Haemophilia A
| Haemophilia A | |
|---|---|
| Other names: Hemophilia A; classical hemophilia;[1] factor VIII deficiency[2] | |
 | |
| Inherited in an X-linked manner (in majority of cases) | |
| Specialty | Haematology |
| Symptoms | Easy bruising and bleeding[1] |
| Complications | Bleeding into joints or brain[1] |
| Types | Mild, moderate, severe[1] |
| Causes | Genetic disorder[2] |
| Diagnostic method | Blood tests (aPTT, factor VIII activity level)[1] |
| Differential diagnosis | Von Willebrand disease, haemophilia B, haemophilia C, vitamin K deficiency]][1][3][4] |
| Treatment | Factor VIII concentrate[1] |
| Prognosis | Often decreased life expectancy[5] |
| Frequency | 1 in 5,000 newborn males[6] |
Haemophilia A is a bleeding disorder that occurs due to a genetic mutation.[1] Symptoms include easy bruising and bleeding.[1] Complications can include bleeding into joints or the brain.[1] Depending on the severity, the disease may become apparent between the newborn period and adulthood.[1]
It generally occurs due to an inherited mutation, which occurs in an X-linked recessive pattern.[2] About a third of cases occur due to a new mutation.[2] Rarely it may occur later in life due to the production of antibodies against factor VIII.[1] The underlying mechanism involves a lack of factor VIII, a part of the coagulation cascade.[4] Diagnosis may be suspected based on a blood test for aPTT and confirmed by a factor VIII activity level.[1] It is divided into mild, moderate, and severe depending on a person's factor levels.[2]
Treatment is with factor VIII concentrate, with recombinant factor VIII products often recommended.[1] These may be used preventatively or when bleeding occurs.[1] Desmopressin (DDAVP) or aminocaproic acid may also be used.[1][4] Fresh frozen plasma or cryoprecipitate may be used if factor VIII is not available.[4] In those who develop inhibitors, recombinant factor VIIa or emicizumab are options.[1] Despite availability of treatment life expectancy is reduced by about 30%.[5]
Haemophilia A affects about 1 in 5,000 newborn males.[6] Males are primarily affected; though, a few females may also have the disease.[1] Of X-linked recessive disorders, it is the most common; and is 5 times more common than haemophilia B.[1][4] Descriptions of inherited bleeding disorders among males date from at least the 100s AD.[7] Medical descriptions occurred in the 1800s, with the separation of haemophilia A and B occurring in 1947.[7]
Signs and symptoms

In terms of the symptoms of haemophilia A, there are internal or external bleeding episodes. Individuals with more severe haemophilia suffer more severe and more frequent bleeding, while others with mild haemophilia typically suffer more minor symptoms except after surgery or serious trauma. Moderate haemophiliacs have variable symptoms which manifest along a spectrum between severe and mild forms.[8]
Prolonged bleeding from a venepuncture or heelprick is another common early sign of haemophilia, these signs may lead to blood tests which indicate haemophilia.[9] In other people, especially those with moderate or mild haemophilia, any trauma will lead to the first serious bleed. Haemophilia leads to a severely increased risk of prolonged bleeding from common injuries, or in severe cases bleeding may be spontaneous and without obvious cause. Bleeding may occur anywhere in the body, superficial bleeding such as those caused by abrasions, or shallow lacerations may be prolonged and the scab may easily be broken up due to the lack of fibrin, which may cause re-bleeding.[8] While superficial bleeding is troublesome, some of the more serious sites of bleeding are:[10]
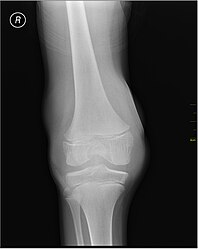
Muscle and joint haemorrhages – or haemarthrosis – are indicative of haemophilia,[11] while digestive tract and cerebral haemorrhages are also germane to other coagulation disorders.
Repeated bleeds into a joint capsule can cause permanent joint damage and disfigurement resulting in chronic arthritis and disability. Joint damage is not a result of blood in the capsule but rather the healing process. When blood in the joint is broken down by enzymes in the body, the bone in that area is also degraded, this exerts a lot of pain upon the person afflicted with the disease.[12][13][14]
Complications
One therapeutic conundrum is the development of inhibitor antibodies against factor VIII due to frequent infusions. These develop as the body recognises the infused factor VIII as foreign, as the body does not produce its own copy. In these individuals, activated factor VII, a precursor to factor VIII in the coagulation cascade, can be infused as a treatment for haemorrhage in individuals with haemophilia and antibodies against replacement factor VIII.[8][15]
Mouth
The oral manifestations are characterized by frequent bleeding of multiple sites, frequently seen as gingival and postextraction haemorrhages. The symptoms depend on the severity of haemophilia. In the case of severe haemophilia, patients may complain of multiple oral bleeding episodes throughout their life. Haemophilia patients are considered to be a special group of patients as routinely done procedures may be fatal in them. It was seen that almost 14% of all haemophilia patients and 30% of cases with a mild type of haemophilia have been diagnosed early following an episode of severe oral bleeding, of which the most common sites were the labial frenum and the tongue.[16]
Genetics
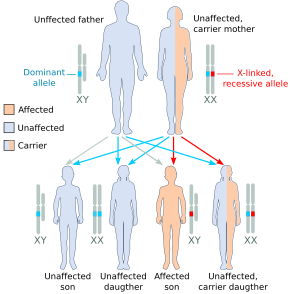
Haemophilia A is inherited as an X-linked recessive trait. It occurs in males and in homozygous females (which is only possible in the daughters of a haemophilic male and a carrier or haemophiliac female[17]). However, mild haemophilia A is known to occur in heterozygous females due to X-inactivation, so it is recommended that levels of factor VIII and IX be measured in all known or potential carriers prior to surgery and in the event of clinically significant bleeding.[8][18]
About 5-10% of people with haemophilia A are affected because they make a dysfunctional version of the factor VIII protein, while the remainder are affected because they produce factor VIII in insufficient amounts (quantitative deficiency).[18] Of those who have severe deficiency (defined as <1% activity of factor VIII), 45-50% have the same mutation, an inversion within the factor VIII gene that results in total elimination of protein production.[18]
Since both forms of haemophilia can be caused by a variety of different mutations, initial diagnosis and classification is done by measurement of protein activity rather than by genetic tests, though genetic tests are recommended for testing of family members once a known case of haemophilia is identified.[8][18]
Approximately 30% of people have no family history; their disease is presumably caused by new mutations.[19]
Diagnosis
The diagnosis of haemophilia A may be suspected as coagulation testing reveals an increased partial thromboplastin time (PTT) in the context of a normal prothrombin time (PT) and bleeding time. PTT tests are the first blood test done when haemophilia is indicated.[20] However, the diagnosis is made in the presence of very low levels of factor VIII. A family history is frequently present, although not essential. Recently, genetic testing has been made available to determine an individual's risk of attaining or passing on haemophilia. Diagnosis of haemophilia A also includes a severity level, which can range from mild to severe based on the amount of active and functioning factor VIII detected in the blood. Factor VIII levels do not typically change throughout an individual's lifetime. Severe haemophilia A is the most common severity, occurring in the majority of affected people. Individuals with mild haemophilia often experience few or no bleeding episodes except in the case of serious trauma (i.e. tooth extraction, or surgery).[8]
-
X-Ray- haemoarthritis
-
Haemarthrosis on lateral view
Severity
There are numerous different mutations which can cause haemophilia A, due to differences in changes to the factor VIII gene (and the resulting protein). Individuals with haemophilia often have some level of active clotting factor. Individuals with less than 1% active factor are classified as having severe haemophilia, those with 1–5% active factor have moderate haemophilia, and those with mild haemophilia have between 5–40% of normal levels of active clotting factor.[21]
Differential diagnosis
Two of the most common differential diagnoses are haemophilia B which is a deficiency in Factor IX and von Willebrand Disease which is a deficiency in von Willebrand factor (needed for the proper functioning of Factor VIII[22]); haemophilia C is also considered.
Treatment

In regards to the treatment of this genetic disorder, most individuals with severe haemophilia require regular supplementation with intravenous recombinant or plasma concentrate Factor VIII. The preventative treatment regime is highly variable and individually determined.[10] In children, an easily accessible intravenous port[23] may have to be inserted to minimise frequent traumatic intravenous cannulation. These devices have made prophylaxis in haemophilia much easier for families because the problems of finding a vein for infusion several times a week are eliminated. However, there are risks involved with their use, the most worrisome being that of infection, studies differ but some show an infection rate that is high.[24] These infections can usually be treated with intravenous antibiotics but sometimes the device must be removed,[25] also, there are other studies that show a risk of clots forming at the tip of the catheter, rendering it useless. Some individuals with severe haemophilia, and most with moderate and mild haemophilia, treat only as needed without a regular prophylactic schedule.[26] Mild haemophiliacs often manage their condition with desmopressin, a drug which releases stored factor VIII from blood vessel walls.[27]
Teeth
The inferior alveolar nerve block should only be given after raising clotting factor levels by appropriate replacement therapy, as there is a risk of bleeding into the muscles along with potential airway compromise due to a haematoma in the retromolar or pterygoid space. The intraligamental technique or interosseous technique should be considered instead of the mandibular block. Articaine has been used as a buccal infiltration to anaesthetize the lower molar teeth. A lingual infiltration also requires appropriate factor replacement since the injection is into an area with a rich plexus of blood vessels and the needle is not adjacent to bone.[28]
Monoclonal antibodies
Monoclonal antibody emicizumab has been approved by the FDA in 2017 for therapy of hemophilia A.[29]
Prognosis
Two Dutch studies have followed haemophilia patients for a number of years.[30][31] Both studies found that viral infections were common in haemophiliacs due to the frequent blood transfusions which put them at risk of acquiring blood borne infections, such as HIV, hepatitis B and hepatitis C. In the latest study which followed people from 1992 to 2001, the male life expectancy was 59 years. If cases with known viral infections were excluded, the life expectancy was 72, close to that of the general population. 26% of the cases died from AIDS and 22% from hepatitis C.[31]
Epidemiology
Haemophilia A occurs in approximately 1 in 5,000 males,[18] while the incidence of haemophilia B is 1 in 30,000 in the male population,[18] of these, 85% have haemophilia A and 15% have haemophilia B.[18]
Research
Gene therapy
In December 2017, it was reported that doctors had used a new form of gene therapy to treat haemophilia A.[32][33][34]
See also
References
- ↑ 1.00 1.01 1.02 1.03 1.04 1.05 1.06 1.07 1.08 1.09 1.10 1.11 1.12 1.13 1.14 1.15 1.16 1.17 1.18 "Hemophilia A - Symptoms, Causes, Treatment | NORD". rarediseases.org. Archived from the original on 2 May 2023. Retrieved 14 September 2023.
- ↑ 2.0 2.1 2.2 2.3 2.4 "Hemophilia A | NBDF". National Hemophilia Foundation. Archived from the original on 5 July 2023. Retrieved 14 September 2023.
- ↑ Salen, Philip; Babiker, Hani M. (2023). "Hemophilia A". StatPearls. StatPearls Publishing. Archived from the original on 16 September 2023. Retrieved 14 September 2023.
- ↑ 4.0 4.1 4.2 4.3 4.4 "Haemophilia A (Factor VIII Deficiency) information | Patient". Patient. Archived from the original on 27 June 2016. Retrieved 24 June 2016.
- ↑ 5.0 5.1 "Estimating hemophilia prevalence and life expectancy | Canadian Blood Services". www.blood.ca. Archived from the original on 5 July 2022. Retrieved 14 September 2023.
- ↑ 6.0 6.1 "Data & Statistics | Hemophilia | NCBDDD | CDC". Centers for Disease Control and Prevention. 1 August 2022. Archived from the original on 12 September 2023. Retrieved 14 September 2023.
- ↑ 7.0 7.1 "History | NBDF". National Hemophilia Foundation. Archived from the original on 7 June 2023. Retrieved 14 September 2023.
- ↑ 8.0 8.1 8.2 8.3 8.4 8.5 Konkle, Barbara A.; Josephson, Neil C.; Nakaya Fletcher, Shelley (1993-01-01). Pagon, Roberta A.; Adam, Margaret P.; Ardinger, Holly H.; Wallace, Stephanie E.; Amemiya, Anne; Bean, Lora J.H.; Bird, Thomas D.; Fong, Chin-To; Mefford, Heather C. (eds.). Hemophilia A. Seattle (WA): University of Washington, Seattle. PMID 20301578. Archived from the original on 2019-10-19. Retrieved 2021-04-25.update 2014
- ↑ Lissauer, Tom; Fanaroff, Avroy A.; Miall, Lawrence; Fanaroff, Jonathan (2015-06-10). Neonatology at a Glance. John Wiley & Sons. p. 135. ISBN 9781118767429. Archived from the original on 2021-04-27. Retrieved 2021-04-25.
- ↑ 10.0 10.1 "How Is Hemophilia Treated? - NHLBI, NIH". www.nhlbi.nih.gov. Archived from the original on 2017-10-05. Retrieved 2016-07-08.
- ↑ "Management of Joint Bleeding in Hemophilia: Management of Acute Hemarthrosis". Medscape. Medscape.org. Archived from the original on 3 August 2016. Retrieved 1 August 2016.
- ↑ Zhou, Jenny Y.; Barnes, Richard F. W.; Foster, Gary; Iorio, Alfonso; Cramer, Thomas J.; von Drygalski, Annette (12 July 2019). "Joint Bleeding Tendencies in Adult Patients With Hemophilia: It's Not All Pharmacokinetics". Clinical and Applied Thrombosis/Hemostasis. 25. doi:10.1177/1076029619862052. ISSN 1076-0296. Archived from the original on 23 September 2021. Retrieved 17 September 2021.
- ↑ "Joint Disease | Introduction to Hemophilia Care | NCBDDD | CDC". www.cdc.gov. 20 May 2020. Archived from the original on 7 April 2021. Retrieved 19 September 2021.
- ↑ Abrol, Pankaj (10 April 2019). Hemophilia: Recent Advances. BoD – Books on Demand. p. 39. ISBN 978-1-78985-817-4. Archived from the original on 23 September 2021. Retrieved 20 September 2021.
- ↑ Ma, Alice D.; Carrizosa, Daniel (2006-01-01). "Acquired Factor VIII Inhibitors: Pathophysiology and Treatment". ASH Education Program Book. 2006 (1): 432–437. doi:10.1182/asheducation-2006.1.432. ISSN 1520-4391. PMID 17124095.
- ↑ Sonu Acharya, Karishma Rathore, Upasana Mahapatra, Sashikant Sethi, Nikita Sahu (2018). "An unusual oral manifestation of hemophilia in a child". Journal of International Oral Health. Retrieved 2019-12-20.
- ↑ Nair, Preethi S.; Shetty, S.; Ghosh, Kanjaksha (2012-01-01). "A homozygous female hemophilia A". Indian Journal of Human Genetics. 18 (1): 134–136. doi:10.4103/0971-6866.96685. ISSN 0971-6866. PMC 3385172. PMID 22754241.
- ↑ 18.0 18.1 18.2 18.3 18.4 18.5 18.6 Kliegman, Robert (2011). Nelson textbook of pediatrics (19th ed.). Philadelphia: Saunders. pp. 1700–1. ISBN 978-1-4377-0755-7.
- ↑ Bowen, D J (2002). "Haemophilia A and haemophilia B: molecular insights". Molecular Pathology. 55 (1): 1–18. doi:10.1136/mp.55.1.1. PMC 1187139. PMID 11836440.
- ↑ "OMIM Entry - # 306700 - HEMOPHILIA A; HEMA". omim.org. Archived from the original on 2016-05-10. Retrieved 2016-07-08.
- ↑ Hemophilia A at eMedicine
- ↑ "Von Willebrand's Disease. About Von Willebrand's Disease | Patient". Patient. Archived from the original on 2016-07-06. Retrieved 2016-07-08.
- ↑ Ma, Alice D.; Roberts, Harold R.; Escobar, Miguel A. (2012-10-03). Hemophilia and Hemostasis: A Case-Based Approach to Management. John Wiley & Sons. ISBN 9781118439302. Archived from the original on 2021-04-27. Retrieved 2021-04-25.Google books no page
- ↑ Santagostino, Elena; Mancuso, Maria Elisa (2008-09-01). "Barriers to primary prophylaxis in haemophilic children: the issue of the venous access". Blood Transfusion. 6 (Suppl 2): s12–s16. doi:10.2450/2008.0031-08. ISSN 1723-2007. PMC 2652218. PMID 19105504.
- ↑ "Guidelines for the Prevention of Intravascular Catheter-Related Infections". www.cdc.gov. Archived from the original on 2 October 2018. Retrieved 8 July 2016.
- ↑ Ljung, Rolf (2007-09-01). "The risk associated with indwelling catheters in children with haemophilia". British Journal of Haematology. 138 (5): 580–586. doi:10.1111/j.1365-2141.2007.06703.x. ISSN 1365-2141. PMID 17686052. S2CID 25748135.
- ↑ Franchini, Massimo; Lippi, Giuseppe (2011-10-01). "The use of desmopressin in acquired haemophilia A: a systematic review". Blood Transfusion. 9 (4): 377–382. doi:10.2450/2011.0113-10. ISSN 1723-2007. PMC 3200405. PMID 21839010.
- ↑ Andrew Brewer, Maria Elvira Correa (May 2006). "Guildelines for Dental Treatment of Patients with Inherited Bleeding Disorders" (PDF). Treatment of Hemophilia. 40: 9 – via World Federation of Hemophilia (WFH).
- ↑ HEMLIBRA- emicizumab injection, solution drug label/data at DailyMed from U.S. National Library of Medicine, National Institutes of Health.
- ↑ Triemstra, Mattanja (1995-12-01). "Mortality in Patients with Hemophilia: Changes in a Dutch Population from 1986 to 1992 and 1973 to 1986". Annals of Internal Medicine. 123 (11): 823–7. doi:10.7326/0003-4819-123-11-199512010-00002. ISSN 0003-4819. PMID 7486463. S2CID 22008880. Archived from the original on 2021-09-23. Retrieved 2021-04-25.
- ↑ 31.0 31.1 Plug, I.; Van Der Bom, J. G.; Peters, M.; Mauser-Bunschoten, E. P.; De Goede-Bolder, A.; Heijnen, L.; Smit, C.; Willemse, J.; Rosendaal, F. R. (2006-03-01). "Mortality and causes of death in patients with hemophilia, 1992–2001: a prospective cohort study1". Journal of Thrombosis and Haemostasis. 4 (3): 510–516. doi:10.1111/j.1538-7836.2006.01808.x. hdl:1887/5021. ISSN 1538-7836. PMID 16460432. S2CID 13651790.
- ↑ "Groundbreaking gene therapy trial set to cure haemophilia A". Barts Health NHS Trust. 14 December 2017. Archived from the original on 15 December 2017. Retrieved 14 December 2017.
- ↑ "Haemophilia A trial results 'mind-blowing'". BBC. 14 December 2017. Archived from the original on 8 March 2019. Retrieved 14 December 2017.
- ↑ Rangarajan, Savita; Walsh, Liron; Lester, Will; Perry, David; Madan, Bella; Laffan, Michael; Yu, Hua; Vettermann, Christian; Pierce, Glenn F.; Wong, Wing Y.; Pasi, K. John (16 December 2017). "AAV5–Factor VIII Gene Transfer in Severe Hemophilia A". New England Journal of Medicine. 377 (26): 2519–2530. doi:10.1056/NEJMoa1708483. hdl:10044/1/57163. PMID 29224506. Archived from the original on 4 August 2020. Retrieved 25 April 2021.
External links
| Classification | |
|---|---|
| External resources |