the original on 2017-04-22. Retrieved 2020-12-12.
- ↑ Sipple, George (March 6, 2009). "Fans mourn loss of "Mrs. Hockey," Colleen Howe". The Detroit Free Press. Archived from the original on October 4, 2015. Retrieved October 3, 2015.
- ↑ Sportsteam. "Grimsby Town legend Kevin Moore passes away". Archived from the original on 2013-05-03. Retrieved 2013-04-29.
- ↑ "Support for Valiants hero Ernie Moss after he is diagnosed with Pick's Disease". The Sentinel. 11 October 2014. Archived from the original on 2 November 2014. Retrieved 11 October 2014.
- ↑ Sally Potter (22 January 2013). "Nic Potter obituary | Music | guardian.co.uk". London: Guardian. Archived from the original on 1 February 2013. Retrieved 22 January 2013.
- ↑ "The Sexy, Proto-Feminist Art Of Christina Ramberg's Tragically Short Life". www.wbur.org. Archived from the original on 2020-10-22. Retrieved 2020-12-12.
- ↑ Matt Roper (2011-04-26). "Robbie Savage's tears for his dad - and the end of his football career". mirror.co.uk. Archived from the original on 2012-01-29. Retrieved 2011-05-11.
- ↑ Gordon Rayner, "Sir Nicholas Wall, once Britain's top family law judge, commits suicide after dementia diagnosis" Archived 2020-11-12 at the Wayback Machine, The Daily Telegraph, 23 February 2017.
- ↑ [1] Archived 2020-09-20 at the Wayback Machine, ‘’The Daily Telegraph’’, 13 Aug 2020
- ↑ Farooq, Mohammad Omar. "Kazi Nazrul Islam: Illness and Treatment". nazrul.org. Archived from the original on 29 May 2015. Retrieved 26 March 2016.
External links
- OMIM on Frontotemporal Dementia and/or Amyotrophic Lateral Sclerosis and Chromosome 9 Open Reading Frame 72; C9ORF72 Archived 2015-09-24 at the Wayback Machine
| Classification | |
|---|---|
| External resources |
- Pages with script errors
- Webarchive template wayback links
- CS1 errors: missing periodical
- All articles with dead external links
- Articles with dead external links from March 2022
- Articles with invalid date parameter in template
- Articles with permanently dead external links
- CS1 maint: multiple names: authors list
- All articles with unsourced statements
- Articles with unsourced statements from December 2020
- Cognitive disorders
- Dementia
- Frontal lobe
- Psychiatric diagnosis
- Temporal lobe
- RTT