Isavuconazonium
 | |
| Names | |
|---|---|
| Trade names | Cresemba |
| Other names | Isavuconazonium sulfate |
| Clinical data | |
| Drug class | Antifungal (triazole)[1] |
| Main uses | Invasive aspergillosis and mucormycosis[2][3] |
| Side effects | Liver problems, nausea, shortness of breath, abdominal pain, diarrhea, headache, low potassium, rash[2] |
| Pregnancy category | |
| Routes of use | By mouth, intravenous (IV) |
| External links | |
| AHFS/Drugs.com | Monograph |
| Legal | |
| Legal status | |
| Chemical and physical data | |
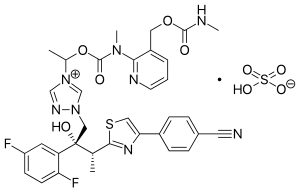
| Formula | C35H35F2N8O5S |
| Molar mass | 717.77 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Isavuconazonium, sold under the brand name Cresemba, is a medication used to treat invasive aspergillosis and mucormycosis.[2][3] For mucormycosis, it is used when amphotericin B is not suitable.[5] It may be taken by mouth or given by gradual injection into a vein.[3]
Common side effects include liver problems, nausea, shortness of breath, abdominal pain, diarrhea, headache, low potassium and rash.[2] Other side effects may include anaphylaxis and short QT.[3][6] Use in pregnancy may harm the baby.[3] It is a antifungal medication of the triazole class and works after being converted into isavuconazole in the body.[1]
Isavuconazonium was approved for medical use in the United States and Europe in 2015.[6][2] In the United Kingdom a vial of 200 mg cost the NHS about £300 as of 2021.[5] This amount in the United States costs about 350 USD.[7]
Medical uses
Isavuconazonium is used to treat invasive aspergillosis and invasive mucormycosis in adults ages 18 years and older. It is available in a capsule for administration by mouth and as a powder for administration via infusion.[3][8][9][10][11]
Dosage
It is given at an initial dose of 372 mg of the sulfate or 200 mg of isavuconazonium (2 capsules or one vial) three times per day for two days.[3] This is than decreased to 372 mg once per day.[3]
Contraindications
Isavuconazonium should not be given to anyone with a history of allergic reaction to it, nor to anyone taking ketoconazole or high-dose ritonavir. It should not be given to anyone with a family history of short QT syndrome. It has not been tested in children.[8][9]
In preclinical studies, isavuconazonium caused birth defects; it has not been tested in pregnant women. Women who are pregnant should not take it and women taking it should not get pregnant. It is excreted in breast milk so people should not breast feed while taking it.[8][9]
Side effects
Common adverse effects (occurring in between 1 and 10% of people) include low potassium, decreased appetite, delirium, headache, sleepiness, vein inflammation, difficulty breathing, acute respiratory failure, vomiting, diarrhea, nausea, stomach pain, elevated results in liver function tests, rash, itchy skin, kidney failure, chest pain, and fatigue. There are several uncommon side effects as well.[8][9]
Interactions
Isavuconazonium is converted into isavuconazole inside the body, and isavuconazole is a substrate for CYP3A4 or CYP3A5. Many other medications inhibit or induce those two enzymes, and isavuconazonium should not be administered with them. Inducers result in levels of isavuconazole that are too low and won't work, and inhibitors can cause high levels of isavuconazole which will in turn cause increased adverse events and toxicity. Likewise isavuconazonium can interfere with appropriate dosing of other drugs that are substrates for those enzymes.[8][9]
In addition, isavuconazole induces CYP2B6 and can decrease the amount of drugs that are metabolized by the enzyme. Isavuconazole inhibits P-glycoprotein (P-gp), BCRP, SLC22A2, and uridine diphosphate-glucuronosyltransferases, each of which remove drugs from circulation; isavuconazonium will increase the amount of drugs that are affected by those proteins and may increase their toxicities.[8][9]
Pharmacology
After oral or intravenous (IV) administration, isavuconazonium is rapidly hydrolysed by esterases in blood or the gastrointestinal tract to the active form, isavuconazole.[8][9][12]
Isavuconazole works by inhibition of lanosterol 14α-demethylase, the enzyme responsible for converting lanosterol to ergosterol by demethylation. The resulting depletion of ergosterol and buildup of lanosterol compromise the structure of the fungal cell membrane. Mammalian cells are resistant to demethylation inhibition by azoles, making the drug effects specific to fungi.[8][9][12]
Chemistry
Isavuconazonium comprises an N-(3-acetoxypropyl)-N-methylamino-carboxymethyl group linked through an ester moiety to the triazole nitrogen in isavuconazole.[13][14][15]
History
Isavuconazole and isavuconazonium were discovered in Japan by researchers at Roche's research center in Kamakura.[13][16] Basilea Pharmaceutica, which had been spun out of Roche to develop antimicrobial assets, developed isavuconazonium through Phase II clinical trials. In February 2010, Basilea partnered with Astellas Pharma to complete Phase III trials, obtain regulatory approvals, and market the drug. In 2013 and 2014, the partners won orphan drug designation in the US for isavuconazonium for treating invasive aspergillosis, mucormycosis, and invasive candidiasis.[12][17][18][19]
In 2014, Basilea and Astellas amended the agreement to give Astellas sole marketing authority in North America, and Basilea the rights to market in the rest of the world.[20]
The U.S. Food and Drug Administration (FDA) granted approval in June 2015,[12][21] and the European Medicines Agency (EMA) approved it in October 2015.[22][8][9]
In 2017, Basilea licensed rights to Pfizer to sell in most of Europe and a few other countries.[23]
References
- ↑ 1.0 1.1 Wilkes, Gail M.; Barton-Burke, Margaret (2019). 2020-2021 Oncology Nursing Drug Handbook. Jones & Bartlett Learning. pp. 1874–1876. ISBN 978-1-284-17132-7. Archived from the original on 2021-09-05. Retrieved 2021-09-05.
- ↑ 2.0 2.1 2.2 2.3 2.4 "Cresemba". Archived from the original on 10 August 2020. Retrieved 28 November 2021.
- ↑ 3.0 3.1 3.2 3.3 3.4 3.5 3.6 3.7 3.8 "Cresemba- isavuconazonium sulfate capsule Cresemba- isavuconazonium sulfate injection, powder, lyophilized, for solution". DailyMed. 2 December 2019. Archived from the original on 30 March 2021. Retrieved 26 August 2020.
- ↑ 4.0 4.1 "Isavuconazonium (Cresemba) Use During Pregnancy". Drugs.com. 31 January 2020. Archived from the original on 2 December 2020. Retrieved 26 August 2020.
- ↑ 5.0 5.1 BNF 81: March-September 2021. BMJ Group and the Pharmaceutical Press. 2021. p. 635. ISBN 978-0857114105.
- ↑ 6.0 6.1 "Isavuconazonium Monograph for Professionals". Drugs.com. Archived from the original on 25 January 2021. Retrieved 26 November 2021.
- ↑ "Cresemba Prices, Coupons & Patient Assistance Programs". Drugs.com. Archived from the original on 10 April 2020. Retrieved 28 November 2021.
- ↑ 8.0 8.1 8.2 8.3 8.4 8.5 8.6 8.7 8.8 "Cresemba 100 mg hard capsules - Summary of Product Characteristics (SPC)". UK Electronic Medicines Compendium. February 2017. Archived from the original on 18 June 2017. Retrieved 3 July 2017.
- ↑ 9.0 9.1 9.2 9.3 9.4 9.5 9.6 9.7 9.8 "Cresemba 200 mg powder for concentrate for solution for infusion. Summary of Product Characteristics (SPC)". UK Electronic Medicines Compendium. February 2017. Archived from the original on 18 June 2017. Retrieved 3 July 2017.
- ↑ Donnelley MA, Zhu ES, Thompson GR (2016-06-02). "Isavuconazole in the treatment of invasive aspergillosis and mucormycosis infections". Infection and Drug Resistance. 9: 79–86. doi:10.2147/IDR.S81416. PMC 4898026. PMID 27330318.
- ↑ Miceli MH, Kauffman CA (November 2015). "Isavuconazole: A New Broad-Spectrum Triazole Antifungal Agent". Clinical Infectious Diseases. 61 (10): 1558–65. doi:10.1093/cid/civ571. PMID 26179012.
- ↑ 12.0 12.1 12.2 12.3 Pettit NN, Carver PL (July 2015). "Isavuconazole: A New Option for the Management of Invasive Fungal Infections". The Annals of Pharmacotherapy. 49 (7): 825–42. doi:10.1177/1060028015581679. PMID 25940222. Archived from the original on 2021-09-05. Retrieved 2021-05-06.
- ↑ 13.0 13.1 Guinea J, Bouza E (December 2008). "Isavuconazole: a new and promising antifungal triazole for the treatment of invasive fungal infections". Future Microbiology. 3 (6): 603–15. doi:10.2217/17460913.3.6.603. PMID 19072177.
- ↑ "Proposed INN: List 96" (PDF). WHO Drug Information. 20 (4). 2006. Archived (PDF) from the original on 2021-09-05. Retrieved 2021-05-06.
- ↑ "Recommended INN: List 58" (PDF). WHO Drug Information. 21 (3). 2007. Archived (PDF) from the original on 2020-09-17. Retrieved 2021-05-06.
- ↑ Ohwada J, Tsukazaki M, Hayase T, Oikawa N, Isshiki Y, Fukuda H, et al. (January 2003). "Design, synthesis and antifungal activity of a novel water soluble prodrug of antifungal triazole". Bioorganic & Medicinal Chemistry Letters. 13 (2): 191–6. doi:10.1016/s0960-894x(02)00892-2. PMID 12482421.
- ↑ "Isavuconazonium sulfate Orphan Drug Designations and Approvals". U.S. Food and Drug Administration (FDA). 6 May 2013. Archived from the original on 5 September 2021. Retrieved 26 August 2020.
- ↑ "Isavuconazonium sulfate Orphan Drug Designations and Approvals". U.S. Food and Drug Administration (FDA). 20 October 2014. Archived from the original on 5 September 2021. Retrieved 26 August 2020.
- ↑ "Isavuconazonium sulfate Orphan Drug Designations and Approvals". U.S. Food and Drug Administration (FDA). 25 October 2013. Archived from the original on 5 September 2021. Retrieved 26 August 2020.
- ↑ "Astellas Takes Over Isavuconazole Manufacturing and Promotion in U.S., Canada". Genetic Engineering & Biotechnology News. February 28, 2014. Archived from the original on March 5, 2014. Retrieved May 6, 2021.
- ↑ "Cresemba Capsules & Cresemba Powder for Injection". U.S. Food and Drug Administration (FDA). 3 June 2015. Archived from the original on 1 April 2021. Retrieved 26 August 2020.
- Lay summary in: (PDF) https://www.accessdata.fda.gov/drugsatfda_docs/nda/2015/207500Orig1207501Orig1s000Lbl.pdf.
{{cite web}}: Missing or empty|title=(help)
- Lay summary in: (PDF) https://www.accessdata.fda.gov/drugsatfda_docs/nda/2015/207500Orig1207501Orig1s000Lbl.pdf.
- ↑ "Cresemba". European Medicines Agency (EMA). Archived from the original on 10 August 2020. Retrieved 26 August 2020.
- ↑ Elvidge S (June 16, 2017). "Pfizer builds out anti-infective reach with Basilea deal". BioPharma Dive. Archived from the original on August 5, 2019. Retrieved May 6, 2021.
External links
| External sites: | |
|---|---|
| Identifiers: |
|
- Pages using duplicate arguments in template calls
- CS1 errors: missing title
- CS1 errors: bare URL
- Drugs with non-standard legal status
- Chemical articles with unknown parameter in Infobox drug
- Infobox drug articles with non-default infobox title
- Chemical articles without CAS registry number
- Articles without EBI source
- Chemical pages without ChemSpiderID
- Chemical pages without DrugBank identifier
- Articles without KEGG source
- Articles without UNII source
- Chemicals using indexlabels
- Drugs missing an ATC code
- Drugboxes which contain changes to verified fields
- Drugboxes which contain changes to watched fields
- Astellas Pharma
- Fluoroarenes
- Lanosterol 14α-demethylase inhibitors
- Nitriles
- Prodrugs
- Tertiary alcohols
- Thiazoles
- Triazole antifungals
- RTT