Triple X syndrome
| Triple X syndrome | |
|---|---|
| Other names: Triplo-X syndrome, trisomy X, XXX syndrome, 47,XXX syndrome[1] | |
 | |
| Increased distance between the eyes in 9 year old girl with trisomy X[2] | |
| Specialty | Medical genetics |
| Symptoms | Taller than average[1] |
| Complications | Learning difficulties, decreased muscle tone, seizures, kidney problems[1] |
| Causes | Random event[1] |
| Diagnostic method | Chromosomal analysis[3] |
| Treatment | Speech therapy, physical therapy, counseling[3] |
| Frequency | 1 per 1,000 female births[2] |
Triple X syndrome, also known as trisomy X and 47,XXX, is characterized by the presence of an extra X chromosome in each cell of a female.[1] Those affected are often taller than average.[1] Usually there are no other physical differences and normal fertility.[1] Occasionally there are learning difficulties, decreased muscle tone, seizures, or kidney problems.[1]
Triple X is due to a random event.[1] Triple X can result either during the division of the mother's reproductive cells or during division of cells during early development.[2] It is not typically inherited from one generation to the next.[1] A form where only a percentage of the body cells contain XXX can also occur.[1] Diagnosis is by chromosomal analysis.[3]
Treatment may include speech therapy, physical therapy, and counseling.[3] It occurs in about one in every 1,000 female births.[2] It is estimated that 90% of those affected are not diagnosed as they either have no or only few symptoms.[2] It was first identified in 1959 by geneticist Patricia Jacobs.[4]
Signs and symptoms
Trisomy X has variable effects, ranging from no symptoms at all to significant disability.[5] Severity varies between people diagnosed prenatally (before birth) and postnatally (after birth), and postnatal cases are more severe on average.[6] Symptoms associated with trisomy X include tall stature, mild developmental delay, subtle physical and skeletal anomalies, increased rates of mental health concerns, and earlier age of menopause.[5][7]
-
Epicanthal folds and increased distance between the eyes in 2-year-old girl with trisomy X[2]
-
The type of finger curvature frequently seen in triple X syndrome
-
An adult with trisomy X
Physical
The physical and physiological impacts of trisomy X tend to be subtle.[5] Tall stature is one of the major physical associations of trisomy X. Prior to age four, most girls with trisomy X are average height; growth picks up after this age, and is particularly rapid between the ages of four and eight. Of girls with trisomy X aged six to thirteen, 40% are above the 90th percentile in height.[7] The average adult height in trisomy X has been estimated as 172 cm (5 ft 7+1⁄2 in) and head circumference as 20th percentile.[8][note 1] The added height in trisomy X is primarily in the limbs, with long legs and a shorter sitting height.[5] Though head circumference is generally below the 50th percentile,[7] microcephaly, a head circumference below the 5th percentile, is rare.[5]
Minor skeletal and craniofacial anomalies are associated with trisomy X. Subtle dysmorphisms seen in some females with trisomy X include hypertelorism (wide-spaced eyes), epicanthic folds (an additional fold of skin in the corners of the eyes), and upslanting palpebral fissures (the opening between the eyelids). These differences are usually minor and do not impact the daily lives of girls and women with the condition.[5] Other skeletal anomalies associated with trisomy X include clinodactyly (incurved little fingers), radioulnar synostosis (the fusion of the long bones in the forearm),[11] flat feet, and hyper-extensible joints.[12] These findings are not unique to trisomy X, but rather are seen in sex chromosome aneuploidy disorders as a whole.[13]
Severe internal disease is rare in trisomy X. Genitourinary conditions are more common than in the general population, particularly kidney and ovary malformations.[5] One study found that the autoimmune disorders lupus and Sjögren syndrome are more common in trisomy X than in the general population.[14] Conditions such as sleep apnea, asthma, scoliosis, and hip dysplasia have also been linked to sex chromosome aneuploidies as a whole, including trisomy X.[13] Although heart defects are common in pentasomy X,[15] they are no more frequent in trisomy X than the general population.[7]
Puberty starts around the expected age and progresses as normal.[16] Fertility is normal when not complicated by early menopause;[5] a large population study found women with 47,XXX karyotypes to average 1.9 lifetime pregnancies, compared to 2.3 for women with "normal" 46,XX karyotypes, and to be no more likely to miscarry.[17] Premature ovarian failure (POF), or early menopause, is a known complication of trisomy X. Premature ovarian failure is defined as menopause before the age of 40; in the general population, 1 in 100 women experience menopause before this age, 1 in 1,000 before age 30, and 1 in 10,000 before age 20. Amongst women with POF, 3% have trisomy X, compared to 1 in 1,000 in the general population.[18] The average age of menopause for women with trisomy X is 45 years, compared to 50 years for women with 46,XX karyotypes.[17] POF is more common in women with trisomy X who also have autoimmune disorders.[5]
Neurological

General cognitive functioning is reduced in trisomy X, with an average intelligence quotient of 85–90.[5] Performance IQ tends to be higher than verbal IQ.[19] Though intellectual disability is rare, it is more prevalent than in the general population, occurring in about 5–10% of females with trisomy X[5] compared to approximately 1% of the broader population.[20] While the average is depressed, the effect of trisomy X varies substantially, and some women are highly intelligent.[21]
Infant milestones are normal to slightly delayed. A patient support organization reports crawling around the age of ten months and walking around sixteen to eighteen months, with first words acquired shortly after one year of age and fluent speech around age two.[16] Speech therapy is needed in 40%–90% of girls with trisomy X at some point in their lives,[19] compared to around 3–8% of children in the general population.[22][23] Expressive language skills tend to be more affected than receptive skills.[24]
Neuroimaging in trisomy X demonstrates decreased whole brain volumes, correlated with overall intellectual functioning. Amygdala volume may be smaller than expected after controlling for whole brain size. White matter abnormalities have been reported, although their significance is unknown. These findings are common to X-chromosome polysomy syndromes, and is seen in males with Klinefelter syndrome.[5][19] Epilepsy or electroencephalogram abnormalities may be more common in those with trisomy X, particularly those who are also intellectually disabled.[5][25] Epilepsy in sex chromosome aneuploidies as a whole is mild, amenable to treatment, and often attenuates or disappears with time. Tremor is reported in approximately a quarter of women with trisomy X and responds to the same treatments as in the general population.[25]
Autism spectrum disorders are more common in trisomy X, occurring in approximately 15% of girls with trisomy X[24] compared to less than 1% of girls in the general population.[26] Though much of the research is in children, research in adult women with trisomy X suggests higher rates of autistic symptomatology than the general population.[27] Executive dysfunction, where people have difficulty regulating their actions and emotions, is more prevalent amongst those with trisomy X than the general population.[19][24]
Psychological
The psychosocial adaptation of girls and women with trisomy X is dependent on environmental factors. Girls growing up in stable environments with healthy home lives tend to have relatively high adaptive and social functioning, while significant behavioural and psychological issues are predominantly seen in those from troubled social environments.[7] Though girls with trisomy X usually have good relationships with peers, they trend towards immaturity;[19] some behavioural issues in children with trisomy X are thought to be a consequence of the disconnect between apparent age, as understood via increased height, and cognitive and emotional maturity encouraging hard-to-reach expectations.[7] Girls whose motor and language skills are more severely affected by trisomy X often experience low confidence and self-esteem.[28] These traits vary in severity; though some women with trisomy X are significantly impaired, many are within the normal range of variance, and some are high-functioning and high-achieving.[21]
Some mental health issues are more frequent in women with trisomy X. Dysthymia and cyclothymia, milder forms of depression and bipolar disorder respectively, are more common than in the general population.[5][7] Women with trisomy X average higher schizotypy, reporting higher levels of introversion, magical thinking, and impulsivity.[19] Sex chromosome aneuploidies are associated with psychosis, and schizophrenic women are more likely to have trisomy X than the general female population.[29] The prevalence of trisomy X in women with adult-onset schizophrenia is estimated to be around 1 in 400, compared to 1 in 1,000 in women as a whole; the prevalence in childhood onset schizophrenia is unclear, but may be as high as 1 in 40.[30] Approximately one-fifth of women with trisomy X report clinically significant levels of anxiety.[24] Women with trisomy X are often "late bloomers", experiencing high rates of psychological distress into early adulthood, but by their mid-thirties having stronger interpersonal bonds and healthy relationships.[19] The study of mental health in trisomy X is complicated by the fact that girls and women who were diagnosed before birth seem to be more mildly affected than those diagnosed after. For instance, psychogenic stomach pains are reported in a disproportionate number of postnatally diagnosed patients, but fewer prenatally diagnosed ones.[16]
Mosaic forms
The most common karyotype in trisomy X is 47,XXX, where all cells have an additional copy of the X chromosome. Mosaicism, where both 47,XXX and other cell lines are present, occurs in approximately 10% of cases. Mosaic trisomy X has different outcomes to the non-mosaic condition. Common mosaic forms observed include 46,XX/47,XXX, 45,X0/47,XXX (with a Turner syndrome cell line), and 47,XXX/48,XXXX (with a tetrasomy X cell line). Complex mosaicism, with cell lines such as 45,X0/46,XX/47,XXX, can also be seen.[5]
46,XX/47,XXX
The simplest form of mosaic trisomy X, with a 46,XX/47,XXX karyotype, is milder compared to full trisomy X. Cognitive development is more typical, with improved long-term life outcomes. Although generally milder, 46,XX/47,XXX mosaicism is associated with a higher risk of chromosome anomalies in offspring than full trisomy X.[28] The increased risk of abnormal offspring in mosaicism has been hypothesized to be a consequence of oocyte abnormality in 46,XX/47,XXX women not seen in full 47,XXX. Some writers have recommended women with 46,XX/47,XXX karyotypes undergo screening for chromosomal disorders during pregnancy.[5][31]
45,X0/47,XXX

Between 3% and 15% of females with Turner syndrome, defined by a karyotype with a single copy of the X chromosome, have a 47,XXX cell line.[5][32] Mosaic karyotypes with both 45,X0 and 47,XXX cells are considered Turner syndrome rather than trisomy X, but the presence of 47,XXX cells influences the disorder. Non-mosaic Turner syndrome is characterized by primary amenorrhea and failure to begin or complete puberty, while 80–90% of women with 45,X0/47,XXX mosaicism begin puberty naturally and approximately 60–80% have spontaneous menses.[33] Around two-thirds of 45,X0/47,XXX mosaics have clinically significant short stature, compared with virtually all women with non-mosaic Turner syndrome.[33] Although women with trisomy X have lower IQs than the general population and women with Turner syndrome do not, intellectual disability does not appear to be more common in the mosaic than for non-mosaic Turner's.[34] Women with mosaic Turner syndrome tend to have similar dysmorphic features to those with non-mosaic Turner's syndrome, but less marked, and some have none of the traditional Turner's stigmata.[35]
Turner syndrome is characteristically associated with sterility, and only 2–7% of women with Turner's, including mosaic cases, are capable of pregnancy. In contrast, Turner's women with 47,XXX cell lines often complete puberty and are typically fertile.[36] Women with this mosaicism experience premature ovarian failure; the average age of menopause in these women is around 30, with the onset of likely perimenopausal irregular menses occurring around age 20.[33]
47,XXX/48,XXXX
Mosaicism with a tetrasomy X cell line generally appears more severe than typical trisomy X.[28] Like trisomy X, tetrasomy X has a variable phenotype muddled by underdiagnosis. The tetrasomy is generally more severe than the trisomy; intellectual disability is characteristic, dysmorphic features more visible, and puberty often altered.[5][28]
Causes
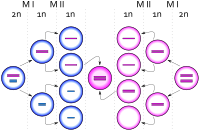
Trisomy X, like other aneuploidy disorders, is caused by a process called nondisjunction. Nondisjunction occurs when homologous chromosomes or sister chromatids fail to separate properly during meiosis, the process that produces gametes (eggs or sperm), and result in gametes with too many or too few chromosomes.[39] Nondisjunction can occur during gametogenesis, where the trisomy is present from conception, or zygote development, where it occurs after conception.[5] When nondisjunction occurs after conception, the resulting karyotype is generally mosaic, with both 47,XXX and other cell lines.[40]
Most cases of trisomy X occur through maternal nondisjunction, with around 90% of cases traced to errors in oogenesis.[28] The vast majority of cases of trisomy X occur randomly; they have nothing to do with the chromosomes of the parents and little chance of recurring in the family.[16] Nondisjunction is related to advanced maternal age, and trisomy X specifically appears to have a small but significant maternal age effect.[5] In a cohort of women with trisomy X born in the 1960s, the average maternal age was 33.[7] The risk of women with full trisomy X having chromosomally abnormal children is low, likely below 1%. Recurrence may occur if the mother has mosaicism for trisomy X, particularly in ovarian cells, but this makes up a small fraction of cases.[16]
Diagnosis
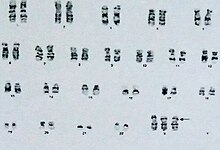
Chromosome aneuploidies such as trisomy X are diagnosed via karyotype,[41] the process in which chromosomes are tested from blood, bone marrow, amniotic fluid, or placental cells.[42] As trisomy X is generally mild or asymptomatic, most cases are never diagnosed. Around 10% of cases of trisomy X are diagnosed in the person's lifetime; many are ascertained coincidentally during prenatal testing via amniocentesis or chorionic villi sampling, which is routinely performed for advanced maternal age.[5] Postnatal testing is typically prompted by tall stature,[43] hypotonia, developmental disability or neurodivergence, mild dysmorphic features such as hypertelorism or clinodactyly, and premature ovarian failure.[5]
Differential diagnosis
Tetrasomy X, characterized by four copies of the X chromosome, has some signs in common with more severe cases of trisomy X. Intellectual disability, generally mild, is more frequently seen in the tetrasomy than the trisomy. There is more of a tendency towards noticeable dysmorphic features such as hypertelorism, clinodactyly, and epicanthic folds. Unlike trisomy X, approximately half of women with tetrasomy X have no or incomplete pubertal development. Although in most cases tetrasomy X is significantly more severe than trisomy X, some cases of tetrasomy X are mild, and some cases of trisomy X severe. Like trisomy X, the full phenotypic range of tetrasomy X is unknown due to underdiagnosis.[11][44] Pentasomy X, with five X chromosomes, may rarely be a differential diagnosis for trisomy X. The phenotype of pentasomy X is more severe than the trisomy or tetrasomy, with significant intellectual disability, heart defects, microcephaly, and short stature.[11]
Due to overlapping dysmorphic features, such as epicanthic folds and upslanting palpebral fissures, some cases of trisomy X may be ascertained due to suspicion of Down syndrome.[5] When the primary symptom is tall stature, trisomy X may be considered alongside other conditions depending on the rest of the phenotype. Marfan syndrome may be considered due to the disproportion between limb and torso length observed in both syndromes, as well as both experiencing joint issues. Beckwith-Wiedemann syndrome, another disproportionate tall stature syndrome, can cause developmental disability similar to that seen in some cases of trisomy X.[43]
As karyotypic diagnosis is conclusive, differential diagnosis can be abandoned after karyotype in most cases of trisomy X. However, due to the relatively high prevalence of trisomy X, other congenital disorders may occur alongside a 47,XXX karyotype. Differential diagnosis remains indicated when the phenotype is particularly severe for what a 47,XXX karyotype alone explains, such as severe intellectual disability or significant malformation.[5]
Treatment
Treatment may include speech therapy, physical therapy, and counseling.[3] A stable home environment can improve some of the symptoms.[45]
Prognosis
Parent of a daughter with trisomy X[46]
The prognosis of trisomy X is broadly good, with adult independence most often achieved, if delayed. Most adults achieve normal life outcomes, pursuing education, employment, or homemaking.[46] Childhood and adolescence, particularly in compulsory education, tends to be more difficult for those with trisomy X than adult life. Parents report their daughters' struggling both academically and socially at school,[47] particularly during secondary education,[46] while adults report better adaptation after leaving education and entering the workforce.[7] Of the women in the cohort studies followed to early adulthood, 7 of 37 dropped out of high school, while three attended university.[7] Compared to age-matched women in the general population, women with trisomy X are 68% as likely to live with a partner, 64% as likely to have children, 36% as likely to hold higher education qualifications, and almost twice as likely to be retired from the workforce.[48]
Physical health is generally good and many women with trisomy X live into old age.[16] Little data exists on aging in trisomy X.[7] Data from the Danish Cytogenetic Central Register, which covers 13% of women with trisomy X in Denmark,[49] suggests a life expectancy of 71 for women with full trisomy X and 78 for mosaics, compared to 84 for controls.[50] The limited sample, composed only of women with trisomy X who have come to medical attention, has led to speculation this number is an underestimate.[48]
Women with trisomy X who were diagnosed prenatally have better outcomes as a group than those diagnosed postnatally, and 46,XX/47,XXX mosaics better than those with full trisomy X.[5] Some of the improved outcome in prenatal diagnosis appears to be a function of higher socioeconomic status amongst parents.[7]
Epidemiology
Trisomy X is a relatively common genetic disorder, occurring in around 1 in 1,000 female births. Despite this prevalence, only around 10% of cases are diagnosed during their lifetime.[5] Large cytogenetic studies in Denmark find a diagnosed prevalence of 6 in 100,000 females, around 7% of the actual number of girls and women with trisomy X expected to exist in the general population.[49] Diagnosis in the United Kingdom is particularly low, with an estimated 2% of cases medically recognized.[48] Amongst the 244,000 women in the UK Biobank research sample, 110 were found to have 47,XXX karyotypes, corresponding to approximately half the number expected in the population. The fact this number is still reduced compared to the broader population is thought to be an effect of UK Biobank participants being less likely to be of low IQ and low socioeconomic status than the general population, both of which are more frequent in trisomy X.[17]
Trisomy X only occurs in females, as the Y chromosome is in most cases necessary for male sexual development.[16][note 2] In addition to its high base rate, trisomy X is more common in some clinical subpopulations. The karyotype occurs in an estimated 3% of women with early menopause,[18] 1 in 350 with Sjögren syndrome, and 1 in 400 with systemic lupus erythematosus.[14]
-
Expected and observed number of people diagnosed with trisomy X and Turner syndrome in Denmark
-
Violin plots of age at diagnosis for Klinefelter syndrome, trisomy X, and XYY syndrome
History
The first known case of trisomy X, in a 176 cm (5 ft 9+1⁄2 in) woman who experienced premature ovarian failure at the age of 19, was diagnosed in 1959 by a team led by Patricia Jacobs.[7][54] The late 1950s and early 1960s were a period of frequent ascertainment of previously unknown sex chromosome aneuploidies, with the 47,XXX karyotype discovered alongside 45,X0 and 47,XXY the same year.[7][55][56] Early studies on sex chromosome aneuploidy screened patients residing in institutions, depicting the karyotypes as incapacitating; even at the time, this research was criticized for giving an inaccurate portrait of sex chromosome aneuploidy.[57] Early reports of women with trisomy X have since been criticized for a dehumanizing ableist perspective, showing nude photographs of institutionalized women described as "mental deficiency patients".[58]
In response to the biased early studies, a newborn screening program for sex chromosome aneuploidy disorders was implemented in the 1960s.[59] Almost 200,000 neonates were screened in Aarhus, Toronto, New Haven, Denver, Edinburgh, and Winnipeg; those found to have sex chromosome aneuploidies were followed up for 20 years for most of the cohorts, and longer for the Edinburgh and Denver cohorts.[7] The children with trisomy X and Klinefelter's had their karyotypes disclosed to their parents, but due to the then-present perception that XYY syndrome was associated with violent criminality, those diagnoses were hidden from the family.[59]
These studies dispelled the idea that sex chromosome aneuploidies were "tantamount to a life of serious handicaps" and revealed their high prevalence in the population.[60] They provided extensive information on the outcomes of trisomy X and other sex chromosome aneuploidies, forming much of the medical literature on the topic to this day. However, the small sample sizes of the long-term follow-ups in particular stymies extrapolation; by 1999, only 16 women in Edinburgh were still being followed.[16] In 2007, Nicole Tartaglia founded the eXtraordinarY Kids Clinic in Denver to study children with sex chromosome aneuploidies; around one-fifth of patients at the clinic had trisomy X as of 2015[update].[13] In 2020, she introduced the eXtraordinarY Babies Study, a planned cohort study on people prenatally diagnosed with sex chromosome aneuploidies.[61]
The first description of trisomy X used the term 'superfemale' to describe the karyotype by analogy to Drosophila flies, a term that was immediately disputed. Curt Stern proposed the use of 'metafemale', which Jacobs criticized as both medically inaccurate and an "illegitimate product of a Graeco-Roman alliance". Bernard Lennon, opposing the use of 'superfemale' as misleading and possessed of an inappropriate "emotional element", suggested 'XXX syndrome'.[62][63] For some years, the disorder was predominantly known as 'triple X syndrome' or 'triple X', though the latter is now discouraged.[16]
Society and culture
Awareness and diagnosis of sex chromosome aneuploidies is increasing.[64] In the late 2010s, several state governments across the United States declared May to be National X & Y Chromosome Variation Awareness Month.[65]
Descriptions of trisomy X overwhelmingly consider the karyotype from a medical perspective, rather than a sociological or educational one.[47] One topic in the sociological discussion of trisomy X and other sex chromosome aneuploidies is disability-selective abortion. Fetuses with sex chromosome aneuploidies are more likely to be aborted, though fetuses with trisomy X are less likely than for such conditions as a whole. A literature review of 19 studies found that nearly one-third of pregnancies with a child with trisomy X were aborted; it also found that parents who were counselled by a genetic counseller with expertise in sex chromosome aneuploidies, rather than an obstetrician or gynecologist, were less likely to abort.[66] Abortion rates in sex chromosome aneuploidies have decreased over time with improved counselling.[67][68]
Other animals
Trisomy X has been observed in other species that use the XY sex-determination system. Six cases of trisomy X have been recorded in dogs, for which the karyotype is 79,XXX compared to 78,XX for an euploid female dog.[69] Unlike in humans, trisomy X in dogs is strongly linked to infertility, either primary anestrus or infertility with an otherwise normal estrous cycle. Canine trisomy X is thought to be underascertained, as most pet dogs are desexed and so underlying infertility will not be discovered.[70] Three of the six known cases of canine trisomy X demonstrated behavioural issues such as fearfulness, inciting speculation about a link between the karyotype and psychological concerns as seen in humans with the condition. An additional dog with normal fertility and no reported behavioural issues was found to have a mosaic 78,XX/79,XXX karyotype. The canine X chromosome has a particularly large pseudoautosomal region, and dogs accordingly have a lower rate of monosomy X than observed in other species; however, a large pseudoautosomal region is not considered a contraindication for trisomy X, and canine trisomy X may have a comparable prevalence to the human form.[69]
Trisomy X is also observed in cattle, where it corresponds to a 61,XXX karyotype. A survey of 71 heifers who failed to become pregnant after two breeding seasons found two cases of trisomy X.[71] As of 2021 a total of eight heifers with Trisomy X have been identified, seven of them were infertile. The condition also affect the river buffalo where the three known cases were sterile.[72]
Notes
- ↑ As comparison, the average adult height for women in the Anglosphere is around 162 cm (5 ft 4 in).[9][10]
- ↑ Male phenotypes, innate or induced, with forms of X chromosome polysomy that are usually phenotypically female do occur. For trisomy X, a trans man and several men with sex reversal have been recorded.[51][52][53]
References
- ↑ 1.00 1.01 1.02 1.03 1.04 1.05 1.06 1.07 1.08 1.09 1.10 "triple X syndrome". GHR. June 2014. Archived from the original on 27 July 2016. Retrieved 26 September 2016.
- ↑ 2.0 2.1 2.2 2.3 2.4 2.5 Tartaglia, NR; Howell, S; Sutherland, A; Wilson, R; Wilson, L (11 May 2010). "A review of trisomy X (47,XXX)". Orphanet Journal of Rare Diseases. 5: 8. doi:10.1186/1750-1172-5-8. PMC 2883963. PMID 20459843.
- ↑ 3.0 3.1 3.2 3.3 3.4 "47 XXX syndrome". GARD. 16 March 2016. Archived from the original on 5 November 2016. Retrieved 26 September 2016.
- ↑ Wright, David (2011). Downs: The history of a disability. OUP Oxford. p. 159. ISBN 9780191619786. Archived from the original on 2017-09-11.
- ↑ 5.00 5.01 5.02 5.03 5.04 5.05 5.06 5.07 5.08 5.09 5.10 5.11 5.12 5.13 5.14 5.15 5.16 5.17 5.18 5.19 5.20 5.21 5.22 5.23 5.24 5.25 Tartaglia NR, Howell S, Sutherland A, Wilson R, Wilson L (11 May 2010). "A review of trisomy X (47,XXX)". Orphanet Journal of Rare Diseases. 5 (8): 8. doi:10.1186/1750-1172-5-8. PMC 2883963. PMID 20459843.
- ↑ Wigby K, D'Epagnier C, Howell S, Reicks A, Wilson R, Cordeiro L, Tartaglia N (November 2016). "Expanding the phenotype of Triple X syndrome: A comparison of prenatal versus postnatal diagnosis". American Journal of Medical Genetics Part A. 170 (11): 2870–2881. doi:10.1002/ajmg.a.37688. PMC 6501572. PMID 27644018.
- ↑ 7.00 7.01 7.02 7.03 7.04 7.05 7.06 7.07 7.08 7.09 7.10 7.11 7.12 7.13 7.14 Otter M, Schrander-Stumpel CTRM, Curfs LMG (1 July 2009). "Triple X syndrome: a review of the literature". European Journal of Human Genetics. 18 (3): 265–271. doi:10.1038/ejhg.2009.109. PMC 2987225. PMID 19568271.
- ↑ Jones, Kenneth Lyons; Jones, Marilyn Crandall; del Campo, Miguel (2013). "Chromosomal Abnormality Syndromes Identifiable on Routine Karyotype". Smith's Recognizable Patterns of Human Malformation (7 ed.). Amsterdam: Elsevier. p. 74. ISBN 978-0323638821.
- ↑ US Dept. of Health and Human Services; et al. (August 2016). "Anthropometric reference data for children and adults: United States, 2011–2014" (PDF). National Health Statistics Reports. 11. Archived (PDF) from the original on 2 February 2017. Retrieved 23 March 2021.
- ↑ "Australian health survey: first results". Australian Bureau of Statistics. 29 October 2012. Archived from the original on 20 January 2017. Retrieved 23 March 2021.
- ↑ 11.0 11.1 11.2 NORD, Samango-Sprouse C (2020). "Trisomy X". National Organization for Rare Diseases. Archived from the original on 23 April 2021. Retrieved 10 May 2021.
- ↑ Wilson, Rebecca; Bennett, Elizabeth; Howell, Susan E; Tartaglia, Nicole (2012). "Sex Chromosome Aneuploidies". Psychopathology of Childhood and Adolescence: A Neuropsychological Approach. New York: Springer Publishing. pp. 594–596. ISBN 978-0826109200.
- ↑ 13.0 13.1 13.2 Tartaglia N, Howell S, Wilson R, Janusz J, Boada R, Martin S, Frazier JB, Pfeiffer M, Regan K, McSwegin S, Zeitler P (17 July 2015). "The eXtraordinarY Kids Clinic: an interdisciplinary model of care for children and adolescents with sex chromosome aneuploidy". Journal of Multidisciplinary Healthcare. 8 (1): 323–334. doi:10.2147/JMDH.S80242. PMC 4514383. PMID 26229481.
- ↑ 14.0 14.1 Liu K, Kurien BT, Zimmerman SL, Kaufman KM, Taft DH, Kottyan LC, Lazaro S, Weaver CA, Ice JA, Adler AJ, Chodosh J, Radfar L, Rasmussen A, Stone DU, Lewis DM, Li S, Koelsch KA, Igoe A, Talsania M, Kumar J, Maier-Moore JS, Harris VM, Gopalakrishnan R, Jonsson R, Lessard JA, Lu X, Gottenberg JE, Anaya JM, Cunninghame-Graham DS, Huang AJW, Brennan MT, Hughes P, Xie G, Ng WF, Nordmark G, Eriksson P, Omdal R, Rhodus NL, Rischmueller M, Rohrer M, Segal BM, Vyse TJ, Wahren-Herlenius M, Witte T, Pons-Estel B, Alarcón-Riquelme M, Guthridge JM, James JA, Lessard CJ, Kelly JA, Thompson SD, Gaffney PM, Montgomery CG, Edberg JC, Kimberly RP, Alarcón GS, Langefeld CL, Gilkeson GS, Kamen DL, Tsao BP, McCune WJ, Salmon JE, Merrill JT, Weisman MH, Wallace DJ, Utset TO, Bottinger EP, Amos CI, Siminovitch KA, Mariette X, Sivils KL, Harley JB, Scofield RH (29 December 2015). "X chromosome dose and sex bias in autoimmune diseases: increased prevalence of 47,XXX in systemic lupus erythematosus and Sjögren's syndrome". Arthritis & Rheumatology. 68 (5): 1290–1300. doi:10.1002/art.39560. PMC 5019501. PMID 26713507.
- ↑ Milunsky JM (2016). "Prenatal Diagnosis of Sex Chromosome Abnormalities". In Milunksy A, Milunsnky JM (eds.). Genetic Disorders and the Fetus: Diagnosis, Prevention, and Treatment (7 ed.). Hoboken: John Wiley and Sons. Section "49,XXXXX".
- ↑ 16.0 16.1 16.2 16.3 16.4 16.5 16.6 16.7 16.8 Unique, Hultén M, Scerif G (2021). "Triple X syndrome, also called Trisomy X" (PDF). Unique. Archived (PDF) from the original on 20 March 2021. Retrieved 10 May 2021.
- ↑ 17.0 17.1 17.2 Tuke MA, Ruth KS, Wood AR, Beaumont RN, Tyrrell J, Jones SE, Yaghootkar H, Turner CLS, Donohoe ME, Brooke AM, Collinson MN, Freathy RM, Weedon MN, Frayling TM, Murray A (2019). "Mosaic Turner syndrome shows reduced penetrance in an adult population study". Genetics in Medicine. 21 (4): 877–886. doi:10.1038/s41436-018-0271-6. PMC 6752315. PMID 30181606.
- ↑ 18.0 18.1 Cordts EB, Christofolini DM, dos Santos AA, Bianco B, Barbosa CP (2011). "Genetic aspects of premature ovarian failure: a literature review". Archives of Gynecology and Obstetrics. 283 (3): 635–643. doi:10.1007/s00404-010-1815-4. PMID 21188402. S2CID 10472263.
- ↑ 19.0 19.1 19.2 19.3 19.4 19.5 19.6 Leggett V, Jacobs P, Nation K, Scerif G, Bishop DVM (February 2010). "Neurocognitive outcomes of individuals with a sex chromosome trisomy: XXX, XYY, or XXY: a systematic review". Developmental Medicine and Child Neurology. 52 (2): 119–129. doi:10.1111/j.1469-8749.2009.03545.x. PMC 2820350. PMID 20059514.
- ↑ Maulik PK, Mascarenhas MN, Mathers CD, Dua T, Saxena S (April 2011). "Prevalence of intellectual disability: a meta-analysis of population-based studies". Research in Developmental Disabilities. 32 (2): 419–436. doi:10.1016/j.ridd.2010.12.018. PMID 21236634.
- ↑ 21.0 21.1 Kliegman, Robert M; St. Geme, Joseph (2019). "Hypofunction of the Ovaries". Nelson Textbook of Pediatrics (21 ed.). Amsterdam: Elsevier. pp. 3005–3006. ISBN 978-0323529501.
- ↑ Law J, Boyle J, Harris F, Harkness A, Nye C (2000). "Prevalence and natural history of primary speech and language delay: findings from a systematic review of the literature". International Journal of Language & Communication Disorders. 32 (5): 165–188. doi:10.1080/136828200247133. PMID 10912250. S2CID 21499682.
- ↑ Black LI, Vahratian A, Hoffman HJ (June 2015). "Communication disorders and use of intervention services among children aged 3–17 years: United States, 2012". National Center for Health Statistics Data Briefs. Archived from the original on 28 June 2021. Retrieved 28 June 2021.
- ↑ 24.0 24.1 24.2 24.3 van Rijn S (15 July 2019). "A review of neurocognitive functioning and risk for psychopathology in sex chromosome trisomy (47,XXY, 47,XXX, 47,XYY)". Current Opinion in Psychiatry. 32 (2): 79–84. doi:10.1097/YCO.0000000000000471. PMC 6687415. PMID 30689602.
- ↑ 25.0 25.1 AXYS, Berry Kravis E (December 2020). "Seizures and tremor in people with X & Y chromosome variations" (PDF). AXYS: Association for X and Y Chromosome Variations. Archived (PDF) from the original on 18 April 2021. Retrieved 11 May 2021.
- ↑ Maenner MJ, Shaw KA, Baio J, Washington A, Patrick M, DiRienzo M, Christensen DL, Wiggins LD, Pettygrove S, Andrews JG, Lopez M, Hudson A, Baroud T, Schwenk Y, White T, Rosenberg CR, Lee LC, Harrington RA, Huston M, Hewitt A, Esler A, Hall-Lande J, Poynter JN, Hallas-Muchow L, Constantino JN, Fitzgerald RT, Zahorodny W, Shenouda J, Daniels JL, Warren Z, Vehorn A, Salinas A, Durkin MS, Dietz PM (27 March 2020). "Prevalence of autism spectrum disorder among children aged 8 years — Autism and Developmental Disabilities Monitoring Network, 11 sites, United States". MMWR. Surveillance Summaries. 69 (4): 1–12. doi:10.15585/mmwr.ss6904a1. PMC 7119644. PMID 32214087.
- ↑ Otter M, Crins PML, Campforts BCM, Stumpel CTRM, van Amelsvoort T, Vingerhoets C (15 February 2021). "Social functioning and emotion recognition in adults with triple X syndrome". BJPsych Open. 7 (2): e51. doi:10.1192/bjo.2021.8. PMC 8058878. PMID 33583482.
- ↑ 28.0 28.1 28.2 28.3 28.4 Milunsky JM (2016). "Prenatal Diagnosis of Sex Chromosome Abnormalities". In Milunksy A, Milunsnky JM (eds.). Genetic Disorders and the Fetus: Diagnosis, Prevention, and Treatment (7 ed.). Hoboken: John Wiley and Sons. Triple X and poly-X syndromes.
- ↑ DeLisi LE, Friedrich U, Wahlstrom J, Boccio-Smith A, Forsman A, Eklund K, Crow TJ (1994). "Schizophrenia and sex chromosome anomalies". Schizophrenia Bulletin. 20 (3): 495–505. doi:10.1093/schbul/20.3.495. PMID 7973466.
- ↑ Eckstrand K, Addington AM, Stromberg T, Merriman B, Miller R, Gochman P, Long R, Dutra A, Chen Z, Meltzer P, Nelson SF, Rapaport JL (18 September 2008). "Sex chromosome anomalies in childhood onset schizophrenia: an update". Molecular Psychiatry. 13 (10): 910–911. doi:10.1038/mp.2008.67. PMC 4316819. PMID 18800051.
- ↑ Neri G, Opitz JM (1984). "A possible explanation for the low incidence of gonosomal aneuploidy among the offspring of triplo-X individuals". American Journal of Medical Genetics. 18 (2): 357–364. doi:10.1002/ajmg.1320180220. PMID 6465205.
- ↑ Lim HH, Kil HR, Koo SH (July 2017). "Incidence, puberty, and fertility in 45,X/47,XXX mosaicism: Report of a patient and a literature review". American Journal of Medical Genetics Part A. 173 (7): 1961–1964. doi:10.1002/ajmg.a.38276. PMID 28485514. S2CID 32472061.
- ↑ 33.0 33.1 33.2 Tang R, Lin L, Guo Z, Hou H, Yu Q (July 2019). "Ovarian reserve evaluation in a woman with 45,X/47,XXX mosaicism: A case report and a review of literature". Molecular Genetics & Genomic Medicine. 7 (7): e00732. doi:10.1002/mgg3.732. PMC 6625135. PMID 31070017.
- ↑ Sybert VP (March 2002). "Phenotypic effects of mosaicism for a 47,XXX cell line in Turner syndrome". Journal of Medical Genetics. 39 (3): 217–220. doi:10.1136/jmg.39.3.217. PMC 1735059. PMID 11897829.
- ↑ Blair J, Tolmie J, Hollman AS, Donaldson MDC (November 2001). "Phenotype, ovarian function, and growth in patients with 45,X/47,XXX Turner mosaicism: Implications for prenatal counseling and estrogen therapy at puberty". Journal of Pediatrics. 139 (5): 724–728. doi:10.1067/mpd.2001.118571. PMID 11713453.
- ↑ Milunsky JM (2016). "Prenatal Diagnosis of Sex Chromosome Abnormalities". In Milunksy A, Milunsnky JM (eds.). Genetic Disorders and the Fetus: Diagnosis, Prevention, and Treatment (7 ed.). Hoboken: John Wiley and Sons. Turner syndrome.
- ↑ May KM, Jacobs PA, Lee M, Ratcliffe S, Robinson A, Nielsen J, Hassold TJ (April 1990). "The parental origin of the extra X chromosome in 47,XXX females". American Journal of Human Genetics. 46 (4): 754–761. PMC 1683670. PMID 2316522.
- ↑ Gottlieb SF, Tupper C, Kerndt CC, Tegay DH (26 September 2020). "Genetics, Nondisjunction". NCBI Bookshelf. PMID 29489267. Archived from the original on 9 April 2022. Retrieved 21 June 2021.
- ↑ Mikwar M, MacFarlane AJ, Marchetti F (4 July 2020). "Mechanisms of oocyte aneuploidy associated with advanced maternal age". Mutation Research/Reviews in Mutation Research. 785: 108320. doi:10.1016/j.mrrev.2020.108320. PMID 32800274.
- ↑ Kuliev A, Verlinsky Y (1 October 2004). "Meiotic and mitotic nondisjunction: lessons from preimplantation genetic diagnosis". Human Reproduction Update. 10 (5): 401–407. doi:10.1093/humupd/dmh036. PMID 15319376.
- ↑ O'Connor C (2008). "Chromosomal abnormalities: aneuploidies". Nature Education. Archived from the original on 3 November 2020. Retrieved 16 May 2021.
- ↑ Edens Hurst AC, Zieve D, Conaway B (2 April 2021). "Karyotyping". MedlinePlus. Archived from the original on 7 November 2020. Retrieved 16 May 2021.
- ↑ 43.0 43.1 Meazza C, Gertosio C, Giacchero R, Pagani S, Bozzola M (3 August 2017). "Tall stature: a difficult diagnosis?". Italian Journal of Pediatrics. 43 (1): 66. doi:10.1186/s13052-017-0385-5. PMC 5543750. PMID 28774346.
- ↑ Unique, Rooman R, Hultén M (2005). "Tetrasomy X" (PDF). Unique. Archived (PDF) from the original on 18 March 2021. Retrieved 16 May 2021.
- ↑ Otter, M; Schrander-Stumpel, CT; Curfs, LM (March 2010). "Triple X syndrome: a review of the literature". European Journal of Human Genetics. 18 (3): 265–71. doi:10.1038/ejhg.2009.109. PMC 2987225. PMID 19568271.
- ↑ 46.0 46.1 46.2 Isaacs Cover, Virginia (2012). "Trisomy X, Tetrasomy X and Pentasomy X". Living with Klinefelter Syndrome (47,XXY) Trisomy X (47, XXX) and 47, XYY: A Guide for Families and Individuals Affected by Extra X and Y Chromosome Variations. Altona, Manitoba: Friesens. pp. 107–114. ISBN 978-0615574004.
- ↑ 47.0 47.1 Attfield K (25 May 2020). "Triple X supergirls: Their special educational needs and social experience". International Journal of Educational Research. 102 (1): 101588. doi:10.1016/j.ijer.2020.101588. S2CID 219811098.
- ↑ 48.0 48.1 48.2 Berglund A, Stochholm K, Gravholt CH (2020). "The epidemiology of sex chromosome abnormalities". American Journal of Medical Genetics Part C: Seminars in Medical Genetics. 184 (2): 202–215. doi:10.1002/ajmg.c.31805. PMID 32506765. S2CID 219537282.
- ↑ 49.0 49.1 Berglund A, Viuff MN, Skakkebæk A, Chang S, Stochholm K, Gravholt CH (2019). "Changes in the cohort composition of Turner syndrome and severe non-diagnosis of Klinefelter, 47,XXX and 47,XYY syndrome: a nationwide cohort study". Orphanet Journal of Rare Diseases. 14 (1): 16. doi:10.1186/s13023-018-0976-2. PMC 6332849. PMID 30642344.
- ↑ Stochholm K, Juul S, Gravholt CH (2010). "Mortality and incidence in women with 47,XXX and variants". American Journal of Medical Genetics Part A. 152A (2): 367–372. doi:10.1002/ajmg.a.33214. PMID 20101696. S2CID 12004487.
- ↑ Turan MT, Eşel E, Dündar M, Candemir Z, Baştürk M, Sofuoğlu S, Özkul Y (1 December 2000). "Female-to-male transsexual with 47,XXX karyotype". Biological Psychiatry. 48 (1): 1116–1117. doi:10.1016/S0006-3223(00)00954-9. PMID 11094147. S2CID 16396520.
- ↑ Ogata T, Matsuo M, Muroya K, Koyama Y, Fukutani K (1 February 2001). "47,XXX male: A clinical and molecular study". American Journal of Medical Genetics. 98 (4): 353–356. doi:10.1002/1096-8628(20010201)98:4<353::AID-AJMG1110>3.0.CO;2-D. PMID 11170081.
- ↑ Müller U, Latt SA, Donlon T, Opitz JM (October 1987). "Y‐specific DNA sequences in male patients with 46,XX and 47,XXX karyotypes". American Journal of Medical Genetics. 28 (2): 393–401. doi:10.1002/ajmg.1320280218. PMID 2827475.
- ↑ Jacobs PA, Baikie AG, Court Brown WM, MacGregor TN, Harnden DG (26 September 1959). "Evidence for the existence of the human 'super female'". Lancet. 274 (7100): 423–425. doi:10.1016/S0140-6736(59)90415-5. PMID 14406377.
- ↑ Ford CE, Jones KW, Polani PE, de Almeida JCC, Briggs JH (1959). "A sex-chromosome anomaly in a case of gonadal dysgenesis (Turner's syndrome)". Lancet. 273 (7075): 711–713. doi:10.1016/S0140-6736(59)91893-8. PMID 13642858.
- ↑ Jacobs PA, Strong JA (31 January 1959). "A case of human intersexuality having a possible XXY sex-determining mechanism". Nature. 183 (4657): 302–303. Bibcode:1959Natur.183..302J. doi:10.1038/183302a0. PMID 13632697. S2CID 38349997.
- ↑ Barr ML, Sergovich FR, Carr DH, Saver EL (6 September 1969). "The triplo-X female: an appraisal based on a study of 12 cases and a review of the literature". Canadian Medical Association Journal. 101 (5): 247–258. PMC 1946229. PMID 5812107.
- ↑ Attfield K (25 January 2021). "Triple X superwomen: their post-compulsory education and employability". Journal of Education and Work. 34 (1): 81–94. doi:10.1080/13639080.2021.1875126. S2CID 231990866.
- ↑ 59.0 59.1 Ratcliffe S (1999). "Long term outcome in children of sex chromosome abnormalities". Archives of Disease in Childhood. 80 (2): 192–195. doi:10.1136/adc.80.2.192. PMC 1717826. PMID 10325742.
- ↑ Cohen FL, Durham JD (March 1985). "Sex chromosome variations in school-age children". Journal of School Health. 55 (3): 99–102. doi:10.1111/j.1746-1561.1985.tb04089.x. PMID 3845264.
- ↑ Tartaglia N, Howell S, Davis S, Kowal K, Tanda T, Brown M, Boada C, Alston A, Crawford L, Thompson T, van Rijn S, Wilson R, Janusz J, Ross J (June 2020). "Early neurodevelopmental and medical profile in children with sex chromosome trisomies: Background for the prospective eXtraordinarY Babies Study to identify early risk factors and targets for intervention". American Journal of Medical Genetics Part C: Seminars in Medical Genetics. 184 (2): 428–443. doi:10.1002/ajmg.c.31807. PMC 7413625. PMID 32506668.
- ↑ Jacobs PA, Baikie AG, Court Brown WM, Harnden DG, MacGregor TN, MacLean N (19 December 1959). "Use of the term "superfemale"". The Lancet. 274 (7112): 1145. doi:10.1016/S0140-6736(59)90132-1.
- ↑ Lennox B (2 January 1960). "Use of the term "superfemale"". The Lancet. 275 (7114): 55. doi:10.1016/S0140-6736(60)92744-6.
- ↑ Milunsky JM (2016). "Prenatal Diagnosis of Sex Chromosome Abnormalities". In Milunksy A, Milunsnky JM (eds.). Genetic Disorders and the Fetus: Diagnosis, Prevention, and Treatment (7 ed.). Hoboken: John Wiley and Sons. Chapter introduction.
- ↑ "National X & Y Chromosome Variation Awareness Month". AXYS: Association for X and Y Chromosome Variations. Archived from the original on 23 May 2021. Retrieved 23 May 2021.
- ↑ Jeon KC, Chen LS, Goodson P (7 October 2011). "Decision to abort after a prenatal diagnosis of sex chromosome abnormality: a systematic review of the literature". Genetics in Medicine. 14 (1): 27–38. doi:10.1038/gim.0b013e31822e57a7. PMID 22237429.
- ↑ Linden MG, Bender BG, Robinson A (March 1996). "Intrauterine diagnosis of sex chromosome aneuploidy". Obstetrics & Gynecology. 87 (3): 468–475. doi:10.1016/0029-7844(95)00419-x. PMID 8598978. S2CID 32257832.
- ↑ Gruchy N, Blondeel E, Le Meur N, Joly-Hélas G, Chambon P, Till M, Herbeaux M, Vigouroux-Castera A, Coussement A, Lespinasse J, Amblard F, Jiminez Pocquet M, Lebel-Roy C, Carré-Pigeon F, Flori E, Mugneret F, Jaillard S, Yardin C, Harbuz R, Collonge-Rame MA, Vago P, Valduga M, Leporrier N, Vialard F (28 March 2016). "Pregnancy outcomes in prenatally diagnosed 47, XXX and 47, XYY syndromes: a 30-year French, retrospective, multicentre study". Prenatal Diagnosis. 36 (6): 523–529. doi:10.1002/pd.4817. PMID 27018091. S2CID 29814110.
- ↑ 69.0 69.1 Szczerbal I, Switonski M (27 March 2021). "Clinical cytogenetics of the dog: a review". Animals. 11 (4): 947. doi:10.3390/ani11040947. PMC 8066086. PMID 33801756.
- ↑ O'Connor CL, Schweizer C, Gradil C, Schlafer D, Lopate C, Prociuk U, Meyers-Wallen VN, Casal ML (15 July 2011). "Trisomy-X with estrous cycle anomalies in two female dogs". Theriogenology. 76 (2): 374–380. doi:10.1016/j.theriogenology.2011.02.017. PMC 3115384. PMID 21550105.
- ↑ Swartz HA, Vogt DW (September 1983). "Chromosome abnormalities as a cause of reproductive inefficiency in heifers". Journal of Heredity. 74 (5): 320–324. doi:10.1093/oxfordjournals.jhered.a109802.
- ↑ Iannuzzi A, Parma P, Iannuzzi L (12 March 2021). "Chromosome Abnormalities and Fertility in Domestic Bovids: A Review". Animals. 11 (3): 802. doi:10.3390/ani11030802. hdl:2434/822898. PMC 8001068. PMID 33809390.
External links
| Classification |
|---|
- NLM (2008). Triple X syndrome Archived 2007-03-12 at the Wayback Machine Genetics Home Reference
![Epicanthal folds and increased distance between the eyes in 2-year-old girl with trisomy X[2]](https://upload.wikimedia.org/wikipedia/commons/thumb/c/cb/XXXSyndromeA.png/250px-XXXSyndromeA.png)