Benzo(a)pyrene
![Benzo[a]pyrene](http://upload.wikimedia.org/wikipedia/commons/thumb/f/fa/Benzo-a-pyrene.svg/220px-Benzo-a-pyrene.svg.png) | |
 | |
 | |
| Names | |
|---|---|
| Preferred IUPAC name
Benzo[pqr]tetraphene[1] | |
Other names
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.000.026 |
| EC Number |
|
| KEGG | |
PubChem CID
|
|
| RTECS number |
|
| UNII | |
| UN number | 3077, 3082 |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C20H12 | |
| Molar mass | 252.316 g·mol−1 |
| Density | 1.24 g/cm3 (25 °C) |
| Melting point | 179[2] °C (354 °F; 452 K) |
| Boiling point | 495 °C (923 °F; 768 K) |
| 0.2 to 6.2 μg/L | |
| -135.7·10−6 cm3/mol | |
| Hazards[3] | |
| GHS labelling: | |
  
| |
| Danger | |
| H317, H340, H350, H360, H410 | |
| P201, P202, P261, P272, P273, P280, P281, P302+P352, P308+P313, P321, P333+P313, P363, P391, P405, P501 | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Benzo[a]pyrene (BaP or B[a]P) is a polycyclic aromatic hydrocarbon and the result of incomplete combustion of organic matter at temperatures between 300 °C (572 °F) and 600 °C (1,112 °F). The ubiquitous compound can be found in coal tar, tobacco smoke and many foods, especially grilled meats. The substance with the formula C20H12 is one of the benzopyrenes, formed by a benzene ring fused to pyrene. Its diol epoxide metabolites, more commonly known as BPDE, react with and bind to DNA, resulting in mutations and eventually cancer. It is listed as a Group 1 carcinogen by the IARC. In the 18th century a scrotal cancer of chimney sweepers, the chimney sweeps' carcinoma, was already known to be connected to soot.
Description
Benzo[a]pyrene (BaP) is a polycyclic aromatic hydrocarbon found in coal tar with the formula C20H12. The compound is one of the benzopyrenes, formed by a benzene ring fused to pyrene, and is the result of incomplete combustion at temperatures between 300 °C (572 °F) and 600 °C (1,112 °F).
Sources
The main source of atmospheric BaP is residential wood burning.[4] It is also found in coal tar, in automobile exhaust fumes (especially from diesel engines), in all smoke resulting from the combustion of organic material (including cigarette smoke), and in charbroiled food. A 2001 National Cancer Institute study found levels of BaP to be significantly higher in foods that were cooked well-done on the barbecue, particularly steaks, chicken with skin, and hamburgers: Cooked meat products have been shown to contain up to 4 ng/g of BaP,[5] and up to 5.5 ng/g in fried chicken[6] and 62.6 ng/g in overcooked charcoal barbecued beef.[7]
BaP is discharged in wastewater by industries such as smelters, particularly iron and steel mills[8] and aluminium smelters.[9]
History
In the 18th century, young British chimney sweeps who climbed into chimneys suffered from chimney sweeps' carcinoma, a scrotal cancer peculiar to their profession, and this was connected to the effects of soot in 1775,[10] in the first work of occupational cancer epidemiology and also the first connection of any chemical mixture to cancer formation. Frequent skin cancers were noted among fuel industry workers in the 19th century. In 1933, BaP was determined to be the compound responsible for these cases, and its carcinogenicity was demonstrated when skin tumors occurred in laboratory animals repeatedly painted with coal tar.[11] BaP has since been identified as a prime carcinogen in cigarette smoke.[12]
Toxicity
Nervous system
Prenatal exposure of BaP in rats is known to affect learning and memory in rodent models. Pregnant rats eating BaP were shown to negatively affect the brain function in the late life of their offspring. At a time when synapses are first formed and adjusted in strength by activity, BaP diminished NMDA receptor-dependent nerve cell activity measured as mRNA expression of the NMDA NR2B receptor subunit.[13]
Immune system
BaP has an effect on the number of white blood cells, inhibiting some of them from differentiating into macrophages, the body's first line of defense to fight infections. In 2016, the molecular mechanism was uncovered as damage to the macrophage membrane's lipid raft integrity by decreasing membrane cholesterol at 25%. This means less immunoreceptors CD32 (a member of the Fc family of immunoreceptors) could bind to IgG and turn the white blood cell into a macrophage. Therefore, macrophage membranes become susceptible to bacterial infections.[14]
Reproductive system
In experiments with male rats, subchronic exposure to inhaled BaP has been shown to generally reduce the function of testicles and epididymis with lower sex steroid/testosterone production and sperm production.[15]
Carcinogenicity
BaP's metabolites are mutagenic and highly carcinogenic, and it is listed as a Group 1 carcinogen by the IARC. Chemical agents and related occupations, Volume 10, A review of Human Carcinogens, IARC Monographs, Lyon France 2009 [16]
In June 2016, BaP was added as benzo[def]chrysene to the REACH Candidate List of Substances of very high concern for Authorisation.[17]
Numerous studies since the 1970s have documented links between BaP and cancers.[18] It has been more difficult to link cancers to specific BaP sources, especially in humans, and difficult to quantify risks posed by various methods of exposure (inhalation or ingestion).[19] A link between vitamin A deficiency and emphysema in smokers was described in 2005 to be due to BaP, which induces vitamin A deficiency in rats.[20]
A 1996 study provided molecular evidence linking components in tobacco smoke to lung cancer. BaP was shown to cause genetic damage in lung cells that was identical to the damage observed in the DNA of most malignant lung tumours.[21]
Regular consumption of cooked meats has been epidemiologically associated with increased levels of colon cancer[22] (although this in itself does not prove carcinogenicity),[23] A 2005 NCI study found an increased risk of colorectal adenomas was associated with BaP intake, and more strongly with BaP intake from all foods.[24]
The detoxification enzymes cytochrome P450 1A1 (CYP1A1) and cytochrome P450 1B1 (CYP1B1) are both protective and necessary for benzo[a]pyrene toxicity. Experiments with strains of mice engineered to remove (knockout) CYP1A1 and CYP1B1 reveal that CYP1A1 primarily acts to protect mammals from low doses of BaP, and that removing this protection accumulates large concentrations of BaP. Unless CYP1B1 is also knocked out, toxicity results from the bioactivation of BaP to benzo[a]pyrene -7,8-dihydrodiol-9,10-epoxide, the ultimate toxic compound.[25][better source needed]
Interaction with DNA

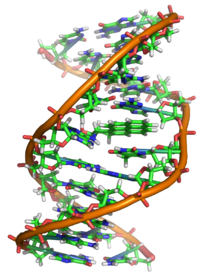
Properly speaking, BaP is a procarcinogen, meaning that its mechanism of carcinogenesis depends on its enzymatic metabolism to BaP diol epoxide[27] It intercalates in DNA, and the electrophilic epoxide is attacked by nucleophilic guanine bases, forming a bulky guanine adduct.[27]

X-ray crystallographic and nuclear magnetic resonance structure studies have shown how this binding distorts the DNA[28] by confusing the double-helical DNA structure. This disrupts the normal process of copying DNA and causes mutations, which explains the occurrence of cancer after exposure. This mechanism of action is similar to that of aflatoxin which binds to the N7 position of guanine.[29]
There are indications that benzo[a]pyrene diol epoxide specifically targets the protective p53 gene.[30] This gene is a transcription factor that regulates the cell cycle and hence functions as a tumor suppressor. By inducing G (guanine) to T (thymidine) transversions in transversion hotspots within p53, there is a probability that benzo[a]pyrene diol epoxide inactivates the tumor suppression ability in certain cells, leading to cancer.
Benzo[a]pyrene-7,8-dihydrodiol-9,10-epoxide is the carcinogenic product of three enzymatic reactions:[31]
- Benzo[a]pyrene is first oxidized by cytochrome P450 1A1 to form a variety of products, including (+)benzo[a]pyrene-7,8-epoxide.[32]
- This product is metabolized by epoxide hydrolase, opening up the epoxide ring to yield (−)benzo[a]pyrene-7,8-dihydrodiol.
- The ultimate carcinogen is formed after another reaction with cytochrome P450 1A1 to yield the (+)benzo[a]pyrene-7,8-dihydrodiol-9,10-epoxide. It is this diol epoxide that covalently binds to DNA.
BaP induces cytochrome P450 1A1 (CYP1A1) by binding to the AHR (aryl hydrocarbon receptor) in the cytosol.[33] Upon binding the transformed receptor translocates to the nucleus where it dimerises with ARNT (aryl hydrocarbon receptor nuclear translocator) and then binds xenobiotic response elements (XREs) in DNA located upstream of certain genes. This process increases transcription of certain genes, notably CYP1A1, followed by increased CYP1A1 protein production.[33] This process is similar to induction of CYP1A1 by certain polychlorinated biphenyls and dioxins. Seemingly, CYP1A1 activity in the intestinal mucosa prevents major amounts of ingested benzo[a]pyrene to enter portal blood and systemic circulation.[34] Intestinal, but not hepatic, expression of CYP1A1 depends on TOLL-like receptor 2 (TLR2),[35] which is a eukaryotic receptor for bacterial surface structures such as lipoteichoic acid.
Moreover, BaP has been found to activate a transposon, LINE1, in humans.[36]
Nucleotide excision repair
As illustrated above, (+)benzo[a]pyrene-7,8-dihydrodiol-9,10-epoxide (BPDE) forms bulky covalent DNA adducts with guanines. Most of these adducts can be efficiently eliminated from DNA by the process of nucleotide excision repair.[37] Those adducts that are not removed can cause errors during DNA replication leading to carcinogenic mutations.
See also
- Benzopyrene
- Benzo[e]pyrene
- Pyrene, a four-ring analogue
- Toxification
References
- ^ Henri A. Favre, Warren H. Powell (2013). Nomenclature of Organic Chemistry: IUPAC Recommendations and Preferred Names 2013. Royal Society of Chemistry. p. 232. ISBN 978-0-85404-182-4.
- ^ William M. Haynes (2016). CRC Handbook of Chemistry and Physics (97th ed.). Boca Raton: CRC Press. pp. 3–42. ISBN 978-1-4987-5429-3.
- ^ "benzo[a]pyrene". pubchem.ncbi.nlm.nih.gov.
- ^ "Assessment of Benzo-alpha-pyrene Emissions in the Great Lakes Region" (PDF). pp. 23–24.
- ^ Kazerouni, N; Sinha, R; Hsu, CH; et al. (2002). "Analysis of 200 food items for benzo[a]pyrene and estimation of its intake in an epidemiologic study". Food and Chemical Toxicology. 40 (1): 423–36. doi:10.1016/S0278-6915(00)00158-7. PMID 11313108.
- ^ Lee, BM; Shim, GA (Aug 2007). "Dietary exposure estimation of benzo[a]pyrene and cancer risk assessment". Journal of Toxicology and Environmental Health Part A. 70 (15–16): 1391–4. doi:10.1080/15287390701434182. PMID 17654259. S2CID 21302834.
- ^ Aygün, SF; Kabadayi, F (December 2005). "Determination of benzo[a]pyrene in charcoal grilled meat samples by HPLC with fluorescence detection". Int J Food Sci Nutr. 56 (8): 581–5. doi:10.1080/09637480500465436. PMID 16638662. S2CID 35095622.
- ^ U.S. Environmental Protection Agency (EPA), Washington, D.C. (2002). "Iron and Steel Manufacturing Point Source Category." Code of Federal Regulations, 40 CFR Part 420.
- ^ EPA (1984). "Nonferrous Metals Manufacturing Point Source Category." Code of Federal Regulations, 40 CFR Part 421.
- ^ Pott, Percivall (1775). Chirurgical Observations …. London, England: L. Hawes, W. Clarke, and R. Collins. pp. 63–68. From p. 67: "The disease, in these people [i.e., chimney sweeps], seems to derive its origin from a lodgment of soot in the rugae of the scrotum, … "
- ^ Cook, J. W.; Hewett, C. L.; Hieger, I. (1933). "The isolation of a cancer producing Hydrocarbon from coal tar". J. Chem. Soc. 1933: 395–405. doi:10.1039/jr9330000395.
- ^ Hecht, SS (1999). "Tobacco smoke carcinogens and lung cancer". J Natl Cancer Inst. 91 (14): 1194–210. doi:10.1093/jnci/91.14.1194. PMID 10413421.
- ^ McCallister, M. M.; Maguire, M; Ramesh, A; et al. (2008). "Prenatal Exposure to Benzo[a]pyrene Impairs Later-Life Cortical Neuronal Function". NeuroToxicology. 29 (5): 846–854. doi:10.1016/j.neuro.2008.07.008. PMC 2752856. PMID 18761371..
- ^ Clark RS, Pellom ST, Booker B, et al. (2016). "Validation of research trajectory 1 of an Exposome framework: Exposure to benzo[a]pyrene confers enhanced susceptibility to bacterial infection". Environ Research. 146: 173–84. Bibcode:2016ER....146..173C. doi:10.1016/j.envres.2015.12.027. PMC 5523512. PMID 26765097.
- ^ Ramesh A1, Inyang F; Lunstra, DD; Niaz, MS; et al. (Aug 2008). "Alteration of fertility endpoints in adult male F-344 rats by subchronic exposure to inhaled benzo(a)pyrene". Exp Toxicol Pathol. 60 (4–5): 269–80. doi:10.1016/j.etp.2008.02.010. PMC 3526104. PMID 18499416.
{{cite journal}}: CS1 maint: numeric names: authors list (link) - ^ A review of human carcinogens—part F: chemical agents and related occupations
- ^ European Chemicals Agency. "ED/21/2016". ECHA. Retrieved 21 June 2016.
- ^ Kleiböhmer, W. (2001). "Polycyclic Aromatic Hydrocarbon (PAH) Metabolites". Environmental Analysis (Volume 3 of Handbook of Analytical Separations). Elsevier. pp. 99–122. ISBN 978-0-08-050576-3.
- ^ Armstrong, B.; Hutchinson, E.; Unwin, J.; Fletcher, T. (2004). "Lung Cancer Risk After Exposure to Polycyclic Aromatic Hydrocarbons: A Review and Meta-Analysis". Environmental Health Perspectives. 112 (9): 970–978. doi:10.1289/ehp.6895. PMC 1247189. PMID 15198916.
- ^ "Benzopyrene and Vitamin A deficiency". Researcher links cigarettes, vitamin A and emphysema. Retrieved March 5, 2005.
- ^ Denissenko, M. F.; Pao, A.; Tang, M.; Pfeifer, G. P. (1996-10-18). "Preferential formation of benzo[a]pyrene adducts at lung cancer mutational hotspots in P53". Science. 274 (5286): 430–432. Bibcode:1996Sci...274..430D. doi:10.1126/science.274.5286.430. PMID 8832894. S2CID 3589066.
- ^ Le Marchand, L; Hankin, JH; Pierce, LM; et al. (September 2002). "Well-done red meat, metabolic phenotypes and colorectal cancer in Hawaii". Mutat. Res. 506–507: 205–14. doi:10.1016/s0027-5107(02)00167-7. PMID 12351160.
- ^ Truswell, AS (Mar 2002). "Meat consumption and cancer of the large bowel". European Journal of Clinical Nutrition. 56 (Suppl 1): S19–24. doi:10.1038/sj.ejcn.1601349. PMID 11965518. S2CID 23886438.
- ^ Sinha, Rashmi; Kulldorff, Martin; Gunter, Marc J.; et al. (August 2005). "Dietary benzo[a]pyrene intake and risk of colorectal adenoma". Cancer Epidemiology, Biomarkers & Prevention. 14 (8): 2030–2034. doi:10.1158/1055-9965.EPI-04-0854. PMID 16103456. S2CID 33819830.
- ^ Data presented by Daniel W. Nebert in research seminars 2007
- ^ Created from PDB 1JDG Archived 2008-09-22 at the Wayback Machine
- ^ a b Bogdan, Dennis Paul (February 1, 1973). Interactions Of Benzopyrene And Nucleic Acids In The Presence Of A Mixed-Function Oxidase System - Ph.D. Dissertation (requires login) (Thesis). State University of New York at Buffalo. ISBN 979-8661021359. ProQuest 302781013. Retrieved May 9, 2021.
- ^ Volk, David E.; Thiviyanathan, Varatharasa; Rice, Jeffrey S.; et al. (2003-02-18). "Solution structure of a cis-opened (10R)-N6-deoxyadenosine adduct of (9S,10R)-9,10-epoxy-7,8,9,10-tetrahydrobenzo[a]pyrene in a DNA duplex". Biochemistry. 42 (6): 1410–1420. doi:10.1021/bi026745u. PMID 12578353.
- ^ Eaton, D. L.; Gallagher, E. P. (1994). "Mechanisms of aflatoxin carcinogenesis". Annual Review of Pharmacology and Toxicology. 34: 135–172. doi:10.1146/annurev.pa.34.040194.001031. PMID 8042848.
- ^ Pfeifer, Gerd P.; Denissenko, Mikhail F.; Olivier, Magali; et al. (2002-10-21). "Tobacco smoke carcinogens, DNA damage and p53 mutations in smoking-associated cancers". Oncogene. 21 (48): 7435–7451. doi:10.1038/sj.onc.1205803. PMID 12379884. S2CID 6134471.
- ^ Jiang, Hao; Gelhaus, Stacy L.; Mangal, Dipti; et al. (2007). "Metabolism of Benzo[a]pyrene in Human Bronchoalveolar H358 Cells Using Liquid Chromatography-Mass Spectrometry". Chem. Res. Toxicol. 20 (9): 1331–1341. doi:10.1021/tx700107z. PMC 2423818. PMID 17702526.
- ^ Shou, M; Gonzalez, FJ; Gelboin, HV (December 1996). "Stereoselective epoxidation and hydration at the K-region of polycyclic aromatic hydrocarbons by cDNA-expressed cytochromes P450 1A1, 1A2, and epoxide hydrolase". Biochemistry. 35 (49): 15807–13. doi:10.1021/bi962042z. PMID 8961944.
- ^ a b Whitlock, JP Jr. (April 1999). "Induction of cytochrome P4501A1". Annual Review of Pharmacology and Toxicology. 39: 103–125. doi:10.1146/annurev.pharmtox.39.1.103. PMID 10331078.
- ^ Uno, S.; Dragin, N; Miller, ML; et al. (2008). "Basal and inducible CYP1 mRNA quantitation and protein localization throughout the mouse gastrointestinal tract". Free Radic Biol Med. 44 (4): 570–83. doi:10.1016/j.freeradbiomed.2007.10.044. PMC 2754765. PMID 17997381.
- ^ Do, KN; Fink, LN; Jensen, TE; et al. (2012). "TLR2 controls intestinal carcinogen detoxication by CYP1A1". PLOS ONE. 7 (3): e32309. Bibcode:2012PLoSO...732309D. doi:10.1371/journal.pone.0032309. PMC 3307708. PMID 22442665.
- ^ Stribinskis, Vilius; Ramos, Kenneth S. (2006). "Activation of Human Long Interspersed Nuclear Element 1 Retrotransposition by Benzo(a)pyrene, a Ubiquitous Environmental Carcinogen". Cancer Res. 66 (5): 2616–20. doi:10.1158/0008-5472.CAN-05-3478. PMID 16510580.
- ^ Li, Wentao; Hu, Jinchuan; Adebali, Ogun; et al. (2017-06-27). "Human genome-wide repair map of DNA damage caused by the cigarette smoke carcinogen benzo[a]pyrene". Proceedings of the National Academy of Sciences of the United States of America. 114 (26): 6752–6757. Bibcode:2017PNAS..114.6752L. doi:10.1073/pnas.1706021114. PMC 5495276. PMID 28607059.
External links
- International Chemical Safety Card 0104
- National Pollutant Inventory – Polycyclic Aromatic Hydrocarbon Fact Sheet
- "Lung cancer as consequence by Benzopyrene in smokers". Lung Cancer. Archived from the original on April 14, 2005. Retrieved March 5, 2005.
- "Levels of Benzopyrene in Burnt toasts". Guardian Unlimited, Special reports: Close encounters. Retrieved March 5, 2005.
- Karle, I. L. (2004). "Crystal and molecular structure of a benzo[a]pyrene-7,8-diol-9,10-epoxide N2-deoxyguanosine adduct: Absolute configuration and conformation". Proceedings of the National Academy of Sciences. 101 (6): 1433–8. Bibcode:2004PNAS..101.1433K. doi:10.1073/pnas.0307305101. PMC 341736. PMID 14757823.
- CS1 maint: numeric names: authors list
- CS1: long volume value
- Webarchive template wayback links
- Articles with short description
- Short description is different from Wikidata
- Restricted titles (brackets)
- All articles with unsourced statements
- Articles with unsourced statements from May 2019
- ECHA InfoCard ID from Wikidata
- Chembox having GHS data
- Articles containing unverified chemical infoboxes
- Short description matches Wikidata
- All articles lacking reliable references
- Articles lacking reliable references from July 2016
- IARC Group 1 carcinogens
- Polycyclic aromatic hydrocarbons
- PBT substances