Acetylcholinesterase inhibitor
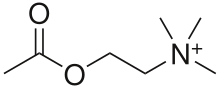

Acetylcholinesterase inhibitors (AChEIs) also often called cholinesterase inhibitors,[1] inhibit the enzyme acetylcholinesterase from breaking down the neurotransmitter acetylcholine into choline and acetate,[2] thereby increasing both the level and duration of action of acetylcholine in the central nervous system, autonomic ganglia and neuromuscular junctions, which are rich in acetylcholine receptors.[2] Acetylcholinesterase inhibitors are one of two types of cholinesterase inhibitors; the other being butyryl-cholinesterase inhibitors.[2] Acetylcholinesterase is the primary member of the cholinesterase enzyme family.[3]
Acetylcholinesterase inhibitors are classified as reversible, irreversible, or quasi-irreversible (also called pseudo-irreversible).[4][5]
Mechanism of action
Organophosphates
Organophosphates like TEPP and sarin inhibit cholinesterases, enzymes that hydrolyze the neurotransmitter acetylcholine.
The active centre of cholinesterases feature two important sites, namely the anionic site and the esteratic site. After the binding of acetylcholine to the anionic site of the cholinesterase, the acetyl group of acetylcholine can bind to the esteratic site. Important amino acid residues in the esteratic site are a glutamate, a histidine, and a serine. These residues mediate the hydrolysis of the acetylcholine.

At the esteratic site the acetylcholine is cleaved, which results in a free choline moiety and an acetylated cholinesterase. This acetylated state requires hydrolysis to regenerate itself.[6][7]
Inhibitors like TEPP modify the serine residue in the esteratic site of the cholinesterase.

This phosphorylation inhibits the binding of the acetyl group of the acetylcholine to the esteratic site of the cholinesterase. Because the acetyl group can't bind the cholinesterase, the acetylcholine can't be cleaved. Therefore, the acetylcholine will remain intact and will accumulate in the synapses. This results in continuous activation of acetylcholine receptors, which leads to the acute symptoms of TEPP poisoning.[8] The phosphorylation of cholinesterase by TEPP (or any other organophosphate) is irreversible. This makes the inhibition of the cholinesterase permanent.[6][7]
The cholinesterase gets irreversible phosphorylated according to the following reaction scheme
In this reaction scheme the E indicates the cholinesterase, PX the TEPP molecule, E–PX the reversible phosphorylated cholinesterase, k3 the reaction rate of the second step, EP the phosphorylated cholinesterase and X the leaving group of the TEPP.
The irreversible phosphorylation of the cholinesterase occurs in two steps. In the first step the cholinesterase gets reversibly phosphorylated. This reaction is very fast. Then the second step takes place. The cholinesterase forms a very stable complex with TEPP, in which TEPP is covalently bound to the cholinesterase. This is a slow reaction. But after this step the cholinesterase is irreversibly inhibited.[6]
The time dependent irreversible inhibition of the cholinesterase can be described by the following equation.[6]
In this formula, E is the remaining enzyme activity, E0 is the initial enzyme activity, t is the time interval after mixing of the cholinesterase and the TEPP, KI is the dissociation constant for cholinesterase-TEPP complex (E–PX) and I is the TEPP concentration.
The reaction mechanism and the formula above are both also compatible for other organophosphates. The process occurs in the same way.
Furthermore, certain organophosphates can cause OPIDN, organophosphate-induced delayed polyneuropathy. This is a disease, which is characterized by degeneration of axons in the peripheral and central nervous system. This disease will show a few weeks after contamination with the organophosphate. It is believed that the neuropathy target esterase (NTE) is affected by the organophosphate which induces the disease. However, there are no references found, which indicate that TEPP is one of the organophosphates that can cause OPIDN.[9]
Uses
Acetylcholinesterase inhibitors:[6]
- Occur naturally as venoms and poisons (e.g., onchidal)
- Are used as weapons (nerve agents)
- Are used medicinally:
- To treat myasthenia gravis, where they are used to increase neuromuscular transmission.
- To treat glaucoma
- To treat postural tachycardia syndrome
- As an antidote to anticholinergic poisoning
- To reverse the effect of non-depolarising muscle relaxants
- To treat neuropsychiatric symptoms of diseases such as Alzheimer's disease, particularly apathy
- To increase chances of lucid dreaming (by prolonging REM sleep)[10]
- To treat Alzheimer's disease, the Lewy body dementias and Parkinson's disease. In these neurodegenerative conditions AChEIs are primarily used to treat the cognitive (memory and learning deficits mostly) symptoms of dementia. These symptoms are attenuated due to the role of acetylcholine in cognition in the CNS. There is some evidence to suggest that AChEIs may attenuate psychotic symptoms (especially visual hallucinations) in Parkinson's disease.[11]
- To treat cognitive impairments in patients with schizophrenia. There is some evidence to suggest efficacy in treating positive, negative and affective symptoms.[12][13][14]
- As a treatment for autism and to increase the percentage of rapid eye movement sleep in autistic children, in line with the mechanism by which they encourage lucid dreaming.[15][16]
- Are used as insecticides (e.g., malathion):
- Resistance: The hunt for resistance genes in Rhipicephalus microplus has been hampered by high copy number in the three AChEs involved.[17] Bellgard et al. 2012, Temeyer et al. 2012, and Bendele et al. 2015 all investigate such populations and encounter difficulty confirming the involvement of these three due to copy number.[17]
Guideline recommendations
The clinical guidelines for medication management in people with dementia recommend trialing an AChE inhibitor for people with early- to mid-stage dementia. These guidelines, known as the Medication Appropriateness Tool for Comorbid Health conditions in Dementia (MATCH-D), suggest that these medicines are at least considered.[18][failed verification]
Side effects
| Potential side effects of acetylcholinesterase inhibitors[19][20] | |||
|---|---|---|---|
| mild – generally diminishes | potentially serious | ||
| |||
Some major effects of cholinesterase inhibitors:
- Actions on the parasympathetic nervous system, (the parasympathetic branch of the autonomic nervous system) may cause bradycardia, hypotension, hypersecretion, bronchoconstriction, GI tract hypermotility, and decrease intraocular pressure, increase lower esophageal sphincter (LES) tone
- Cholinergic crisis.
- Actions on the neuromuscular junction may result in prolonged muscle contraction.[21]
- The effects of neostigmine on postoperative nausea and vomiting are controversial and there is not a clear linkage in clinical practice, however, there is good evidence to support the reduction in risk when anticholinergic agents are administered.[22]
Administration of reversible cholinesterase inhibitors is contraindicated with those that have urinary retention due to urethral obstruction.
Overdose
Hyperstimulation of nicotinic and muscarinic receptors.[4]
Titration phase
When used in the central nervous system to alleviate neurological symptoms, such as rivastigmine in Alzheimer's disease, all cholinesterase inhibitors require doses to be increased gradually over several weeks, and this is usually referred to as the titration phase. Many other types of drug treatments may require a titration or stepping up phase. This strategy is used to build tolerance to adverse events or to reach a desired clinical effect.[20] This also prevents accidental overdose and is therefore recommended when initiating treatment with drugs that are extremely potent and/or toxic (drugs with a low therapeutic index).
Examples
Reversible inhibitor
Compounds which function as reversible competitive or noncompetitive inhibitors of cholinesterase are those most likely to have therapeutic uses. These include:
- Some organophosphates not listed under "Irreversible" below
- Carbamates
- Phenanthrene derivatives
- Caffeine – noncompetitive (also an adenosine receptor antagonist)[23]
- Rosmarinic acid – ester of caffeic acid. Found in plants species of family Lamiaceae.[24]
- Alpha-pinene – noncompetitive reversible [25][26]
- Piperidines
- Tacrine, also known as tetrahydroaminoacridine (THA)
- Edrophonium
- Huperzine A[27][28]
- Ladostigil
- Ungeremine[29]
- Lactucopicrin
- Acotiamide
- Hybrid/bitopic ligands[30]
Comparison table
| Inhibitor | Duration | Main site of action | Clinical use | Adverse effects |
|---|---|---|---|---|
| Edrophonium | short (10 min.)[31] | neuromuscular junction[31] | diagnosis of myasthenia gravis[31] | |
| Neostigmine | medium (1–2 hrs.)[31] | neuromuscular junction[31] |
|
visceral[31] |
| Physostigmine | medium (0.5–5 hrs.)[31] | postganglionic parasympathetic[31] | treat glaucoma (eye drops)[31] | |
| Pyridostigmine | medium (2–3 hrs.)[31] | neuromuscular junction[31] |
|
|
| Dyflos | long[31] | postganglionic parasympathetic[31] | historically to treat glaucoma (eye drops)[31] | toxic[31] |
| Echothiophate (irreversible) | long[31] | postganglionic parasympathetic[31] | treat glaucoma (eye drops)[31] | systemic effects[31] |
| Parathion (irreversible) | long[31] | none[31] | toxic[31] |
Quasi-irreversible inhibitor
Compounds which function as quasi-irreversible inhibitors of cholinesterase are those most likely to have use as chemical weapons or pesticides.
- Organophosphates
- Carbamates
- Atypical inhibitors
See also
References
- ^ "Medications for treating people with dementia" (PDF). Retrieved 1 January 2021.
- ^ a b c English, Brett A.; Webster, Andrew A. (2012). "Acetylcholinesterase and its Inhibitors". Primer on the Autonomic Nervous System. Elsevier. pp. 631–633. doi:10.1016/b978-0-12-386525-0.00132-3. ISBN 978-0-12-386525-0.
- ^ Seth (2009-11-18). "23". Textbook Of Pharmacology. Elsevier India. p. III.87. ISBN 978-8131211588.
Anaesthesia: Cholinesterase inhibitors are likely to exaggerate succinylcholine-type muscle relaxation during anaesthesia. 5. Genitourinary system: It may ...
- ^ a b Colović MB, Krstić DZ, Lazarević-Pašti TD, Bondžić AM, Vasić VM (May 2013). "Acetylcholinesterase inhibitors: pharmacology and toxicology". Current Neuropharmacology. 11 (3). Bentham Science Publishers Ltd.: 315–35. doi:10.2174/1570159x11311030006. PMC 3648782. PMID 24179466.
- ^ McGleenon BM, Dynan KB, Passmore AP (October 1999). "Acetylcholinesterase inhibitors in Alzheimer's disease". British Journal of Clinical Pharmacology. 48 (4): 471–80. doi:10.1046/j.1365-2125.1999.00026.x. PMC 2014378. PMID 10583015.
- ^ a b c d e f g Colović MB, Krstić DZ, Lazarević-Pašti TD, Bondžić AM, Vasić VM (May 2013). "Acetylcholinesterase inhibitors: pharmacology and toxicology". Current Neuropharmacology. 11 (3): 315–35. doi:10.2174/1570159X11311030006. PMC 3648782. PMID 24179466.
- ^ a b O'Brien RD (2013-10-22). Toxic Phosphorus Esters: Chemistry, Metabolism, and Biological Effects. Elsevier. ISBN 978-1-4832-7093-7.
- ^ Roberts SM, James RC, Williams PL (2014-12-08). Principles of Toxicology: Environmental and Industrial Applications. John Wiley & Sons. ISBN 978-1-118-98248-8.
- ^ Lotti M, Moretto A (2005-01-01). "Organophosphate-induced delayed polyneuropathy". Toxicological Reviews. 24 (1): 37–49. doi:10.2165/00139709-200524010-00003. PMID 16042503. S2CID 29313644.
- ^ Yuschak T (2006). Advanced Lucid Dreaming: The Power of Supplements. Lulu. ISBN 978-1430305422.
- ^ Taylor D, Paton C, Shitij K (2012). Maudsley Prescribing Guidelines in Psychiatry (11th ed.). West Sussex: Wiley-Blackwell. ISBN 978-0-47-097948-8.
- ^ Singh J, Kour K, Jayaram MB (January 2012). "Acetylcholinesterase inhibitors for schizophrenia". The Cochrane Database of Systematic Reviews. 1 (1): CD007967. doi:10.1002/14651858.CD007967.pub2. PMC 6823258. PMID 22258978.
- Lay summary in: Singh J, Kour K, Jayaram MB. "Acetylcholinesterase inhibitors versus antipsychotics for schizophrenia either alone or in combination with antipsychotics". Cochrane (Abstract).
- ^ Choi KH, Wykes T, Kurtz MM (September 2013). "Adjunctive pharmacotherapy for cognitive deficits in schizophrenia: meta-analytical investigation of efficacy". The British Journal of Psychiatry. 203 (3): 172–8. doi:10.1192/bjp.bp.111.107359. PMC 3759029. PMID 23999481.
- ^ Ribeiz SR, Bassitt DP, Arrais JA, Avila R, Steffens DC, Bottino CM (April 2010). "Cholinesterase inhibitors as adjunctive therapy in patients with schizophrenia and schizoaffective disorder: a review and meta-analysis of the literature". CNS Drugs. 24 (4): 303–17. doi:10.2165/11530260-000000000-00000. PMID 20297855. S2CID 45807136.
- ^ Buckley AW, Sassower K, Rodriguez AJ, Jennison K, Wingert K, Buckley J, Thurm A, Sato S, Swedo S (August 2011). "An open label trial of donepezil for enhancement of rapid eye movement sleep in young children with autism spectrum disorders". Journal of Child and Adolescent Psychopharmacology. 21 (4): 353–7. doi:10.1089/cap.2010.0121. PMC 3157749. PMID 21851192.
- ^ Handen BL, Johnson CR, McAuliffe-Bellin S, Murray PJ, Hardan AY (February 2011). "Safety and efficacy of donepezil in children and adolescents with autism: neuropsychological measures". Journal of Child and Adolescent Psychopharmacology. 21 (1): 43–50. doi:10.1089/cap.2010.0024. PMC 3037196. PMID 21309696.
- ^ a b Chatonnet, Arnaud; Lenfant, Nicolas; Marchot, Pascale; Selkirk, Murray E. (2017-04-05). "Natural genomic amplification of cholinesterase genes in animals". Journal of Neurochemistry. 142. International Society for Neurochemistry (Wiley): 73–81. doi:10.1111/jnc.13990. hdl:10044/1/48129. ISSN 0022-3042. PMID 28382676. S2CID 34155509.
- ^ Page AT, Potter K, Clifford R, McLachlan AJ, Etherton-Beer C (October 2016). "Medication appropriateness tool for co-morbid health conditions in dementia: consensus recommendations from a multidisciplinary expert panel". Internal Medicine Journal. 46 (10): 1189–1197. doi:10.1111/imj.13215. PMC 5129475. PMID 27527376.
- ^ Consumer Reports; Drug Effectiveness Review Project (May 2012). "Evaluating Prescription Drugs Used to Treat: Alzheimer's Disease Comparing Effectiveness, Safety, and Price" (PDF). Best Buy Drugs: 2. Archived (PDF) from the original on 5 September 2012. Retrieved 1 May 2013., which claims Alzheimer's Association guidance as a source
- ^ a b Inglis F (June 2002). "The tolerability and safety of cholinesterase inhibitors in the treatment of dementia". International Journal of Clinical Practice. Supplement (127): 45–63. PMID 12139367.
- ^ Singh, Ravneet; Sadiq, Nazia M. (2020), "Cholinesterase Inhibitors", StatPearls, Treasure Island (FL): StatPearls Publishing, PMID 31335056, retrieved 2020-10-12
- ^ Barash PG, Cullen BF, Stoelting RK, Cahalan MK, Stock MC (15 April 2013). Clinical Anesthesia (7th ed.). Lippincott Williams & Wilkins. pp. 552–554. ISBN 978-1-4511-4419-2.
- ^ Karadsheh N, Kussie P, Linthicum DS (March 1991). "Inhibition of acetylcholinesterase by caffeine, anabasine, methyl pyrrolidine and their derivatives". Toxicology Letters. 55 (3): 335–42. doi:10.1016/0378-4274(91)90015-X. PMID 2003276.
- ^ Vladimir-Knežević S, Blažeković B, Kindl M, Vladić J, Lower-Nedza AD, Brantner AH (January 2014). "Acetylcholinesterase inhibitory, antioxidant and phytochemical properties of selected medicinal plants of the Lamiaceae family". Molecules. 19 (1): 767–82. doi:10.3390/molecules19010767. PMC 6271370. PMID 24413832.
- ^ Miyazawa M, Yamafuji C (March 2005). "Inhibition of acetylcholinesterase activity by bicyclic monoterpenoids". Journal of Agricultural and Food Chemistry. 53 (5): 1765–8. doi:10.1021/jf040019b. PMID 15740071.
- ^ Perry NS, Houghton PJ, Theobald A, Jenner P, Perry EK (July 2000). "In-vitro inhibition of human erythrocyte acetylcholinesterase by salvia lavandulaefolia essential oil and constituent terpenes". The Journal of Pharmacy and Pharmacology. 52 (7): 895–902. doi:10.1211/0022357001774598. PMID 10933142. S2CID 34457692.
- ^ Bauer BA. "Huperzine A: Can it treat Alzheimer's?". Mayo Clinic. Archived from the original on 2012-08-19.
- ^ Wang BS, Wang H, Wei ZH, Song YY, Zhang L, Chen HZ (April 2009). "Efficacy and safety of natural acetylcholinesterase inhibitor huperzine A in the treatment of Alzheimer's disease: an updated meta-analysis". Journal of Neural Transmission. 116 (4): 457–65. doi:10.1007/s00702-009-0189-x. PMID 19221692. S2CID 8655284.
- ^ Rhee IK, Appels N, Hofte B, Karabatak B, Erkelens C, Stark LM, Flippin LA, Verpoorte R (November 2004). "Isolation of the acetylcholinesterase inhibitor ungeremine from Nerine bowdenii by preparative HPLC coupled on-line to a flow assay system". Biological & Pharmaceutical Bulletin. 27 (11): 1804–9. doi:10.1248/bpb.27.1804. PMID 15516727.
- ^ Messerer R, Dallanoce C, Matera C, Wehle S, Flammini L, Chirinda B, et al. (June 2017). "Novel bipharmacophoric inhibitors of the cholinesterases with affinity to the muscarinic receptors M1 and M2". MedChemComm. 8 (6): 1346–1359. doi:10.1039/c7md00149e. PMC 6072511. PMID 30108847.
- ^ a b c d e f g h i j k l m n o p q r s t u v w x y Rang HP (2003). Pharmacology. Edinburgh: Churchill Livingstone. p. 156. ISBN 978-0-443-07145-4.
External links
- Acetylcholinesterase+inhibitors at the U.S. National Library of Medicine Medical Subject Headings (MeSH)
- Acetylcholinesterase: A gorge-ous enzyme QUite Interesting PDB Structure article at PDBe
![{\displaystyle {\ce {E + PX <=> E-PX ->[k_3] EP + X}}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/896525e22a1d1b440d2154f6a1cde9e1ddbef67d)
