Megalencephaly
| Megalencephaly | |
|---|---|
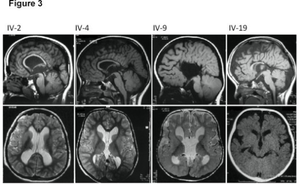 | |
| MRI images showing megalencephaly in four family members who all have unusually large skulls (the family is affected by an autosomal recessive syndrome caused by a KIF7 mutation that induces multiple epiphyseal dysplasia)[1] | |
| Usual onset | Congenital |
| Duration | Long-term |
Megalencephaly (or macrencephaly; abbreviated MEG) is a growth development disorder in which the brain is abnormally large. It is characterized by a brain with an average weight that is 2.5 standard deviations above the mean of the general population.[2] Approximately 1 out of 50 children (2%) are said to have the characteristics of megalencephaly in the general population.[3]
A mutation in the PI3K-AKT pathway is believed to be the primary cause of brain proliferation and ultimately the root cause of megalencephaly. This mutation has produced a classification of brain overdevelopment that consists of two syndromes including megalencephaly-capillary malformation (MCAP) and megalencephaly-polydactyly-polymicrogyria-hydrocephalus (MPPH).[4] Megalencephaly is usually diagnosed at birth and is confirmed with an MRI.
There are several neuropsychiatric disorders linked with megalencephaly; however, studies have shown that autism is the most prevalent association with the malformation of MEG.[5] Although no treatment currently exists for megalencephaly, management methods are focused at reducing deficits linked with autism. Most recent research is targeted at creating inhibitors to reduce the mutational pathway that causes megalencephaly.[6]
Classification
Macrocephaly
Macrocephaly is a term used to refer to a person who has an abnormally large head. The circumference of the head must be above the 95th percentile or at least 2.5 standard deviations from the mean of normal weight and gender groups in the United States.[2][7] A person with macrocephaly does not necessarily indicate that megalencephaly is also present. Large skulls usually exhibit no neurodevelopment conditions at all, meaning most individuals with macrocephaly are healthy.[citation needed]
Hemimegalencephaly
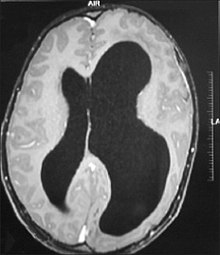
Hemimegalencephaly is an extremely rare form of macrocephaly and is characterized by uneven development of brain hemispheres (one-half of brain is larger than other).[9] The syndrome can be presented by itself or in association with phakomatosis or hemigigantism. Additionally, hemimegalencephaly will frequently cause severe epilepsy, focal neuro-logical deficits, macrocrania, and mild to severe intellectual disability.[10]
MCAP
Megalencephaly-capillary (MCAP) is one of the two major syndromes of megalencephaly. Typically, MCAP and MPPH can be distinguished by somatic features.[11] MCAP includes many characteristics that are observed at birth including: cutaneous vascular malformations, especially capillary malformations of the face and cutis marmorata, polydactyly, connective tissue dysplasia, and focal or segmental body overgrowth.[12] Furthermore, MCAP can occasionally be linked with asymmetric brain overgrowth (hemimegalencephaly) as well as segmental overgrowth of the body (hemihypertrophy).[4]
MPPH
Megalencephaly-polymicrogyria-polydactyly-hydrocephalus (MPPH) is one of the two major syndromes contributing to megalencephaly. Typically MCAP and MPPH can be distinguished by somatic features.[11] In differentiation to MCAP, MPPH lacks consistent somatic features other than postaxial polydactyly.[12] Furthermore, brain and body development is normally symmetric in the majority of patients that appear to have MPPH symptoms.[4]
Signs and symptoms
Autism
There is an extremely high association between megalencephaly and autism. Approximately 20% of autistic children have megalencephaly, making it the most common physical characteristic of autism.[6] People who present both megalencephaly and autistic characteristics usually also show signs of hyperactivity as a major symptom. However, there is no definitive evidence that autism is the primary cause/result of megalencephaly.[citation needed]
Since most children with megalencephaly also have autism, the goal of treatment for MEG is focused on managing the signs and symptoms associated with autism.[citation needed]
Other associations
- Achondroplasia
- Beckwith–Wiedemann syndrome
- Neurofibromatosis type I (NF1)
- Tuberous sclerosis (TS)
- Klippel–Trénaunay syndrome
- Epidermal nevus syndrome
Causes
Although very little is still known as to the direct cause of megalencephaly, recent studies have begun to provide early indications of possible sources for its formation. Recent research has shown that there is a strong link between genetic pathways that cause brain develop and mutations in that pathway that result in brain overgrowth.[citation needed]
PI3K-AKT
Recent studies have shown that mutations in phosphoinositide 3-kinase (PI3K) and AKT (also known as protein kinase B) pathway have been identified in MCAP and MPPH. This pathway has proven to be an integral part of brain growth and development and is an area of interest to many researchers who study the cause of megalencephaly. Mutations in this pathway have been shown to cause a gain of function in the activation of the PI3K-AKT pathway.[13] This cellular pathway is critical in the regulation of diverse cell functions including, cell growth, proliferation, metabolism, survival, apoptosis, angiogenesis, tumorigenesis and most importantly in regards to megalencephaly, brain development.[14] AKT is a crucial signaling molecule part of the PI3K pathway and is also involved in many cellular functions. These functions include, brain development, synaptic plasticity and neurodevelopment. Loss of function in AKT can cause microcephaly in humans while inactivation of the pathway can cause hemimegalencephaly.[4] There are also several cancers that have been shown to be linked with mutations in the AKT pathway, including melanoma and lung cancer.[4][15][16]
Pur-alpha
Pur-alpha (purα) is a sequence-specific single-stranded DNA and RNA-binding protein. Studies have shown that the protein is primarily active during early development and is believed to have a role in brain enlargement.[17] Although the exact function is still controversial, it is believed that pur-alpha is responsible for neuronal proliferation during neurogenesis as well as the maturation of dendrites.[18] Thus, pur-alpha is also considered a potential root cause of megalencephaly and brain overgrowth.[citation needed]
Pathophysiology
One impact of megalencephaly is the complete lack of motor development. One medical study assessed three patients presenting megalencephaly who showed severely impaired motor and speech development as well as distinct facial abnormalities including skull bossing, a low nasal bridge and large eyes.[19]
Diagnosis
Diagnosis of megalencephaly has changed over the years, however, with the development of more advanced equipment, physicians have been able to confirm the disorder with better accuracy. Usually, a physical exam is first performed when characteristics of megalencephaly have appeared. This typically occurs at birth or during early child development. A physician will then take head measurements in order to determine the circumference. This is known as the head circumference.[7] Then a family background will be recorded in order to determine if there has been a history of megalencephaly in the family.[20]
A neurological exam will then be performed using the technology of an MRI machine in order to confirm the diagnosis of megalencephaly. These imaging tests give detailed information regarding brain size, volume asymmetry and other irregular developments linked with MCAP, MPPH and hemimegalencephaly.[2][20]
There is also a strong correlation of epilepsy and megalencephaly and this can aid doctors in their diagnosis.[19]
If a diagnosis of megalencephaly is confirmed, the child is referred to a specialist who focuses on managing the symptoms and improving lifestyle. Since megalencephaly is usually presented with autism, the goal of treatment is to improve deficiencies associated with autistic causes. Additionally, since each patient has unique symptoms, there is no one specific treatment method and therefore is heavily reliant on symptoms associated with an individual.[citation needed]
Prevention
Since there are very few treatment methods focused on managing megalencephaly, future research is targeted at inhibiting mutation of the pathway. However, this next step could be met with several complications as understanding the underlying mechanism of the mutation is a difficult task. The genetic coding that initiates a single mutation is sporadic and patterns are hard to detect in many cases.[6] Even though very little research has been done to create inhibitors of the PI3K-AKT pathway, several pharmaceutical companies have begun to focus their interests in designing a prevention method for this purpose.[6]
Treatment
There is currently no specific treatment for megalencephaly, however periodic head measurements may be assessed to determine the rate of brain growth. Those individuals who develop neurological disorders may be prescribed anti-epileptic drugs for seizures.[2] Studies have shown that reducing epilepsy can increase cell apoptosis and reduce the proliferation of neurons that ultimately leads to brain overgrowth.[21]
Prognosis
The prognosis of megalencephaly depends heavily on the underlying cause and associated neurological disorders.[20] Because the majority of megalencephaly cases are linked with autism, the prognosis is equivalent to the corresponding condition. Since hemimegalencephaly is associated with severe seizures, hemiparesis and intellectual disability, the result is a poor prognosis. In most cases, those diagnosed with this type of megalencephaly usually do not survive through adulthood.[22]
Epidemiology
Approximately one out of every 50 (2%) children in the general population are said to have megalencephaly.[3] Additionally, it is said that megalencephaly affects 3–4 times more males than females. Those individuals that are classified with macrocephaly, or general head overgrowth, are said to have megalencephaly at a rate of 10–30% of the time.[citation needed]
History
It is believed that megalencephaly was discovered in 1972. Prior to diagnoses that used MRI scanning as a way to confirm brain overgrowth, cases of megalencephaly were diagnosed by autopsy in which the physical brain was measured and weighed.[6]
Research
Future research is targeted at further understanding mutations and how they lead to MCAP and MPPH syndromes. The majority of studies of megalencephaly have included mice who present brain abnormalities and overgrowth. The next step is to move to clinical trials involving humans in order to determine the exact genetic mutation causing the sequences. Additionally, scientists and pharmaceutical companies have begun to show interest in mutation inhibition and designing preventative methods to eliminate the underlying cause of megalencephaly altogether.[6]
Other relations
Intracranial volume also affects this pathology, since it is related with the size of the brain.[23]
References
- ↑ Ali BR, Silhavy JL, Akawi NA, Gleeson JG, Al-Gazali L (2012). "A mutation in KIF7 is responsible for the autosomal recessive syndrome of macrocephaly, multiple epiphyseal dysplasia and distinctive facial appearance". Orphanet Journal of Rare Diseases. 7: 27. doi:10.1186/1750-1172-7-27. PMC 3492204. PMID 22587682. Archived from the original on 2022-06-27. Retrieved 2022-10-12.
- ↑ 2.0 2.1 2.2 2.3 Striano, P; Federico (October 2012). "Mutations in mTOR pathway linked to megalencephaly syndromes". Nature Reviews Neurology. 8. 8 (10): 542–4. doi:10.1038/nrneurol.2012.178. PMID 22907262. S2CID 33405738.
- ↑ 3.0 3.1 Sandler, A; Knudsen; Brown; Christian (August 1997). "Neurodevelopmental dysfunction among nonreferred children with idiopathic megalencephaly". Journal of Pediatrics. 131 (2): 320–4. doi:10.1016/S0022-3476(97)70176-8. PMID 9290626.
- ↑ 4.0 4.1 4.2 4.3 4.4 Mirzaa, G; Riviere, Dobybns (May 2013). "Megalencephaly syndromes and activating mutations in the PI3K-AKT pathway: MPPH and MCAP". American Journal of Medical Genetics Part C. 163 (2): 122–30. doi:10.1002/ajmg.c.31361. PMID 23592320. S2CID 35211056.
- ↑ Ghaziuddin, M; Zaccagnini; Tsai; Elardo (August 1999). "Is Megalencephaly specific to autism?" (PDF). Journal of Intellectual Disability Research. 43 (4): 279–82. doi:10.1046/j.1365-2788.1999.00211.x. hdl:2027.42/72452. PMID 10466865. Archived from the original on 2023-02-18. Retrieved 2022-10-12.
- ↑ 6.0 6.1 6.2 6.3 6.4 6.5 Interview with Dr. Ghayda Mirzaa Archived 2018-09-19 at the Wayback Machine (October 1st, 2013)
- ↑ 7.0 7.1 Daymont, C; Zabel M; Feudtner C; Rubin D (January 2012). "The test characteristics of head circumference measurements for pathology associated with head enlargement: a retrospective cohort study". BMC Pediatrics. 12 (9): 9. doi:10.1186/1471-2431-12-9. PMC 3331824. PMID 22269214.
- ↑ Acharya N, Reddy MS, Paulson CT, Prasanna D (January 2014). "Cranio-orbital-temporal neurofibromatosis: an uncommon subtype of neurofibromatosis type-1". Oman Journal of Ophthalmology. 7 (1): 43–5. doi:10.4103/0974-620X.127934. PMC 4008903. PMID 24799805.
- ↑ Johns Hopkins Medicine, Neurology and Neurosurgery. "Hemimegalencephaly". Archived from the original on 2014-04-02. Retrieved 2022-10-12.
- ↑ Di Rocco, C; Battaglia, Pietrini; Piastra, Massimi (August 2006). "Hemimegalencephaly: clinical implications and surgical treatment". Child's Nervous System. 22 (8): 852–866. doi:10.1007/s00381-006-0149-9. PMID 16821075. S2CID 23996880.
- ↑ 11.0 11.1 Mirzaa, Ghayda; Conway; Gripp; Lerman-Sagie; Siegel; deVries; Lev; Kramer; Hopkins; Graham; Dobyns (February 2012). "Megalencephaly-capillary malformation (MCAP) and megalencephaly-polydactyly-polymicrogyria-hydrocephalus (MPPH) syndromes: two closely related disorders of brain overgrowth and abnormal brain and body morphogenesis". American Journal of Medical Genetics. 158A (2): 269–91. doi:10.1002/ajmg.a.34402. PMID 22228622.
- ↑ 12.0 12.1 Nakamura, K; Kato; Tohyama; Shiohama; Hayasaka; Nishiyama; Kodera; Nakashima; Tsurusaki; Miyake; Matsumoto; Saitsu (June 2013). "AKT3 and PIK3R2 mutations in two patients with megalencephaly-related syndromes: MCAP and MPPH". Clinical Genetics. 85 (4): 396–398. doi:10.1111/cge.12188. PMID 23745724. S2CID 12057608.
- ↑ Fam, H (December 2012). "Caught in the AKT: identification of a de novo pathway in MCAP and MPPH and its therapeutic implications". Clinical Genetics. 82 (6): 521–2. doi:10.1111/cge.12003. PMID 22989095. S2CID 27129200.
- ↑ Engelman, J (August 2009). "Targeting PI3K signalling in cancer: opportunities, challenges and limitations". Nature Reviews Cancer. 9 (8): 550–62. doi:10.1038/nrc2664. PMID 19629070. S2CID 7632764.
- ↑ Riviere, J; Mirzaa; O'Roak; Beddaui; Alcantara; Conway (June 2012). "De novo germline and postzygotic mutations in AKT3, PIK3R2 and PIK3CA cause a spectrum of related megalencephaly syndromes". Nat Genet. 44 (8): 934–40. doi:10.1038/ng.2331. PMC 3408813. PMID 22729224.
- ↑ Stahl, J; Sharma; Cheung; Zimmerman; Cheng; Bosenberg; Kester; Sandirasegarane; Robertson (October 2004). "Deregulated Akt3 activity promotes development of malignant melanoma". Cancer Research. 64 (19): 7002–10. doi:10.1158/0008-5472.CAN-04-1399. PMID 15466193.
- ↑ Hokkanen, S; Feldmann; Ding; Jung; Bojarski; Renner-Muller; Schuller; Kretzschmar; Wolf; Herms (February 2012). "Lack of Pur-alpha alters postnatal brain development and causes megalencephaly". Human Molecular Genetics. 21 (3): 473–84. doi:10.1093/hmg/ddr476. PMID 22010047.
- ↑ Khalili, K; Del Valle; Muralidharan; Gault; Darbinian; Otte; Meier; Johnson; Daniel; Kinoshita; Amini; Gordon (October 2003). "Puralpha is essential for postnatal brain development and developmentally coupled cellular proliferation as revealed by genetic inactivation in the mouse". Molecular Cell Biology. 23 (19): 6857–75. doi:10.1128/MCB.23.19.6857-6875.2003. PMC 193944. PMID 12972605.
- ↑ 19.0 19.1 Hengst, M; Tucke, Zerres; Blaum, Hausler (September 2010). "Megalencephaly, mega corpus callosum, and complete lack of motor development. But many with megalancephaly have normal intelligence.: Delineation of a rare syndrome". American Journal of Medical Genetics Part A. 152A (9): 2360–4. doi:10.1002/ajmg.a.33577. PMID 20803648. S2CID 205313167.
- ↑ 20.0 20.1 20.2 National Institute of Neurological Disorders and Stroke. "Megalencephaly Information Page". National Institute of Health. Archived from the original on January 3, 2019. Retrieved January 2, 2019.
- ↑ Almgren, M; Schalling, Lavebratt (November 2008). "Idiopathic megalencephaly-possible cause and treatment opportunities: from patient to lab". European Journal of Pediatrics. 12 (6): 438–45. doi:10.1016/j.ejpn.2007.11.008. PMID 18242108.
- ↑ Nakahashi, M; Sato, Yagishita; Ota, Saito; Sugai, Sasaki; Natsume, Tsushima; Amanuma, Endo (December 2009). "Clinical and imaging characteristics of localized megalencephaly: a retrospective comparison of diffuse hemimegalencephaly and multilobar cortical dysplasia". Neuroradiology. 51. 51 (12): 821–30. doi:10.1007/s00234-009-0579-7. PMID 19672585. S2CID 28924230.
- ↑ Adams, Hieab H H; Hibar, Derrek P; Chouraki, Vincent; Stein, Jason L; Nyquist, Paul A; Rentería, Miguel E; Trompet, Stella; Arias-Vasquez, Alejandro; Seshadri, Sudha (2016). "Novel genetic loci underlying human intracranial volume identified through genome-wide association". Nature Neuroscience. 19 (12): 1569–1582. doi:10.1038/nn.4398. PMC 5227112. PMID 27694991.
External links
- Megalencephaly information page Archived 2022-04-12 at the Wayback Machine (provided by National Institute of Neurological Disorders and Stroke)
| Classification | |
|---|---|
| External resources |