Radequinil
Jump to navigation
Jump to search
 | |
| Clinical data | |
|---|---|
| ATC code |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
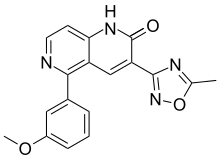
| Formula | C18H14N4O3 |
| Molar mass | 334.335 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Radequinil (INN; AC-3933) is a cognitive enhancer which acts as a partial inverse agonist of the benzodiazepine site of the GABAA receptor.[1] It was under development by Dainippon Sumitomo Pharma for the treatment of Alzheimer's disease and made it to phase II clinical trials but development seems to have been halted and it was never marketed.[1][2]
See also
References
- ^ a b Martocchia A, Falaschi P (2008). "Current Strategies of Therapy in Alzheimer's Disease" (PDF). The Open Neuropsychopharmacology Journal. 1: 19–23. doi:10.2174/1876523800801010019.
- ^ Pogacić V, Herrling P (2007). "List of drugs in development for neurodegenerative diseases. Update June 2007". Neuro-Degenerative Diseases. 4 (6): 443–86. doi:10.1159/000107705. PMID 17934328.
Categories:
- Articles with short description
- Short description matches Wikidata
- Drugs not assigned an ATC code
- Articles without EBI source
- Chemical pages without DrugBank identifier
- Articles without KEGG source
- Drugs with no legal status
- Articles containing unverified chemical infoboxes
- Nootropics
- Oxadiazoles
- Lactams
- GABAA receptor negative allosteric modulators
- Phenol ethers
- Naphthyridines
- All stub articles
- Nervous system drug stubs