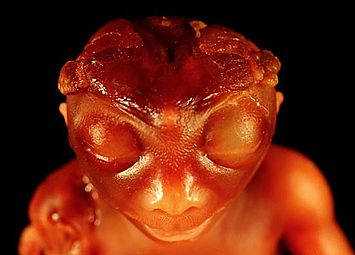
Anencephaly
| Anencephaly | |
|---|---|
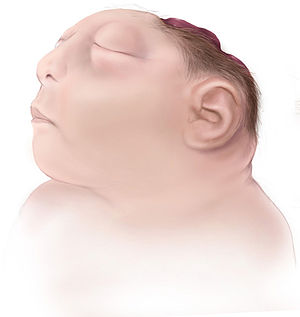 | |
| Illustration of an anencephalic fetus | |
| Specialty | Medical genetics; pediatrics |
| Symptoms | Blind, deaf, unconscious, unable to feel pain[1] |
| Usual onset | Embryonic development[2] |
| Risk factors | Folate deficiency, antiepileptic medication, diabetes, excessive vitamin A, hyperthermia during pregnancy[2] |
| Diagnostic method | Before birth: Suspected based on high levels of alpha fetoprotein in the mother, confirmed by ultrasound[2] After birth: Examination[2] |
| Prevention | Folic acid supplementation[2] |
| Treatment | Supportive care[1] |
| Prognosis | Extremely poor[1] |
| Frequency | 1 in 7,500 births (USA)[3] |
Anencephaly is the absence of most of the brain and skull as a result of issues during early development.[2] Babies with the condition are blind, deaf, unconscious, and unable to feel pain.[1] Reflexes such as breathing and responses to sound or touch may be present.[1]
It is believed to result from a number of factors including folate deficiency, antiepileptic medication, diabetes, excessive vitamin A, and hyperthermia during pregnancy.[2] It is a type of neural tube defect.[2] The underlying mechanism involved failure of closure of the head end of the neural tube.[1] Diagnosis before birth may be suspected based on high levels of alpha fetoprotein in the mother and confirmed by ultrasound.[2]
Prevention is by supplementation of folic acid.[2] The anti epileptic medication, valproate should be avoided during pregnancy, if possible.[2] Treatment involves supportive care.[1] If the baby is born alive, most die within a few hours or days of birth.[1] About 1 in 7,500 births are affected in the United States.[3]
Signs and symptoms
A baby born with anencephaly is usually blind, deaf, unaware of its surroundings and unable to feel pain. Although some individuals with anencephaly may be born with a main brain stem, the lack of a functioning cerebrum permanently rules out the possibility of ever gaining awareness of their surroundings. Reflex actions such as breathing and responses to sound or touch may occur.[4]
Due to the presence of the brainstem, children with anencephaly have almost all the primitive reflexes of a newborn, responding to auditory, vestibular and painful stimuli.[5] This means that the child can move, smile, suckle and breathe without the aid of devices.[6]
Causes
The cause of anencephaly is disputed by medical professionals and researchers.
Folic acid has been shown to be important in neural tube formation since at least 1991,[7][8] and as a subtype of neural tube defect, folic acid may play a role in anencephaly. Studies have shown that the addition of folic acid to the diet of women of child-bearing age may significantly reduce, although not eliminate, the incidence of neural tube defects. Therefore, it is recommended that all women of child-bearing age consume 0.4 mg of folic acid daily,[4] especially those attempting to conceive or who may possibly conceive, as this can reduce the risk to 0.03%.[9] It is not advisable to wait until pregnancy has begun, since, by the time a woman knows she is pregnant, the critical time for the formation of a neural tube defect has usually already passed. A physician may prescribe even higher dosages of folic acid (5 mg/day) for women having had a previous pregnancy with a neural tube defect.[9]
In general, neural tube defects do not follow direct patterns of heredity, though there is some indirect evidence of inheritance,[10] and recent animal models indicate a possible association with deficiencies of the transcription factor TEAD2.[11] Studies show that a woman who has had one child with a neural tube defect such as anencephaly has about a 3% risk of having another child with a neural tube defect,[12] as opposed to the background rate of 0.1% occurrence in the population at large.[13] Genetic counseling is usually offered to women at a higher risk of having a child with a neural tube defect to discuss available testing. It is known that people taking certain anticonvulsants and people with insulin-dependent diabetes have a higher risk of having a child with a neural tube defect.[14]
Relation to genetic ciliopathy
Until recently, medical literature did not indicate a connection among many genetic disorders, both genetic syndromes and genetic diseases, that are now being found to be related. As a result of new genetic research, some of these are, in fact, highly related in their root cause despite the widely varying set of medical symptoms that are clinically visible in the disorders. Anencephaly is one such disease, part of an emerging class of diseases called ciliopathies. The underlying cause may be a dysfunctional molecular mechanism in the primary cilia structures of the cell, organelles present in many cellular types throughout the human body. The cilia defects adversely affect "numerous critical developmental signaling pathways" essential to cellular development and, thus, offer a plausible hypothesis for the often multi-symptom nature of a large set of syndromes and diseases. Known ciliopathies include primary ciliary dyskinesia, Bardet–Biedl syndrome, polycystic kidney and liver disease, nephronophthisis, Alström syndrome, Meckel–Gruber syndrome, and some forms of retinal degeneration.[15]
Diagnosis
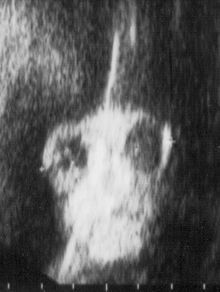
Anencephaly can often be diagnosed before birth through an ultrasound examination. The maternal serum alpha-fetoprotein (AFP screening)[16] and detailed fetal ultrasound[17] can be useful for screening for neural tube defects such as spina bifida or anencephaly.
Meroanencephaly
Meroanencephaly is a rare form of anencephaly characterized by malformed cranial bones, a median cranial defect, and a cranial protrusion called area cerebrovasculosa. Area cerebrovasculosa is a section of abnormal, spongy, vascular tissue admixed with glial tissue ranging from simply a membrane to a large mass of connective tissue, hemorrhagic vascular channels, glial nodules, and disorganized choroid plexuses.[18]
Holoanencephaly
The most common type of anencephaly, where the brain has entirely failed to form, except for the brain stem. Infants rarely survive more than one day after birth with holoanencephaly.[18]
Craniorachischisis
The most severe type of anencephaly where area cerebrovasculosa and area medullovasculosa fill both cranial defects and the spinal column. Craniorachischisis is characterized by anencephaly accompanied by bony defects in the spine and the exposure of neural tissue as the vault of the skull fails to form.[18][19] Craniorachischisis occurs in about 1 of every 1000 live births, but various physical and chemical tests can detect neural tube closure during early pregnancy.[20]
Prognosis
There is no cure or standard treatment for anencephaly. Prognosis is extremely poor, as many anencephalic fetuses do not survive birth and infants that are not stillborn will usually die within a few hours or days after birth from cardiorespiratory arrest.[4]
In almost all cases, anencephalic infants are not aggressively resuscitated because there is no chance of the infants ever achieving a conscious existence. Instead, the usual clinical practice is to offer hydration, nutrition, and comfort measures and to "let nature take its course". Artificial ventilation, surgery (to fix any co-existing congenital defects), and drug therapy (such as antibiotics) are usually regarded as futile efforts. Some clinicians and medical ethicists view even the provision of nutrition and hydration as medically futile.[citation needed]
Epidemiology
In the United States, anencephaly occurs in about 1 out of every 10,000 births.[21] Rates may be higher among Africans with rates in Nigeria estimated at 3 per 10,000 in 1990 while rates in Ghana estimated at 8 per 10,000 in 1992.[22] Rates in China are estimated at 5 per 10,000.[22]
Research has suggested that, overall, female babies are more likely to be affected by the disorder.[23]
Ethical issues
Organ donation
One issue concerning anencephalic newborns is organ donation. Initial legal guidance came from the case of Baby Theresa in 1992, in which the boundaries of organ donation were tested for the first time.[24] Infant organs are scarce, and the high demand for pediatric organ transplants poses a major public health issue. In 1999, it was found that among children who die under the age of two, 30–50% do so while awaiting transplant.[24] In order for infant organs to be viable and used for transplant, they must be removed while the infant still has circulation or very soon after circulation has ceased. However, this poses both legal and ethical issues. In the United States, all states have laws that forbid the removal of organs from a donor until after the donor has died, except in the case of altruistic living donations of a kidney or liver segment.
The goal of those in support of organ donation is that it will further contribute to the pool of suitable organs for transplants while the need for lifesaving organ transplants is so great. It has been estimated that 400–500 infant hearts and kidneys and 500–1,000 infant livers are needed in the United States each year.[25] Because anencephalic neonates have partially functioning brain stems, they possess some brain stem reflexes such as spontaneous respirations. For this reason, these patients cannot be declared brain dead.
Within the medical community, the main ethical issues with organ donation are a misdiagnosis of anencephaly, the slippery slope argument, that anencephalic neonates would rarely be a source of organs, and that it would undermine confidence in organ transplantation.[26] Slippery slope concerns are a major issue in personhood debates, across the board. In regards to anencephaly, those who oppose organ donation argue that it could open the door for involuntary organ donors such as an elderly person with severe dementia. Another point of contention is the number of children who would actually benefit. There are discrepancies in statistics; however, it is known that most anencephalic children are stillborn.[26]
Proposals have been made to bypass the legal and ethical issues surrounding organ donation. These include waiting for death to occur before procuring organs, expanding the definition of death, creating a special legal category for anencephalic infants, and defining them as non-persons.[27]
In the United Kingdom, a child born with anencephaly was reported as the country's youngest organ donor. Teddy Houlston was diagnosed as anencephalic at 12 weeks of gestation. His parents, Jess Evans and Mike Houlston, decided against abortion and instead proposed organ donation. Teddy was born on 22 April 2014, in Cardiff, Wales, and lived for 100 minutes, after which his heart and kidneys were removed. His kidneys were later transplanted into an adult in Leeds. Teddy's twin, Noah, was born healthy.[28]
Brain death
There are four different concepts used to determine brain death: failure of heart, failure of lungs, whole brain death, and neocortical death.
Neocortical death, similar to a persistent vegetative state (PVS), involves loss of cognitive functioning of the brain. A proposal by law professor David Randolph Smith,[29] in an attempt to prove that neocortical death should legally be treated the same as brain death, involved PET scans to determine the similarities. However, this proposal has been criticized on the basis that confirming neocortical death by PET scan may risk indeterminacy.[30]
Pregnancy termination
Anencephaly can be diagnosed before delivery with a high degree of accuracy. Although anencephaly is a fatal condition, the option of abortion is dependent on the abortion laws in the state.[25] According to a 2013 report, 26% of the world's population reside in a country where abortion is generally prohibited.[25] [31] In 2012, Brazil extended the right of abortion to mothers with anencephalic fetuses. This decision is, however, receiving much disapproval by several religious groups.[32]
Legal proceedings
The case of baby Theresa was the beginning of the ethical debate over anencephalic infant organ donation.[24] The story of baby Theresa remains a focus of basic moral philosophy. Baby Theresa was born with anencephaly in 1992. Her parents, knowing that their child was going to die, requested that her organs be given for transplantation. Although her physicians agreed, Florida law prohibited the infant's organs from being removed while she was still alive. By the time she died nine days after birth, her organs had deteriorated past the point of being viable.[33]
Baby K was an anencephalic infant that was kept alive for two years and 174 days under intensive care by order of the US Court of Appeals for the Fourth Circuit.
United States uniform acts
The Uniform Determination of Death Act (UDDA) is a model bill, adopted by many US states, stating that an individual who has sustained either 1) irreversible cessation of circulatory and respiratory functions or 2) irreversible cessation of all functions of the entire brain, including the brain stem, is dead. This bill was a result of much debate over the definition of death and is applicable to the debate over anencephaly. A related bill, the Uniform Anatomical Gift Act (UAGA), grants individuals and, after death, their family members the right to decide whether or not to donate organs. Because it is against the law for any person to pay money for an organ, the person in need of an organ transplant must rely on a volunteer.[25]
There have been two state bills that proposed to change current laws regarding death and organ donation. California Senate Bill 2018 proposed to amend the UDDA to define anencephalic infants as already dead, while New Jersey Assembly Bill 3367 proposed to allow anencephalic infants to be organ sources even if they are not dead.[25][34]
Research
Some genetic research has been conducted to determine the causes of anencephaly. It has been found that cartilage homeoprotein (CART1) is selectively expressed in chondrocytes (cartilage cells). The CART1 gene to chromosome 12q21.3–q22 has been mapped. Also, it has been found that mice homozygous for deficiency in the Cart1 gene manifested acrania and meroanencephaly, and prenatal treatment with folic acid will suppress acrania and meroanencephaly in the Cart1-deficient mutants.[35][36]
See also
References
- ↑ 1.0 1.1 1.2 1.3 1.4 1.5 1.6 1.7 "Anencephaly Information Page | National Institute of Neurological Disorders and Stroke". www.ninds.nih.gov. Archived from the original on 9 January 2021. Retrieved 10 January 2021.
- ↑ 2.00 2.01 2.02 2.03 2.04 2.05 2.06 2.07 2.08 2.09 2.10 Tafuri, SM; Lui, F (January 2020). "Embryology, Anencephaly". PMID 31424828.
{{cite journal}}: Cite journal requires|journal=(help) - ↑ 3.0 3.1 "Anencephaly". NORD (National Organization for Rare Disorders). Archived from the original on 23 November 2020. Retrieved 10 January 2021.
- ↑ 4.0 4.1 4.2 "Anencephaly Information Page: National Institute of Neurological Disorders and Stroke (NINDS)". Archived from the original on 2 December 2016. Retrieved 2016-12-02.
- ↑ DIAMENT, Aron (1996). Neurologia Infantil. 3ª Edição. São Paulo: Atheneu. p. 745.
- ↑ Luyendijk, W; Treffers, PD (1992). "The smile in anencephalic infants". Clinical Neurology and Neurosurgery. 94 Suppl: S113-7. doi:10.1016/0303-8467(92)90042-2. PMID 1320482.
- ↑ Centers for Disease Control (CDC) (1991-08-02). "Use of folic acid for prevention of spina bifida and other neural tube defects--1983-1991". MMWR. Morbidity and Mortality Weekly Report. 40 (30): 513–516. ISSN 0149-2195. PMID 2072886.
- ↑ "Lancet 1991; 338:8760:131-137". www.sjsu.edu. Archived from the original on 2018-04-08. Retrieved 2018-12-10.
- ↑ 9.0 9.1 Ratcliffe, Stephen D., ed. (2008). Family medicine obstetrics (3rd ed.). Philadelphia: Mosby Elsevier. ISBN 978-0-323-04306-9.[page needed]
- ↑ Shaffer, Lisa G.; Marazita, Mary L.; Bodurtha, Joann; Newlin, Anna; Nance, Walter E. (1990). "Evidence for a major gene in familial anencephaly". American Journal of Medical Genetics. 36 (1): 97–101. doi:10.1002/ajmg.1320360119. PMID 2333913.
- ↑ Kaneko, Kotaro J.; Kohn, Matthew J.; Liu, Chengyu; Depamphilis, Melvin L. (2007). "Transcription factor TEAD2 is involved in neural tube closure". Genesis. 45 (9): 577–87. doi:10.1002/dvg.20330. PMC 2765819. PMID 17868131.
- ↑ Cowchock, S.; Ainbender, E.; Prescott, G.; Crandall, B.; Lau, L.; Heller, R.; Muir, W. A.; Kloza, E.; et al. (1980). "The recurrence risk for neural tube defects in the United States: A collaborative study". American Journal of Medical Genetics. 5 (3): 309–14. doi:10.1002/ajmg.1320050314. PMID 7405962.
- ↑ "Archived copy". Archived from the original on 2011-03-04. Retrieved 2011-04-01.
{{cite web}}: CS1 maint: archived copy as title (link)[full citation needed] - ↑ "Anencephaly". Genetics Home Reference. National Institutes of Health. 22 August 2011. Archived from the original on 2011-08-05. Retrieved 28 August 2011.
- ↑ Badano, Jose L.; Mitsuma, Norimasa; Beales, Phil L.; Katsanis, Nicholas (2006). "The Ciliopathies: An Emerging Class of Human Genetic Disorders". Annual Review of Genomics and Human Genetics. 7: 125–48. doi:10.1146/annurev.genom.7.080505.115610. PMID 16722803.
- ↑ Joó JG, Beke A, Papp C, et al. (2007). "Neural tube defects in the sample of genetic counselling". Prenat. Diagn. 27 (10): 912–21. doi:10.1002/pd.1801. PMID 17602445.
- ↑ Cedergren M, Selbing A (2006). "Detection of fetal structural abnormalities by an 11-14-week ultrasound dating scan in an unselected Swedish population". Acta Obstetricia et Gynecologica Scandinavica. 85 (8): 912–5. doi:10.1080/00016340500448438. PMID 16862467.
- ↑ 18.0 18.1 18.2 Isada, Nelson B.; Qureshi, Faisal; Jacques, Suzanne M.; Holzgreve, Wolfgang; Tout, Mary Jo; Johnson, Mark P.; Evans, Mark I. (1993). "Meroanencephaly: Pathology and Prenatal Diagnosis". Fetal Diagnosis and Therapy. 8 (6): 423–8. doi:10.1159/000263862. PMID 8286034.
- ↑ Murlimanju, N; Vishal, K; Maligi, AM; Naveen, NS (2010). "Craniorachischisis totalis". Journal of Neurosciences in Rural Practice. 1 (1): 54–5. doi:10.4103/0976-3147.63108. PMC 3137839. PMID 21799625.
- ↑ Gilbert, Scott F. (2010). Developmental biology (9th ed.). Sunderland, Mass.: Sinauer Associates. p. 338. ISBN 978-0878933846.
- ↑ "Anencephaly". PubMed Health. May 1, 2011. Archived from the original on 2014-03-28. Retrieved 2017-10-31.
- ↑ 22.0 22.1 Neural Tube Defects : From Origin to Treatment: From Origin to Treatment. Oxford University Press. 2005. pp. 125–126. ISBN 9780199775149. Archived from the original on 2016-01-31. Retrieved 2015-08-09.
- ↑ Timson, J. (1970). "The sex ratio in anencephaly". Genetica. 41 (3): 457–65. doi:10.1007/BF00958926. PMID 4922971.
- ↑ 24.0 24.1 24.2 Bard, Jennifer S. (1999). "The diagnosis is anencephaly and the parents ask about organ donation: Now what? A guide for hospital counsel and ethics committees". Western New England Law Review. 21 (1): 49–95. PMID 12774804. Archived from the original on 2014-03-26. Retrieved 2012-07-20.
- ↑ 25.0 25.1 25.2 25.3 25.4 Meinke, Sue (June 1989). "Anencephalic Infants as Potential Organ Sources: Ethical and Legal Issues" (PDF). National Reference Center for Bioethics Literature. Archived (PDF) from the original on 2012-05-22. Retrieved 19 July 2012.
{{cite journal}}: Cite journal requires|journal=(help) - ↑ 26.0 26.1 Glasson, John; Plows, Charles W.; Clarke, Oscar W.; Cosgriff, James H.; Kliger, Craig H.; Ruff, Victoria N.; Tenery, Robert M.; Wilkins, George T.; et al. (1995). "The Use of Anencephalic Neonates as Organ Donors". JAMA. 273 (20): 1614–8. doi:10.1001/jama.1995.03520440068039. PMID 7745776.
- ↑ Ahmad, Ferhaan (1992). "Anencephalic infants as organ donors: Beware the slippery slope". CMAJ. 146 (2): 236–241, 244. PMC 1488368. PMID 1735049.
- ↑ [1] Archived 2018-05-28 at the Wayback Machine. BBC News. Retrieved on 2015-04-23.
- ↑ "Archived copy" (PDF). Archived (PDF) from the original on 2013-10-15. Retrieved 2013-10-15.
{{cite web}}: CS1 maint: archived copy as title (link) - ↑ Beresford, H. Richard (July 1996). "Ethics in Neuroscience: Mind over Matter" (PDF). Indiana University. Archived from the original (PDF) on November 1, 2005.
- ↑ "The World's Abortion Laws Map 2013 Update" (PDF). Archived (PDF) from the original on 2015-12-02. Retrieved 2015-11-30.
- ↑ "Anencephaly in Brazil: A New Decision, Old Issues". Archived from the original on 13 May 2012. Retrieved 19 July 2012.
- ↑ Rachels, James. "What is Morality" (PDF). Archived (PDF) from the original on 2014-12-14. Retrieved 19 July 2012.
- ↑ "Uniform Declaration of Death Act (UDDA)". Archived from the original on 2013-09-06. Retrieved 2013-08-23.
- ↑ Chen, Chih-Ping (2008). "Syndromes, Disorders and Maternal Risk Factors Associated with Neural Tube Defects (III)". Taiwanese Journal of Obstetrics and Gynecology. 47 (2): 131–40. doi:10.1016/S1028-4559(08)60070-4. PMID 18603496.
- ↑ Zhao, Qi; Behringer, Richard R.; De Crombrugghe, Benoit (1996). "Prenatal folic acid treatment suppresses acrania and meroanencephaly in mice mutant for the Cart1 homeobox gene". Nature Genetics. 13 (3): 275–83. doi:10.1038/ng0796-275. PMID 8673125.
External links
| Classification | |
|---|---|
| External resources |
- Pages with script errors
- CS1 errors: missing periodical
- CS1: long volume value
- Wikipedia articles needing page number citations from October 2012
- Articles with invalid date parameter in template
- CS1 maint: archived copy as title
- All articles with incomplete citations
- Articles with incomplete citations from November 2012
- Webarchive template wayback links
- Pages using multiple image with manual scaled images
- Articles with hatnote templates targeting a nonexistent page
- All articles with unsourced statements
- Articles with unsourced statements from November 2011
- Congenital disorders of nervous system
- Brain disorders
- RTT