Isosorbide mononitrate
 | |
| Names | |
|---|---|
| Trade names | Monoket, Imdur, others |
| |
| Clinical data | |
| Pregnancy category | |
| Routes of use | By mouth |
| Defined daily dose | 40 mg[2] |
| External links | |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a682348 |
| Legal | |
| License data | |
| Legal status |
|
| Pharmacokinetics | |
| Bioavailability | >95% |
| Protein binding | <5% |
| Metabolism | Hepatic |
| Elimination half-life | 5 hours |
| Excretion | Kidney (93%) |
| Chemical and physical data | |
| Formula | C6H9NO6 |
| Molar mass | 191.139 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Isosorbide mononitrate, sold under many brand names, is a medication used for heart-related chest pain (angina), heart failure, and esophageal spasms.[3] It can be used both to treat and to prevent heart-related chest pain; however, it is generally less preferred than beta blockers or calcium channel blockers.[3] It is taken by mouth.[3]
Common side effects include headache, low blood pressure with standing, blurry vision, and skin flushing.[3] Serious side effects may include low blood pressure especially if also exposed to PDE5 inhibitors such as sildenafil.[3] Use is not recommended in pregnancy.[4] It is believed to work by relaxing smooth muscle within blood vessels.[3]
It was patented in 1971 and approved for medical use in 1981.[5] It is available as a generic medication.[4] A month's supply in the United Kingdom costs the NHS about one pound sterling per month as of 2019.[4] In the United States the wholesale cost of this amount is about US$4.30.[6] In 2017, it was the 111th most commonly prescribed medication in the United States, with more than six million prescriptions.[7][8]
Medical uses
Isosorbide mononitrate is a nitrate-class drug used for the prevention of angina pectoris.[9]
Dosage
The defined daily dose is 40 mg by mouth.[2]
Side effects
The following Side effects have been reported:
Very common: Headache predominates (up to 30%) necessitating withdrawal of 2 to 3% of patients, but the incidence reduces rapidly as treatment continues.[9]
Common: Tiredness, sleep disturbances (6%) and gastrointestinal disturbances (6%) have been reported during clinical trials with isosorbide mononitrate modified-release tablets, but at a frequency no greater than for placebo. Hypotension (4 to 5%), poor appetite (2.5%), nausea (1%)[9]
Adverse effects associated with the clinical use of the drug are as expected with all nitrate preparations. They occur mainly in the early stages of treatment.[9]
Hypotension (4%) with symptoms such as dizziness and nausea (1%) have been reported. In general, these symptoms disappear during long-term treatment.[9]
Other reactions that have been reported with isosorbide mononitrate-modified release tablets include tachycardia, vomiting, diarrhoea, vertigo, and heartburn.[9]
Interactions
- Sildenafil (Viagra). Concomitant administration of isosorbide mononitrate and sildenafil (Viagra) or other phosphodiesterase inhibitors (Tadalafil and Udenafil) can potentiate the vasodilatory effect of isosorbide mononitrate with the potential result of serious side-effects such as syncope or myocardial infarction. Life-threatening hypotension may also occur. Therefore, sildenafil should not be given to patients already receiving isosorbide mononitrate therapy.[9]
- Sulfhydryl-containing compounds. The metabolism of organic nitrates to nitric oxide is dependent on the presence of sulfhydryl groups in the muscle. The combination of oral N-acetylcysteine and a single dose of sustained-release isosorbide mononitrate 60 mg significantly prolonged the total exercise time in patients with angina pectoris and angiographically proven significant coronary artery disease, when compared with isosorbide mononitrate alone. Concomitant administration of other exogenous sources of sulfhydryl groups such as methionine and captopril may produce a similar interaction.[medical citation needed]
- Phenylalkylamine calcium antagonists. The addition of a calcium channel blocker of the verapamil type, such as gallopamil 75 mg, has been shown to further improve left ventricular functional parameters when given in combination with isosorbide mononitrate in a sustained-release formulation.[medical citation needed]
- Propranolol. The addition of isosorbide mononitrate to propranolol treatment in patients with cirrhosis and portal hypertension caused a marked fall in portal pressure, a reduction in hepatic blood flow, cardiac output and mean arterial blood pressure, but no additional change in azygos blood flow. The additional effect of isosorbide mononitrate was especially evident in patients whose portal pressure was not reduced by propranolol.[medical citation needed]
- Calcium antagonists (general). Marked symptomatic orthostatic hypotension has been reported when calcium antagonists and organic nitrates were used in combination. Dose adjustments of either class of agent may be necessary.[9]
Brand names
It is sold in the US by Lannett Company, under the trade name Monoket,[9][10] and was also sold in the US under the name Imdur,[11] and marketed in the UK under the trade names: Isotard, Monosorb, Chemydur. In India, this drug is available under the brand names of Ismo, Imdur, Isonorm, Monotrate, Solotrate, and Monit. In Russia it is occasionally used under the brand names Monocinque and Pektrol. In Australia, this drug is available under the brand name Duride.[citation needed]
Society and culture
Cost
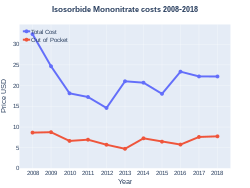
A month's supply in the United Kingdom costs the NHS about one pound sterling per month as of 2019.[4] In the United States the wholesale cost of this amount is about US$4.30.[6]In 2017, it was the 111th most commonly prescribed medication in the United States, with more than six million prescriptions.[7][8]
-
Isosorbide mononitrate costs (US)
-
Isosorbide mononitrate prescriptions (US)
References
- ↑ 1.0 1.1 "Isosorbide mononitrate Use During Pregnancy". Drugs.com. 28 February 2020. Archived from the original on 25 January 2021. Retrieved 14 April 2020.
- ↑ 2.0 2.1 "WHOCC - ATC/DDD Index". www.whocc.no. Archived from the original on 11 July 2020. Retrieved 6 September 2020.
- ↑ 3.0 3.1 3.2 3.3 3.4 3.5 "Isosorbide Dinitrate/Mononitrate Monograph for Professionals". Drugs.com. American Society of Health-System Pharmacists. Archived from the original on 21 December 2016. Retrieved 3 March 2019.
- ↑ 4.0 4.1 4.2 4.3 British national formulary : BNF 76 (76 ed.). Pharmaceutical Press. 2018. pp. 219–220. ISBN 9780857113382.
- ↑ Fischer, Jnos; Ganellin, C. Robin (2006). Analogue-based Drug Discovery. John Wiley & Sons. p. 454. ISBN 9783527607495. Archived from the original on 2016-12-20. Retrieved 2019-02-28.
- ↑ 6.0 6.1 "NADAC as of 2019-02-27". Centers for Medicare and Medicaid Services. Archived from the original on 2019-03-06. Retrieved 3 March 2019.
- ↑ 7.0 7.1 "The Top 300 of 2020". ClinCalc. Archived from the original on 12 February 2021. Retrieved 11 April 2020.
- ↑ 8.0 8.1 "Isosorbide Mononitrate - Drug Usage Statistics". ClinCalc. Archived from the original on 8 July 2020. Retrieved 11 April 2020.
- ↑ 9.0 9.1 9.2 9.3 9.4 9.5 9.6 9.7 9.8 "Monoket- isosorbide mononitrate tablet". DailyMed. 3 February 2015. Archived from the original on 28 August 2021. Retrieved 14 April 2020.
- ↑ "Monoket: FDA-Approved Drugs". U.S. Food and Drug Administration (FDA). Archived from the original on 29 June 2017. Retrieved 14 April 2020.
- ↑ "Imdur: FDA-Approved Drugs". U.S. Food and Drug Administration (FDA). Archived from the original on 29 August 2021. Retrieved 14 April 2020.
External links
| Identifiers: |
|
|---|
- "Isosorbide mononitrate". Drug Information Portal. U.S. National Library of Medicine. Archived from the original on 2020-07-10. Retrieved 2020-04-15.
- Pages using duplicate arguments in template calls
- Chemical articles with unknown parameter in Infobox drug
- Chemical articles without CAS registry number
- Articles without EBI source
- Chemical pages without ChemSpiderID
- Chemical pages without DrugBank identifier
- Articles without KEGG source
- Articles without UNII source
- Drugs missing an ATC code
- All articles with unsourced statements
- Articles with unsourced statements from April 2020
- Articles with invalid date parameter in template
- RTT
- Nitrate esters
- Antianginals
- Furofurans
- Secondary alcohols
- AstraZeneca brands