Monckeberg's arteriosclerosis
| Mönckeberg's sclerosis | |
|---|---|
| Other names: Medial calcific sclerosis or Mönckeberg medial sclerosis | |
 | |
| Microphotography of arterial wall with calcified (violet colour) atherosclerotic plaque (haematoxylin & eosin stain) | |
Mönckeberg's arteriosclerosis, or Mönckeberg's sclerosis, is a form of arteriosclerosis or vessel hardening, where calcium deposits are found in the muscular middle layer of the walls of arteries (the tunica media).[1] It is an example of dystrophic calcification. This condition occurs as an age-related degenerative process. However, it can occur in pseudoxanthoma elasticum and idiopathic arterial calcification of infancy as a pathological condition, as well. Its clinical significance and cause are not well understood and its relationship to atherosclerosis and other forms of vascular calcification are the subject of disagreement.[2] Mönckeberg's arteriosclerosis is named after Johann Georg Mönckeberg,[3][4] who first described it in 1903.
Signs and symptoms
Typically, Mönckeberg's arteriosclerosis is not associated with symptoms unless complicated by atherosclerosis, calciphylaxis, or accompanied by some other disease.[5] However, the presence of Mönckeberg's arteriosclerosis is associated with poorer prognosis.[6] This is probably due to vascular calcification causing increased arterial stiffness, increased pulse pressure and resulting in exaggerated damage to the heart and kidneys.[7]
Cause
Minor degrees of calcification of the cardiovascular system are common in elderly people,[8] and the prevalence of vascular calcification is increased by some diseases (see Epidemiology section). Vascular calcification results from the deposition of calcium phosphate crystals (hydroxyapatite) as a consequence of disordered calcium phosphate regulation in the blood vessel. Hydroxyapatite is secreted in vesicles that bleb out from vascular smooth muscle cells or pericytes in the arterial wall.[2] The mechanism of vascular calcification is not fully understood, but probably involves a phenotypic change in the vascular smooth muscle cells in the wall with activation of bone-forming programs. Numerous regulators of calcification such as osteopontin, osteoprotegerin, matrix gla protein and fetuin-A, receptor activator of NF-kappa-B, receptor activator of NF-kappa-B ligand and tumor necrosis factor (TNF)-related apoptosis-inducing ligand protein have been implicated in this process.[9][10]
It is unclear whether Mönckeberg's arteriosclerosis is a distinct entity or forms part of a spectrum of vascular calcification that includes atherosclerosis and calcification in the inner layer of the artery wall (tunica intima), calcification of the internal elastic lamina, calcification of cardiac valves and widespread soft tissue calcification.[11] The existence of Mönckeberg's arteriosclerosis has been disputed and it has been proposed that it is a part of a continuum of atherosclerotic disease:[9] the majority of atherosclerotic plaques contain some calcium deposits[12][13] and calcification of the internal elastic lamina is common in pathological specimens labelled as Mönckeberg's arteriosclerosis.[2] However studies in animals suggest that a predominantly medial pattern of vascular calcification reflects different underlying mechanisms of disease,[8] and despite involvement of the internal elastic lamina, evidence of inflammation is rare in Mönckeberg's arteriosclerosis.[2]
Diagnosis
Often Mönckeberg's arteriosclerosis is discovered as an incidental finding in an X-ray radiograph, on mammograms, in autopsy, or in association with investigation of some other disease, such as diabetes mellitus or chronic kidney disease. Typically calcification is observed in the arteries of the upper and lower limb although it has been seen in numerous other medium size arteries.[5] In the radial or ulnar arteries it can cause "pipestem" arteries, which present as a bounding pulse at the end of the calcific zone. It may also result in "pulselessness." Epidemiological studies have used the ratio of ankle to brachial blood pressure (ankle brachial pressure index, ABPI or ABI) as an indicator of arterial calcification with ABPI >1.3 to >1.5 being used as a diagnostic criterion depending on the study.[14][15]
-
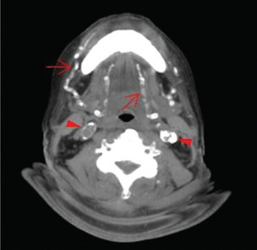
Axial CT slice at the level of C2-C3 soft tissue window setting setting. Extensive calcifications of the bilateral lingual and facial arteries are noted (arrows) as well as calcific plaques at bilateral carotid tree area (arrow heads).
-
A. Pelvic and lower extremity radiograph shows extensive calcification of the femoral arteries. B. Translumbar aortography shows near-total obstruction of the femoral arteries.
Epidemiology
The prevalence of Mönckeberg's arteriosclerosis increases with age and is more frequent in diabetes mellitus, chronic kidney disease, systemic lupus erythematosus, chronic inflammatory conditions, hypervitaminosis D and rare genetic disorders, such as Keutel syndrome.[2] The prevalence of Mönckeberg's arteriosclerosis in the general population has been estimated as <1% on the basis of an ankle brachial pressure index >1.5;[15][16][17] however the validity of this criterion is questionable.[17]
References
- ↑ "Mönckeberg arteriosclerosis" at Dorland's Medical Dictionary
- ↑ 2.0 2.1 2.2 2.3 2.4 Micheletti RG, Fishbein GA, Currier JS, Singer EJ, Fishbein MC (August 2008). "Calcification of the internal elastic lamina of coronary arteries". Mod. Pathol. 21 (8): 1019–28. doi:10.1038/modpathol.2008.89. PMID 18536656.
- ↑ synd/4064 at Who Named It?
- ↑ J. G. Mönckeberg. Über die reine Mediaverkalkung der Extremitätenarterien und ihr Verhalten zur Arteriosklerose. Virchows Archiv für pathologische Anatomie und Physiologie, und für klinische Medicin, Berlin, 1903, 171: 141-167.
- ↑ 5.0 5.1 Steinmann, Beat U.; Royce, Peter M. (2002). Connective tissue and its heritable disorders: molecular, genetic, and medical aspects. New York: Wiley-Liss. ISBN 978-0-471-25185-9.
- ↑ Ono K, Tsuchida A, Kawai H, et al. (June 2003). "Ankle-brachial blood pressure index predicts all-cause and cardiovascular mortality in hemodialysis patients". J. Am. Soc. Nephrol. 14 (6): 1591–8. doi:10.1097/01.asn.0000065547.98258.3d. PMID 12761260.
- ↑ Dao HH, Essalihi R, Bouvet C, Moreau P (May 2005). "Evolution and modulation of age-related medial elastocalcinosis: impact on large artery stiffness and isolated systolic hypertension". Cardiovasc. Res. 66 (2): 307–17. doi:10.1016/j.cardiores.2005.01.012. PMID 15820199.
- ↑ 8.0 8.1 Amann K (November 2008). "Media calcification and intima calcification are distinct entities in chronic kidney disease". Clin J Am Soc Nephrol. 3 (6): 1599–605. doi:10.2215/CJN.02120508. PMID 18815240.
- ↑ 9.0 9.1 McCullough PA, Agrawal V, Danielewicz E, Abela GS (November 2008). "Accelerated atherosclerotic calcification and Mönckeberg's sclerosis: a continuum of advanced vascular pathology in chronic kidney disease". Clin J Am Soc Nephrol. 3 (6): 1585–98. doi:10.2215/CJN.01930408. PMID 18667741.
- ↑ Liu Y, Shanahan CM (2011). "Signalling pathways and vascular calcification". Front. Biosci. 16: 1302–14. doi:10.2741/3790. PMID 21196233.
- ↑ Couri CE, da Silva GA, Martinez JA, Pereira Fde A, de Paula FJ (2005). "Mönckeberg's sclerosis - is the artery the only target of calcification?". BMC Cardiovasc Disord. 5: 34. doi:10.1186/1471-2261-5-34. PMC 1326214. PMID 16343348.
- ↑ Tanimura A, McGregor DH, Anderson HC (1986). "Calcification in atherosclerosis. I. Human studies". J. Exp. Pathol. 2 (4): 261–73. PMID 2946818.
- ↑ Tanimura A, McGregor DH, Anderson HC (1986). "Calcification in atherosclerosis. II. Animal studies". J. Exp. Pathol. 2 (4): 275–97. PMID 3783282.
- ↑ Hirschl M, Francesconi M, Hirschl MM (1991). "[Moenckeberg media sclerosis: clinical aspects in diabetic patients]". VASA (in Deutsch). 20 (3): 216–21. PMID 1950137.
- ↑ 15.0 15.1 Newman AB, Siscovick DS, Manolio TA, et al. (September 1993). "Ankle-arm index as a marker of atherosclerosis in the Cardiovascular Health Study. Cardiovascular Heart Study (CHS) Collaborative Research Group". Circulation. 88 (3): 837–45. doi:10.1161/01.cir.88.3.837. PMID 8353913.
- ↑ Meijer WT, Hoes AW, Rutgers D, Bots ML, Hofman A, Grobbee DE (February 1998). "Peripheral arterial disease in the elderly: The Rotterdam Study". Arterioscler. Thromb. Vasc. Biol. 18 (2): 185–92. doi:10.1161/01.ATV.18.2.185. PMID 9484982.
- ↑ 17.0 17.1 Kröger K, Stang A, Kondratieva J, et al. (2006). "Prevalence of peripheral arterial disease - results of the Heinz Nixdorf recall study". Eur. J. Epidemiol. 21 (4): 279–85. doi:10.1007/s10654-006-0015-9. PMID 16685578.
External links
| Classification |
|---|