Dorzolamide
 | |
 | |
| Names | |
|---|---|
| Trade names | Trusopt, others |
| |
| Clinical data | |
| Pregnancy category |
|
| Routes of use | Topical (eye drops) |
| Defined daily dose | 0.3 ml[1] |
| External links | |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a602022 |
| Legal | |
| Legal status |
|
| Pharmacokinetics | |
| Protein binding | ~33% |
| Elimination half-life | 4 months |
| Chemical and physical data | |
| Formula | C10H16N2O4S3 |
| Molar mass | 324.43 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Dorzolamide, sold under the brand name Trusopt among others, is medications used to treat high pressure inside the eye including glaucoma.[2] It is used as an eye drop.[2] Effects begin within three hours and lasts for at least eight hours.[2] It is also available as the combination dorzolamide/timolol.[2]
Common side effects include eye discomfort, eye redness, taste changes, and blurry vision.[2] Serious side effects include Steven Johnson syndrome.[2] Those allergic to sulfonamides may be allergic to dorzolamide.[2][3] Use is not recommended in pregnancy or breastfeeding.[3] It is a carbonic anhydrase inhibitor and works by decreasing the production of aqueous humour.[2]
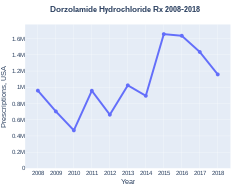
Dorzolamide was approved for medical use in the United States in 1994.[2] It is available as a generic medication.[3] A 5 milliliter bottle in the United Kingdom costs the NHS less than £2 as of 2019.[3] In the United States the wholesale cost of this amount is about US$7.10.[4] In 2017, it was the 281st most commonly prescribed medication in the United States, with more than one million prescriptions.[5][6]
Medical uses
Dorzolamide hydrochloride is used to lower excessive intraocular pressure in open-angle glaucoma and ocular hypertension.
Dosage
The defined daily dose is 0.3 ml.[1]
Side effects
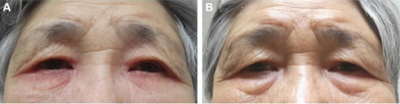
Ocular stinging, burning, itching and bitter taste.[7] It causes shallowing of the anterior chamber and leads to transient myopia.
Pharmacodynamics
It lowers IOP by about 20%.[7] Carbonic Anhydrase can convert H2CO3 into HCO3 (bicarbonate) and H+. The H+ is then exchanged for sodium (Na) which allows you to make aqueous humor. By blocking carbonic anhydrase, the Na/H exchanger can't work, which will decrease Na in the cell and prevent aqueous humor production.
History
This drug, developed by Merck, was the first drug in human therapy (market introduction 1995) that resulted from structure-based drug design. It was developed to circumvent the systemic side effects of acetazolamide which has to be taken orally.[7]
Society and culture
Cost
A 5 milliliter bottle in the United Kingdom costs the NHS less than £2 as of 2019.[3] In the United States the wholesale cost of this amount is about US$7.10.[4] In 2017, it was the 281st most commonly prescribed medication in the United States, with more than one million prescriptions.[5][6]
-
DorzolamideHydrochloride costs (US)
-
DorzolamideHydrochloride prescriptions (US)
References
- ↑ 1.0 1.1 "WHOCC - ATC/DDD Index". www.whocc.no. Archived from the original on 24 August 2019. Retrieved 9 September 2020.
- ↑ 2.0 2.1 2.2 2.3 2.4 2.5 2.6 2.7 2.8 "Dorzolamide Hydrochloride Monograph for Professionals". Drugs.com. American Society of Health-System Pharmacists. Archived from the original on 27 March 2019. Retrieved 26 March 2019.
- ↑ 3.0 3.1 3.2 3.3 3.4 British national formulary : BNF 76 (76 ed.). Pharmaceutical Press. 2018. p. 1148. ISBN 9780857113382.
- ↑ 4.0 4.1 "NADAC as of 2019-02-27". Centers for Medicare and Medicaid Services. Archived from the original on 2019-03-06. Retrieved 3 March 2019.
- ↑ 5.0 5.1 "The Top 300 of 2020". ClinCalc. Archived from the original on 12 February 2021. Retrieved 11 April 2020.
- ↑ 6.0 6.1 "Dorzolamide Hydrochloride - Drug Usage Statistics". ClinCalc. Archived from the original on 23 May 2020. Retrieved 11 April 2020.
- ↑ 7.0 7.1 7.2 KD Tripari MD. Essentials of Medical Pharmacology (5th ed.). Jaypee Brothers Medical Publishers(P) Ltd. p. 88. ISBN 81-8061-187-6.
Further reading
- Kubinyi H (1999). "Chance favors the prepared mind--from serendipity to rational drug design". J Recept Signal Transduct Res. 19 (1–4): 15–39. doi:10.3109/10799899909036635. PMID 10071748.
- Plummer C, MacKay E, Gelatt K (2006). "Comparison of the effects of topical administration of a fixed combination of dorzolamide-timolol to monotherapy with timolol or dorzolamide on IOP, pupil size, and heart rate in glaucomatous dogs". Vet Ophthalmol. 9 (4): 245–9. doi:10.1111/j.1463-5224.2006.00469.x. PMID 16771760.
- Grover S, Apushkin M, Fishman G (2006). "Topical dorzolamide for the treatment of cystoid macular edema in patients with retinitis pigmentosa". Am J Ophthalmol. 141 (5): 850–8. doi:10.1016/j.ajo.2005.12.030. PMID 16546110.
- Almeida G, Faria e Souza S (2006). "Effect of topical dorzolamide on rabbit central corneal thickness". Braz J Med Biol Res. 39 (2): 277–81. doi:10.1590/S0100-879X2006000200015. PMID 16470316.
== External links ==
| Identifiers: |
|
|---|
- Pages using duplicate arguments in template calls
- Chemical articles with unknown parameter in Infobox drug
- Chemical articles without CAS registry number
- Articles without EBI source
- Chemical pages without ChemSpiderID
- Chemical pages without DrugBank identifier
- Articles without KEGG source
- Articles without UNII source
- Drugs missing an ATC code
- Drugboxes which contain changes to verified fields
- Drugboxes which contain changes to watched fields
- RTT
- Carbonic anhydrase inhibitors
- Sulfones
- Sulfonamides
- Merck & Co. brands
- Amines
- Articles with changed CASNo identifier
- Articles with changed KEGG identifier