Anidulafungin
 | |
| Names | |
|---|---|
| Pronunciation | /eɪˌnɪdjʊləˈfʌndʒɪn/ ay-NID-yuu-lə-FUN-jin |
| Trade names | Eraxis, Ecalta, others |
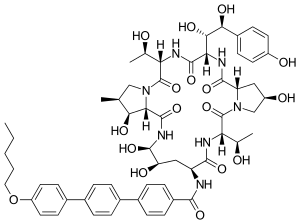
| Other names | (4R,5S)-4,5-Dihydroxy-N2-[[4''-(pentyloxy)-p-terphenyl-4-yl]carbonyl]-L-ornithyl-L-threonyl-trans-4-hydroxy-L-prolyl-(S)-4-hydroxy-4-(p-hydroxyphenyl)-L-threonyl-L-threonyl-(3S,4S)-3-hydroxy-4-methyl-L-proline cyclic (6→1)-peptide[1] 1-[(4R,5R)-4,5-Dihydroxy-N2-[[4''-(pentyloxy)[1',1':4',1''-terphenyl]-4-yl]carbonyl]-L-ornithine]echinocandin B[2] |
| |
| Clinical data | |
| Drug class | Echinocandin[3] |
| Main uses | Invasive candidiasis, esophageal candidiasis[3][4] |
| Side effects | Diarrhea, nausea, low potassium[3] |
| Pregnancy category |
|
| Routes of use | Intravenous |
| External links | |
| AHFS/Drugs.com | Monograph |
| Legal | |
| License data |
|
| Legal status |
|
| Pharmacokinetics | |
| Bioavailability | 100% (intravenous use only) |
| Protein binding | Extensive (>99%) |
| Metabolism | Liver metabolism not observed, CYP system not involved |
| Elimination half-life | 27 hours; 40–50 hours (terminal) |
| Excretion | Feces (~30%), urine (<1%) |
| Chemical and physical data | |
| Formula | C58H73N7O17 |
| Molar mass | 1140.254 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Anidulafungin, sold under the brand name Eraxis and Ecalta among others, is an antifungal used to treat invasive candidiasis and esophageal candidiasis.[3][4] It is given by injection into a vein.[5]
Common side effects include diarrhea, nausea, and low potassium.[3] Other side effects may include liver problems and anaphylaxis.[4] Use in pregnancy may harm the baby.[4] It is an echinocandin and works by interfering with the production of a component of the fungal cell wall known as (1→3)-β-D-glucan.[3]
Anidulafungin was approved for medical use in the United States in 2006 and Europe in 2007.[4][3] It is available as a generic medication.[5] It is on the World Health Organization's List of Essential Medicines as an alternative to micafungin.[6] In the United Kingdom 100 mg costs the NHS about £300 as of 2021.[5] This amount in the United States is about 200 USD.[7]
Medical uses
- Candidemia and other forms of invasive Candida infections (intra-abdominal abscess and peritonitis)
- Esophageal candidiasis
Anidulafungin has not been studied in endocarditis, osteomyelitis, and meningitis due to Candida, and has not been studied in sufficient numbers of neutropenic patients to determine efficacy in this group.[2]
Pharmacology
Anidulafungin significantly differs from other antifungals in that it undergoes chemical degradation to inactive forms at body pH and temperature. Because it does not rely on enzymatic degradation or hepatic or renal excretion, the drug is safe to use in patients with any degree of hepatic or renal impairment.[8]
Volume of distribution: 30–50 L.
Anidulafungin is not evidently metabolized by the liver. This specific drug undergoes slow chemical hydrolysis to an open-ring peptide which lacks antifungal activity. The half-life of the drug is 27 hours. About 30% is excreted in the feces (10% as unchanged drug). Less than 1% is excreted in the urine.[9][10][11]
Mechanism of action
Anidulafungin inhibits glucan synthase, an enzyme important in the formation of (1→3)-β-D-glucan, a major fungal cell wall component. Glucan synthase is not present in mammalian cells, so it is an attractive target for antifungal activity.[12]
Manufacture
Anidulafungin is manufactured via semisynthesis. The starting material is echinocandin B (a lipopeptide fermentation product of Aspergillus nidulans or the closely related species, A. rugulosus), which undergoes deacylation (cleavage of the linoleoyl side chain) by the action of a deacylase enzyme from the bacterium Actinoplanes utahensis;[13] in three subsequent synthetic steps, including a chemical reacylation, the antifungal drug anidulafungin[12][14] is synthesized.
History
Anidulafungin was originally discovered at Lilly laboratories by Turner and Debono and licensed to Vicuron Pharmaceuticals who submitted it to the FDA.[15] Pfizer acquired the drug upon its acquisition of Vicuron in the fall of 2005.[16] Pfizer gained approval by the Food and Drug Administration (FDA) on February 21, 2006.[17]
References
- ↑ "International Nonproprietary Names for Pharmaceutical Substances (INN). Recommended International Nonproprietary names: List 43" (PDF). World Health Organization. 2000. Archived (PDF) from the original on 18 May 2016. Retrieved 11 November 2016.
- ↑ 2.0 2.1 "Eraxis (anidulafungin) for Injection, for Intravenous Use. Full Prescribing Information". Roerig (Division of Pfizer, Inc.), New York, NY 10017. Archived from the original on 30 March 2016. Retrieved 11 November 2016.
- ↑ 3.0 3.1 3.2 3.3 3.4 3.5 3.6 "Ecalta". Archived from the original on 9 August 2021. Retrieved 14 January 2022.
- ↑ 4.0 4.1 4.2 4.3 4.4 "DailyMed - ERAXIS- anidulafungin injection, powder, lyophilized, for solution". dailymed.nlm.nih.gov. Archived from the original on 14 May 2021. Retrieved 14 January 2022.
- ↑ 5.0 5.1 5.2 BNF 81: March-September 2021. BMJ Group and the Pharmaceutical Press. 2021. p. 631. ISBN 978-0857114105.
- ↑ World Health Organization (2021). World Health Organization model list of essential medicines: 22nd list (2021). Geneva: World Health Organization. hdl:10665/345533. WHO/MHP/HPS/EML/2021.02.
- ↑ "Anidulafungin Prices, Coupons & Savings Tips - GoodRx". GoodRx. Retrieved 14 January 2022.
- ↑ "Eraxis at RxList". 2009-06-24. Archived from the original on 2018-01-16. Retrieved 2009-08-01.
- ↑ Trissel LA and Ogundele AB, "Compatibility of Anidulafungin With Other Drugs During Simulated Y-Site Administration,"Am J Health-Sys Pharm, 2005, 62:834-7.
- ↑ Vazquez JA, "Anidulafungin: A New Echinocandin With a Novel Profile," Clin Ther, 2005, 27(6):657-73.
- ↑ Walsh TJ, Anaissie EJ, Denning DW, et al., "Treatment of Aspergillosis: Clinical Practice Guidelines of the Infectious Diseases Society of America," Clin Infect Dis, 2008, 46(3):327-60
- ↑ 12.0 12.1 Denning DW (1997). "Echinocandins and pneumocandins - a new antifungal class with a novel mode of action". J Antimicrob Chemother. 40 (5): 611–614. doi:10.1093/jac/dkf045. PMID 9421307.
- ↑ Lei Shao; Jian Li; Aijuan Liu; Qing Chang; Huimin Lin; Daijie Chen (2013). "Efficient Bioconversion of Echinocandin B to Its Nucleus by Overexpression of Deacylase Genes in Different Host Strains". Applied and Environmental Microbiology. 79 (4): 1126–1133. Bibcode:2013ApEnM..79.1126S. doi:10.1128/AEM.02792-12. PMC 3568618. PMID 23220968.
- ↑ "Anidulafungin EMA Europa" (PDF). Archived (PDF) from the original on 2018-06-19. Retrieved 2021-10-05.
- ↑ PRNewswire. Vicuron Pharmaceuticals Files New Drug Application (NDA) for Anidulafungin for Treatment of Invasive Candidiasis/Candidemia Archived May 16, 2012, at the Wayback Machine 08-18-2005.
- ↑ PRNewswire. Vicuron Pharmaceuticals Stockholders Approve Merger With Pfizer Archived May 16, 2012, at the Wayback Machine 08-15-2005
- ↑ "FDA Approves New Treatment for Fungal Infections". FDA News Release. Food and Drug Administration. 2006-02-21. Archived from the original on 10 July 2009. Retrieved 2009-08-01.
External links
| Identifiers: |
|
|---|
- Pages using duplicate arguments in template calls
- Webarchive template wayback links
- Drugs with non-standard legal status
- Chemical articles with unknown parameter in Infobox drug
- Chemical articles without CAS registry number
- Articles without EBI source
- Chemical pages without ChemSpiderID
- Chemical pages without DrugBank identifier
- Articles without KEGG source
- Articles without UNII source
- Drugs missing an ATC code
- Drug has EMA link
- Drugboxes which contain changes to verified fields
- Drugboxes which contain changes to watched fields
- Articles with changed KEGG identifier
- Articles with changed EBI identifier
- Antifungals
- Echinocandins
- Pfizer brands
- World Health Organization essential medicines (alternatives)
- RTT