Methemoglobinemia
| Methemoglobinemia | |
|---|---|
| Other names: Hemoglobin M disease[1] | |
 | |
| Chocolate-brown blood due to methemoglobinemia | |
| Specialty | Toxicology |
| Symptoms | Headache, dizziness, shortness of breath, nausea, poor muscle coordination, blue-colored skin[2] |
| Causes | Benzocaine, nitrates, dapsone, genetics[3] |
| Diagnostic method | Blood gas[3] |
| Differential diagnosis | Argyria, sulfhemoglobinemia, heart failure[3] |
| Treatment | Oxygen therapy, methylene blue[3] |
| Prognosis | Generally good with treatment[3] |
| Frequency | Relatively uncommon[3] |
Methemoglobinemia is a condition of elevated methemoglobin in the blood.[2] Symptoms may include headache, dizziness, shortness of breath, nausea, poor muscle coordination, and blue-colored skin (cyanosis).[2] Complications may include seizures and heart arrhythmias.[3]
Methemoglobinemia can be due to certain medications, chemicals, or food or it can be inherited from a person's parents.[2] Substances involved may include benzocaine, nitrates, or dapsone.[3] The underlying mechanism involves some of the iron in hemoglobin being converted from the ferrous [Fe2+] to the ferric [Fe3+] form.[3] The diagnosis is often suspected based on symptoms and a low blood oxygen that does not improve with oxygen therapy.[3] Diagnosis is confirmed by a blood gas.[3]
Treatment is generally with oxygen therapy and methylene blue.[3] Other treatments may include vitamin C, exchange transfusion, and hyperbaric oxygen therapy.[3] Outcomes are generally good with treatment.[3] Methemoglobinemia is relatively uncommon, with most cases being acquired rather than genetic.[3]
Signs and symptoms
Signs and symptoms of methemoglobinemia (methemoglobin level above 10%) include shortness of breath, cyanosis, mental status changes (~50%), headache, fatigue, exercise intolerance, dizziness, and loss of consciousness.[citation needed]
People with severe methemoglobinemia (methemoglobin level above 50%) may exhibit seizures, coma, and death (level above 70%).[4] Healthy people may not have many symptoms with methemoglobin levels below 15%. However, patients with co-morbidities such as anemia, cardiovascular disease, lung disease, sepsis, or presence of other abnormal hemoglobin species (e.g. carboxyhemoglobin, sulfhemoglobin or sickle hemoglobin) may experience moderate to severe symptoms at much lower levels (as low as 5–8%).[citation needed]
-
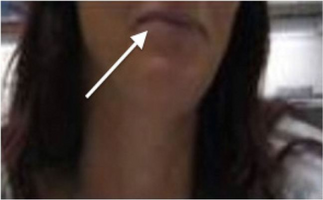
White arrow indicates blue/grey discoloration of lips
-
Chocolate-brown blood due to methemoglobinemia
Cause
Acquired
Methemoglobinemia may be acquired.[5] Classical drug causes of methemoglobinaemia include various antibiotics (trimethoprim, sulfonamides, and dapsone[6]), local anesthetics (especially articaine, benzocaine, prilocaine,[7] and lidocaine[8]), and aniline dyes, metoclopramide, rasburicase, chlorates, and bromates. Nitrates are suspected to cause methemoglobinemia.[9]
In otherwise healthy individuals, the protective enzyme systems normally present in red blood cells rapidly reduce the methemoglobin back to hemoglobin and hence maintain methemoglobin levels at less than one percent of the total hemoglobin concentration. Exposure to exogenous oxidizing drugs and their metabolites (such as benzocaine, dapsone, and nitrates) may lead to an increase of up to a thousandfold of the methemoglobin formation rate, overwhelming the protective enzyme systems and acutely increasing methemoglobin levels.[citation needed]
Infants under 6 months of age have lower levels of a key methemoglobin reduction enzyme (NADH-cytochrome b5 reductase) in their red blood cells. This results in a major risk of methemoglobinemia caused by nitrates ingested in drinking water,[10] dehydration (usually caused by gastroenteritis with diarrhea), sepsis, or topical anesthetics containing benzocaine or prilocaine. Nitrates used in agricultural fertilizers may leak into the ground and may contaminate well water. The current EPA standard of 10 ppm nitrate-nitrogen for drinking water is specifically set to protect infants.[10] Benzocaine applied to the gums or throat (as commonly used in baby teething gels, or sore throat lozenges) can cause methemoglobinemia.[11][12]
Genetic

Due to a deficiency of the enzyme diaphorase I (NADH-cytochrome b5 reductase), methemoglobin levels rise and the blood of met-Hb patients has reduced oxygen-carrying capacity. Instead of being red in color, the arterial blood of met-Hb patients is brown. This results in the skin of Caucasian patients gaining a bluish hue. Hereditary met-Hb is caused by a recessive gene. If only one parent has this gene, offspring will have normal-hued skin, but if both parents carry the gene, there is a chance the offspring will have blue-hued skin.[citation needed]
Another cause of congenital methemoglobinemia is seen in patients with abnormal hemoglobin variants such as hemoglobin M (HbM), or hemoglobin H (HbH), which are not amenable to reduction despite intact enzyme systems.[citation needed]
Methemoglobinemia can also arise in patients with pyruvate kinase deficiency due to impaired production of NADH – the essential cofactor for diaphorase I. Similarly, patients with glucose-6-phosphate dehydrogenase deficiency may have impaired production of another co-factor, NADPH.[citation needed]
Pathophysiology
The affinity for oxygen of ferric iron is impaired. The binding of oxygen to methemoglobin results in an increased affinity for oxygen in the remaining heme sites that are in ferrous state within the same tetrameric hemoglobin unit.[13] This leads to an overall reduced ability of the red blood cell to release oxygen to tissues, with the associated oxygen–hemoglobin dissociation curve therefore shifted to the left. When methemoglobin concentration is elevated in red blood cells, tissue hypoxia may occur.[14]
Normally, methemoglobin levels are <1%, as measured by the CO-oximetry test. Elevated levels of methemoglobin in the blood are caused when the mechanisms that defend against oxidative stress within the red blood cell are overwhelmed and the oxygen carrying ferrous ion (Fe2+) of the heme group of the hemoglobin molecule is oxidized to the ferric state (Fe3+). This converts hemoglobin to methemoglobin, resulting in a reduced ability to release oxygen to tissues and thereby hypoxia. This can give the blood a bluish or chocolate-brown color. Spontaneously formed methemoglobin is normally reduced (regenerating normal hemoglobin) by protective enzyme systems, e.g., NADH methemoglobin reductase (cytochrome-b5 reductase) (major pathway), NADPH methemoglobin reductase (minor pathway) and to a lesser extent the ascorbic acid and glutathione enzyme systems. Disruptions with these enzyme systems lead to methemoglobinemia.Hypoxia occurs due to the decreased oxygen-binding capacity of methemoglobin, as well as the increased oxygen-binding affinity of other subunits in the same hemoglobin molecule, which prevents them from releasing oxygen at normal tissue oxygen levels.[citation needed]
Diagnosis

Arterial blood with elevated methemoglobin levels has a characteristic chocolate-brown color as compared to normal bright red oxygen-containing arterial blood.[4] If methemoglobinemia is suspected, an arterial blood gas and CO-oximetry panel should be obtained.
Differential diagnosis
Other conditions that can cause bluish skin include argyria, sulfhemoglobinemia, and heart failure.[3]
Treatment


Methemoglobinemia can be treated with supplemental oxygen and methylene blue.[15] Methylene blue is given as a 1% solution (10 mg/ml) 1 to 2 mg/kg administered intravenously slowly over five minutes. Although the response is usually rapid, the dose may be repeated in one hour if the level of methemoglobin is still high one hour after the initial infusion. Methylene blue inhibits monoamine oxidase, and serotonin toxicity can occur if taken with an SSRI (selective serotonin reuptake inhibitor) medicine.[16]
Methylene blue restores the iron in hemoglobin to its normal (reduced) oxygen-carrying state. This is achieved by providing an artificial electron acceptor (such as methylene blue or flavin) for NADPH methemoglobin reductase (RBCs usually don't have one; the presence of methylene blue allows the enzyme to function at 5× normal levels).[17] The NADPH is generated via the hexose monophosphate shunt.
Genetically induced chronic low-level methemoglobinemia may be treated with oral methylene blue daily. Also, vitamin C can occasionally reduce cyanosis associated with chronic methemoglobinemia, and may be helpful in settings in which methylene blue is unavailable or contraindicated (e.g., in an individual with G6PD deficiency).[18] Diaphorase (cytochrome b5 reductase) normally contributes only a small percentage of the red blood cell's reducing capacity, but can be pharmacologically activated by exogenous cofactors (such as methylene blue) to five times its normal level of activity.
Epidemiology
Methemoglobinemia mostly affects infants under 6 months of age (particularly those under 4 months) due to low hepatic production of methemoglobin reductase.[19][20] The most at-risk populations are those with water sources high in nitrates, such as wells and other water that's not monitored or treated by a water treatment facility. The nitrates can be hazardous to the infants.[21][22] The link between blue baby syndrome in infants and high nitrate levels is well established for waters exceeding the normal limit of 10 mg/L.[23][24] However, there is also evidence that breastfeeding is protective in exposed populations.[25]
Society and culture
Blue Fugates
The Fugates, a family that lived in the hills of Kentucky, had the hereditary form. They are known as the "Blue Fugates".[26] Martin Fugate and Elizabeth Smith, who had married and settled near Hazard, Kentucky in around 1800, were both carriers of the recessive methemoglobinemia (met-H) gene, as was a nearby clan with whom the Fugates descendants intermarried. As a result, many descendants of the Fugates were born with met-H.[27][28][29][30]
Blue Men of Lurgan
The "blue men of Lurgan" were a pair of Lurgan men suffering from what was described as "familial idiopathic methaemoglobinaemia" who were treated by Dr. James Deeny in 1942. Deeny, who would later become the Chief Medical Officer of the Republic of Ireland, prescribed a course of ascorbic acid and sodium bicarbonate. In case one, by the eighth day of treatments there was a marked change in appearance, and by the twelfth day of treatment the patient's complexion was normal. In case two, the patient's complexion reached normality over a month-long duration of treatment.[31]
See also
References
- ↑ "Methemoglobinemia: MedlinePlus Medical Encyclopedia". medlineplus.gov. Archived from the original on 8 June 2019. Retrieved 8 June 2019.
- ↑ 2.0 2.1 2.2 2.3 "NCI Dictionary of Cancer Terms". National Cancer Institute. 2 February 2011. Archived from the original on 21 December 2019. Retrieved 21 December 2019.
- ↑ 3.00 3.01 3.02 3.03 3.04 3.05 3.06 3.07 3.08 3.09 3.10 3.11 3.12 3.13 3.14 3.15 Ludlow, JT; Wilkerson, RG; Nappe, TM (January 2019). "Methemoglobinemia". PMID 30726002.
{{cite journal}}: Cite journal requires|journal=(help) - ↑ 4.0 4.1 "eMedicine - Methemoglobinemia". Archived from the original on 2008-04-11. Retrieved 2008-09-13.
- ↑ Ash-Bernal R, Wise R, Wright SM (2004). "Acquired methemoglobinemia: a retrospective series of 138 cases at 2 teaching hospitals". Medicine (Baltimore). 83 (5): 265–273. doi:10.1097/01.md.0000141096.00377.3f. PMID 15342970. S2CID 40957843.
- ↑ Zosel A, Rychter K, Leikin JB (2007). "Dapsone-induced methemoglobinemia: case report and literature review". Am J Ther. 14 (6): 585–587. doi:10.1097/MJT.0b013e3180a6af55. PMID 18090884.
- ↑ Adams V, Marley J, McCarroll C (2007). "Prilocaine induced methaemoglobinaemia in a medically compromised patient. Was this an inevitable consequence of the dose administered?". Br Dent J. 203 (10): 585–587. doi:10.1038/bdj.2007.1045. PMID 18037845.
- ↑ Barash M, Reich KA, Rademaker D (2015). "Lidocaine-induced methemoglobinemia: a clinical reminder". J Am Osteopath Assoc. 115 (2): 94–8. doi:10.7556/jaoa.2015.020. PMID 25637615.
- ↑ Powlson, David S.; Addiscott, Tom M.; Benjamin, Nigel; Cassman, Ken G.; De Kok, Theo M.; Van Grinsven, Hans; l'Hirondel, Jean-Louis; Avery, Alex A.; Van Kessel, Chris (2008). "When Does Nitrate Become a Risk for Humans?". Journal of Environmental Quality. 37 (2): 291–5. doi:10.2134/jeq2007.0177. PMID 18268290. Archived from the original on 2018-07-22. Retrieved 2019-12-21.
- ↑ 10.0 10.1 "Basic Information about Nitrate in Drinking Water". United State Environmental Protection Agency. Archived from the original on 17 May 2013. Retrieved 10 May 2013.
- ↑ "FDA Drug Safety Communication: Reports of a rare, but serious and potentially fatal adverse effect with the use of over-the-counter (OTC) benzocaine gels and liquids applied to the gums or mouth". U.S. Food and Drug Administration. 7 April 2011. Archived from the original on 19 April 2012. Retrieved 10 May 2013.
- ↑ "Risk of serious and potentially fatal blood disorder prompts FDA action on oral over-the-counter benzocaine products used for teething and mouth pain and prescription local anesthetics". U.S. FDA. May 23, 2018. Archived from the original on May 25, 2018. Retrieved May 24, 2018.
- ↑ Darling, AU; Roughton, R (1942). "The effect of methemoglobin on the equilibrium between oxygen and hemoglobin". Am J Physiol. 137: 56. doi:10.1152/ajplegacy.1942.137.1.56.
- ↑ Umbreit, Jay (2007). "Methemoglobin—It's not just blue: A concise review". American Journal of Hematology. 82 (2): 134–144. doi:10.1002/ajh.20738. PMID 16986127. S2CID 29107446.
- ↑ Yusim Y, Livingstone D, Sidi A (2007). "Blue dyes, blue people: the systemic effects of blue dyes when administered via different routes". J Clin Anesth. 19 (4): 315–321. doi:10.1016/j.jclinane.2007.01.006. PMID 17572332.
- ↑ Gillman PK (2006). "Methylene blue implicated in potentially fatal serotonin toxicity". Anaesthesia. 61 (10): 1013–4. doi:10.1111/j.1365-2044.2006.04808.x. PMID 16978328.
- ↑ Yubisui T, Takeshita M, Yoneyama Y (Jun 1980). "Reduction of methemoglobin through flavin at the physiological concentration by NADPH-flavin reductase of human erythrocytes". J Biochem. 87 (6): 1715–20. doi:10.1093/oxfordjournals.jbchem.a132915. PMID 7400118.
- ↑ "Archive copy". Archived from the original on 2019-05-14. Retrieved 2019-05-14.
{{cite web}}: CS1 maint: archived copy as title (link) - ↑ Richard, Alyce M.; Diaz, James H.; Kaye, Alan David (1 January 2014). "Reexamining the Risks of Drinking-Water Nitrates on Public Health". The Ochsner Journal. 14 (3): 392–398. PMC 4171798. PMID 25249806.
{{cite journal}}: CS1 maint: url-status (link) - ↑ "Nitrates and drinking water". www.bfhd.wa.gov. Archived from the original on 28 January 2018. Retrieved 10 December 2016.
- ↑ Manassaram, Deana M.; Backer, Lorraine C.; Moll, Deborah M. (1 March 2007). "A review of nitrates in drinking water: maternal exposure and adverse reproductive and developmental outcomes". Ciência & Saúde Coletiva. 12 (3): 153–163. doi:10.1590/S1413-81232007000100018. PMID 17680066.
- ↑ Fan, Anna M; Steinberg, Valerie E (May 27, 1995). "Health implications of nitrate and nitrite in drinking water: An update on methemoglobinemia occurrence and reproductive and developmental toxicity". Regulatory Toxicology and Pharmacology. 23 (1 Pt 1): 35–43. doi:10.1006/rtph.1996.0006. PMID 8628918.
- ↑ "Nitrate and Nitrite in Drinking-Water" (PDF). www.who.int. WHO Press. 2011. Archived (PDF) from the original on August 2, 2018. Retrieved December 10, 2016.
- ↑ EPA, OW, US. "Table of Regulated Drinking Water Contaminants". www.epa.gov. Archived from the original on 2017-03-01. Retrieved 2016-12-12.
- ↑ Pollock, J (1994). "Long term associations with infant feeding in a clinically advantaged population of babies". Developmental Medicine & Child Neurology. 36 (5): 429–440. doi:10.1111/j.1469-8749.1994.tb11869.x. PMID 8168662.
- ↑ "Blue-skinned family baffled science for 150 years". MSN. 24 February 2012. Archived from the original on 22 January 2013. Retrieved 10 May 2013.
- ↑ Straight Dope article on the Fugates of Appalachia, an extended family of blue-skinned people Archived 2006-11-27 at the Wayback Machine
- ↑ Tri City Herald, November 7, 1974 p.32 Newspaper reports on the Blue Fugates[permanent dead link]
- ↑ "Fugates of Kentucky: Skin Bluer than Lake Louise". Archived from the original on 2021-03-07. Retrieved 2022-03-14.
- ↑ "Martin Fuqatenin nəsli: genetik problemə görə dünyaya gələn mavi uşaqlar — Mavi Fuqatelər". Archived from the original on 2020-04-27. Retrieved 2018-06-17.
- ↑ Deeny, James (1995). The End of an Epidemic. Dublin: A.& A.Farmar. ISBN 978-1-899047-06-2.
External links
| Classification | |
|---|---|
| External resources |
- Pages with script errors
- CS1 errors: missing periodical
- CS1 maint: archived copy as title
- CS1 maint: url-status
- Webarchive template wayback links
- All articles with dead external links
- Articles with dead external links from March 2022
- Articles with invalid date parameter in template
- Articles with permanently dead external links
- All articles with unsourced statements
- Articles with unsourced statements from July 2020
- Articles with hatnote templates targeting a nonexistent page
- Red blood cell disorders
- Autosomal recessive disorders
- RTT