Folinic acid
 | |
 | |
| Names | |
|---|---|
| Pronunciation | Leucovorin /ˌljuːkoʊˈvɔːrɪn/ |
| Trade names | Many |
| Other names | Calcium folinate, citrovorum factor, 5-formyltetrahydrofolate |
| |
| Clinical data | |
| Drug class | Folic acid analogue |
| Main uses | To decrease toxicity of methotrexate or pyrimethamine[1] |
| Side effects | Trouble sleeping, allergic reactions, fever[1][2] |
| Pregnancy category |
|
| Routes of use | Intravenous, IM, by mouth |
| Defined daily dose | 60 mg[3] |
| External links | |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a608038 |
| Legal | |
| License data | |
| Legal status |
|
| Pharmacokinetics | |
| Bioavailability | Dose dependent |
| Protein binding | ~15% |
| Elimination half-life | 6.2 hours |
| Excretion | Urinary |
| Chemical and physical data | |
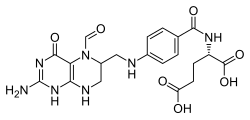
| Formula | C20H23N7O7 |
| Molar mass | 473.446 g·mol−1 |
| 3D model (JSmol) | |
| Melting point | 245 °C (473 °F) decomp |
| Solubility in water | ~0.3[4] mg/mL (20 °C) |
| |
| |
Folinic acid, also known as leucovorin, is a medication used to decrease the toxic effects of methotrexate and pyrimethamine.[1][2] It is also used in combination with 5-fluorouracil to treat colorectal cancer, may be used to treat folate deficiency that results in anemia, and methanol poisoning.[2][5] It is taken by mouth, injection into a muscle, or injection into a vein.[2]
Side effects may include trouble sleeping, allergic reactions, or fever.[1][2] Use in pregnancy or breastfeeding is generally regarded as safe.[1] When used for anemia it is recommended that pernicious anemia as a cause be ruled out first.[2] Folinic acid is a form of folic acid that does not require activation by dihydrofolate reductase to be useful to the body.[2]
Folinic acid was first made in 1945.[6] It is on the World Health Organization's List of Essential Medicines.[7] In the United Kingdom it costs the NHS about £4.62 per 30 mg vial.[1]
Medical use

Folinic acid is given following methotrexate as part of a total chemotherapeutic plan, where it may protect against bone marrow suppression or gastrointestinal mucosa inflammation. No apparent effect is seen on pre-existing methotrexate-induced nephrotoxicity.[8] Folinic acid can be taken as a pill (orally) or injected into a vein (intravenously) or muscle (intramuscularly).[9]
While not specifically an antidote for methotrexate, folinic acid may also be useful in the treatment of acute methotrexate overdose. Different dosing protocols are used, but folinic acid should be redosed until the methotrexate level is less than 5 x 10−8 M.[10]
Additionally, folinic acid is sometimes used to reduce the side effects of methotrexate in rheumatoid arthritis patients. This includes reductions in nausea, abdominal pain, abnormal liver blood tests, and mouth sores.[11]
Folinic acid is also used in combination with the chemotherapy agent 5-fluorouracil in treating colon cancer. In this case, folinic acid is not used for "rescue" purposes; rather, it enhances the effect of 5-fluorouracil by inhibiting thymidylate synthase.
Folinic acid is also sometimes used to prevent toxic effects of high doses of antimicrobial dihydrofolate reductase inhibitors such as trimethoprim and pyrimethamine. It may be prescribed in the treatment of toxoplasmosis retinitis, in combination with the folic acid antagonists pyrimethamine and sulfadiazine.
Folinic acid is also used in the treatment of cerebral folate deficiency, a syndrome in which the use of folic acid cannot normalize cerebrospinal fluid levels of 5-MTHF.[12]
Dosage
The defined daily dose is 60 mg by mouth or by injection.[3] The typical dose for isosporiasis is 5 to 15 mg once per day, while for toxoplasmosis it is 10 to 25 mg once per day, and when pyrimethamine is used it is 25 to 30 mg once per week.[13]
Side effects
Folinic acid should not be administered intrathecally. This may produce severe adverse effects or even death.[14]
Drug interactions
Fluorouracil: Folinic acid may increase the toxicity associated with fluorouracil if the two are administered together. Some adverse effects that have occurred, particularly in elderly patients, include severe enterocolitis, diarrhea, and dehydration.[15]
Sulfamethoxazole-trimethoprim: A potential drug interaction exists with concomitant use of sulfamethoxazole-trimethoprim and folinic acid. Folinic acid has been shown to decrease the efficacy of sulfamethoxazole-trimethoprim in the treatment of Pneumocystis jirovecii (formerly known as Pneumocystis carinii), a common cause of pneumonia in AIDS patients.[16]
Mechanism of action
Folinic acid is a 5-formyl derivative of tetrahydrofolic acid. It is readily converted to other reduced folic acid derivatives (e.g., 5,10-methylenetetrahydrofolate, 5-methyltetrahydrofolate), thus has vitamin activity equivalent to that of folic acid. Since it does not require the action of dihydrofolate reductase for its conversion, its function as a vitamin is unaffected by inhibition of this enzyme by drugs such as methotrexate. This is the classical view of folinic acid rescue therapy. In 1980s, however, folinic acid was found to reactivate the dihydrofolate reductase itself even when methotrexate exists. Although the mechanism is not very clear, the polyglutamylation of methotrexate and dihydrofolate in malignant cells is considered to play an important role in the selective reactivation of dihydrofolate reductase by folinic acid in normal cells.[17]
Folinic acid, therefore, allows for some purine/pyrimidine synthesis to occur in the presence of dihydrofolate reductase inhibition, so some normal DNA replication processes can proceed.
Folinic acid has dextro- and levorotary isomers. Both levoleucovorin (the levorotary isomer) and racemic folinic acid (a mixture of both isomers) have similar efficacy and tolerability.[18] Levoleucovorin was approved by the FDA in 2008.[19]
History
Folinic acid was discovered as a needed growth factor for the bacterium Leuconostoc citrovorum in 1948, by Sauberlich and Baumann. This resulted in it being called "citrovorum factor," meaning citrovorum growth factor. It had an unknown structure, but was found to be a derivative of folate that had to be metabolized in the liver before it could support growth of L. citrovorum. The synthesis of citrovorum factor by liver cells in culture was eventually accomplished from pteroylglutamic acid in the presence of suitable concentrations of ascorbic acid. The simultaneous addition of sodium formate to such systems resulted in increased citrovorum factor activity in the cell-free supernatants (producing, as now known, the 5-formyl derivative), and from this method of preparation of large amounts of the factor, its structure as levo-folinic acid (5-formyl tetrahydrofolic acid) was eventually deduced.[citation needed]
Names
Folinic acid should be distinguished from folic acid (vitamin B9). However, folinic acid is a vitamer for folic acid and has the full vitamin activity of this vitamin. Levofolinic acid and its salts are the 2S- form of the molecule. They are the only forms of the molecule that are known to be biologically active.
It is generally administered as the calcium or sodium salt (calcium folinate (INN), sodium folinate, leucovorin calcium, leucovorin sodium).
References
- ↑ 1.0 1.1 1.2 1.3 1.4 1.5 British national formulary : BNF 69 (69 ed.). British Medical Association. 2015. pp. 576–577. ISBN 9780857111562.
- ↑ 2.0 2.1 2.2 2.3 2.4 2.5 2.6 "Leucovorin Calcium". The American Society of Health-System Pharmacists. Archived from the original on 10 May 2017. Retrieved 8 December 2016.
- ↑ 3.0 3.1 "WHOCC - ATC/DDD Index". www.whocc.no. Archived from the original on 15 September 2020. Retrieved 26 August 2020.
- ↑ "Safety Data Sheet Folinic Acid (calcium salt)" (PDF). Archived (PDF) from the original on 15 September 2020. Retrieved 25 January 2018.
- ↑ Munjal YP, Sharm SK (2012). API Textbook of Medicine, Ninth Edition, Two Volume Set. JP Medical Ltd. p. 1945. ISBN 9789350250747. Archived from the original on 2017-05-10.
- ↑ Sneader, Walter (2005). Drug Discovery: A History. John Wiley & Sons. p. 235. ISBN 9780471899792. Archived from the original on 2017-05-10.
- ↑ World Health Organization (2019). World Health Organization model list of essential medicines: 21st list 2019. Geneva: World Health Organization. hdl:10665/325771. WHO/MVP/EMP/IAU/2019.06. License: CC BY-NC-SA 3.0 IGO.
- ↑ Therapeutic Information Resources Australia (2004). Calcium Folinate (Systemic) in AUSDI: Australian Drug Information for the Health Care Professional. Castle Hill: Therapeutic Information Resources Australia.[page needed]
- ↑ McGuire BW, Sia LL, Leese PT, Gutierrez ML, Stokstad EL (January 1988). "Pharmacokinetics of leucovorin calcium after intravenous, intramuscular, and oral administration". Clinical Pharmacy. 7 (1): 52–8. PMID 3257913.
- ↑ "Leucovorin" (PDF). CCO Formulary. Archived from the original (PDF) on 2008-10-10. Retrieved 2014-01-07.
- ↑ Shea B, Swinden MV, Tanjong Ghogomu E, Ortiz Z, Katchamart W, Rader T, et al. (May 2013). "Folic acid and folinic acid for reducing side effects in patients receiving methotrexate for rheumatoid arthritis". The Cochrane Database of Systematic Reviews. 5 (5): CD000951. doi:10.1002/14651858.CD000951.pub2. PMC 7046011. PMID 23728635.
- ↑ Gordon N (March 2009). "Cerebral folate deficiency". Developmental Medicine and Child Neurology. 51 (3): 180–2. doi:10.1111/j.1469-8749.2008.03185.x. PMID 19260931.
- ↑ "CALCIUM FOLINATE = FOLINIC acid oral - Essential drugs". medicalguidelines.msf.org. Archived from the original on 28 August 2021. Retrieved 26 August 2020.
- ↑ Jardine LF, Ingram LC, Bleyer WA (August 1996). "Intrathecal leucovorin after intrathecal methotrexate overdose". Journal of Pediatric Hematology/Oncology. 18 (3): 302–4. doi:10.1097/00043426-199608000-00014. PMID 8689347.
- ↑ Trissel LA, Martinez JF, Xu QA (April 1995). "Incompatibility of fluorouracil with leucovorin calcium or levoleucovorin calcium". American Journal of Health-System Pharmacy. 52 (7): 710–5. doi:10.1093/ajhp/52.7.710. PMID 7627739.
- ↑ Razavi B, Lund B, Allen BL, Schlesinger L (January 2002). "Failure of trimethoprim/sulfamethoxazole prophylaxis for Pneumocystis carinii pneumonia with concurrent leucovorin use". Infection. 30 (1): 41–2. doi:10.1007/s15010-001-1172-0. PMID 11876516.
- ↑ Goldman ID, Matherly LH (1987). "Biochemical factors in the selectivity of leucovorin rescue: selective inhibition of leucovorin reactivation of dihydrofolate reductase and leucovorin utilization in purine and pyrimidine biosynthesis by methotrexate and dihydrofolate polyglutamates". NCI Monographs (5): 17–26. PMID 2448654.
- ↑ Kovoor, P. A.; Karim, S. M.; Marshall, J. L. (2009). "Is levoleucovorin an alternative to racemic leucovorin? A literature review". Clinical Colorectal Cancer. 8 (4): 200–6. doi:10.3816/CCC.2009.n.034. PMID 19822510.
- ↑ Drugs.com (2008-05-07). "FDA Approves Levoleucovorin". Archived from the original on 2009-07-02. Retrieved 2009-06-07.
External links
- "Leucovorin". Drug Information Portal. U.S. National Library of Medicine. Archived from the original on 2020-02-03. Retrieved 2020-02-03.
- "Leucovorin". MedlinePlus. Archived from the original on 2020-09-15. Retrieved 2020-05-19.
| Identifiers: |
|
|---|
- Pages using duplicate arguments in template calls
- Wikipedia articles needing page number citations from February 2015
- Articles with invalid date parameter in template
- Chemical articles with unknown parameter in Infobox drug
- Chemical articles without CAS registry number
- Articles without EBI source
- Chemical pages without ChemSpiderID
- Chemical pages without DrugBank identifier
- Articles without KEGG source
- Articles without UNII source
- Drugs missing an ATC code
- Drugboxes which contain changes to verified fields
- Drugboxes which contain changes to watched fields
- All articles with unsourced statements
- Articles with unsourced statements from February 2015
- Articles with changed DrugBank identifier
- Chemotherapeutic adjuvants
- Folates
- World Health Organization essential medicines
- RTT