Chronic traumatic encephalopathy
| Chronic traumatic encephalopathy | |
|---|---|
| Other names: Traumatic encephalopathy syndrome, dementia pugilistica,[1] punch drunk syndrome | |
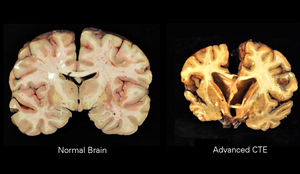 | |
| A normal brain (left) and one with CTE (right) | |
| Specialty | Neurology, psychiatry, sports medicine |
| Symptoms | Behavioral problems, mood problems, problems with thinking[1] |
| Complications | Dementia,[2] aggression, depression, suicidal thoughts[3] |
| Usual onset | Years after initial injuries[2] |
| Causes | Repeated head injuries[1] |
| Risk factors | Contact sports, military, domestic abuse, repeated banging of the head[1] |
| Diagnostic method | Autopsy[1] |
| Differential diagnosis | Alzheimer's disease, Parkinson's disease[3] |
| Treatment | Supportive care[3] |
| Frequency | Uncertain[2] |
Chronic traumatic encephalopathy (CTE) is a neurodegenerative disease caused by repeated head injuries.[1] Symptoms do not typically begin until years after the injuries and can include behavioral problems, mood problems, and problems with thinking.[1][2] The disease often gets worse over time and can result in dementia.[2] It is unclear if the risk of suicide is altered.[1]
Most documented cases have occurred in athletes involved in contact sports such as boxing, American football, professional wrestling, ice hockey, rugby, and association football (soccer).[1][4] Other risk factors include being in the military, prior domestic violence, and repeated banging of the head.[1] The exact amount of trauma required for the condition to occur is unknown, and definitive diagnosis can only occur at autopsy.[1] The disease is classified as a tauopathy.[1]
There is no specific treatment for the disease.[3] Rates of CTE have been found to be about 30% among those with a history of multiple head injuries,[1] however population rates are unclear.[2] Research in brain damage as a result of repeated head injuries began in the 1920s, at which time the condition was known as dementia pugilistica or "punch drunk syndrome".[1][3] It has been proposed that the rules of some sports be changed as a means of prevention.[1]
Signs and symptoms
Symptoms of CTE, which occur in four stages, generally appear eight to ten years after an individual experiences repetitive mild traumatic brain injuries.[5]
First-stage symptoms are confusion, disorientation, dizziness, and headaches. Second-stage symptoms include memory loss, social instability, impulsive behavior, and poor judgment. Third and fourth stages include progressive dementia, movement disorders, hypomimia, speech impediments, sensory processing disorder, tremors, vertigo, deafness, depression and suicidality.
Additional symptoms include dysarthria, dysphagia, cognitive disorders such as amnesia, and ocular abnormalities, such as ptosis.[6]
The condition manifests as dementia, or declining mental ability, problems with memory, dizzy spells or lack of balance to the point of not being able to walk under one's own power for a short time and/or Parkinsonism, or tremors and lack of coordination. It can also cause speech problems and an unsteady gait. Patients with CTE may be prone to inappropriate or explosive behavior and may display pathological jealousy or paranoia.[7]
Cause
Most documented cases have occurred in athletes with mild repetitive brain trauma (RBT) over an extended period of time. Specifically contact sports such as boxing, American football, wrestling, ice hockey, rugby, and association football.[1][4] In soccer, whether this is just associated with prolific headers or other injuries is unclear as of 2017.[8] Other potential risk factors include military personnel (repeated exposure to concussions charges or large caliber ordnance), domestic violence, and repeated banging of the head.[1] The exact amount of trauma required for the condition to occur is unknown although it is believed that it would take many years to develop it.[1]
Pathology
The neuropathological appearance of CTE is distinguished from other tauopathies, such as Alzheimer's disease. The four clinical stages of observable CTE disability have been correlated with tau pathology in brain tissue, ranging in severity from focal perivascular epicenters of neurofibrillary tangles in the frontal neocortex to severe tauopathy affecting widespread brain regions.[9]
The primary physical manifestations of CTE include a reduction in brain weight, associated with atrophy of the frontal and temporal cortices and medial temporal lobe. The lateral ventricles and the third ventricle are often enlarged, with rare instances of dilation of the fourth ventricle.[10] Other physical manifestations of CTE include anterior cavum septi pellucidi and posterior fenestrations, pallor of the substantia nigra and locus ceruleus, and atrophy of the olfactory bulbs, thalamus, mammillary bodies, brainstem and cerebellum.[11] As CTE progresses, there may be marked atrophy of the hippocampus, entorhinal cortex, and amygdala.[5]
On a microscopic scale, the pathology includes neuronal loss, tau deposition, TAR DNA-binding Protein 43 (TDP 43)[9] deposition, white matter changes, and other abnormalities. The tau deposition occurs as dense neurofibrillary tangles (NFT), neurites, and glial tangles, which are made up of astrocytes and other glial cells[10] Beta-amyloid deposition is a relatively uncommon feature of CTE.
A small group of individuals with CTE has chronic traumatic encephalomyopathy (CTEM), which is characterized by symptoms of motor-neuron disease and which mimics amyotrophic lateral sclerosis (ALS). Progressive muscle weakness and balance and gait problems (problems with walking) seem to be early signs of CTEM.[10]
Exosome vesicles created by the brain are potential biomarkers of TBI, including CTE.[12] A subtype of CTE is dementia pugilistica or boxer's dementia (from Latin pugilator - boxer) as it was initially found in those with a history of boxing, also called "punch-drunk syndrome".
Loss of neurons, scarring of brain tissue, collection of proteinaceous, senile plaques, hydrocephalus, attenuation of the corpus callosum, diffuse axonal injury, neurofibrillary tangles, and damage to the cerebellum are implicated in the syndrome. The condition may be etiologically related to Alzheimer's disease.[13] Neurofibrillary tangles have been found in the brains of dementia pugilistica patients, but not in the same distribution as is usually found in people with Alzheimer's.[14] One group examined slices of brain from patients having had multiple mild traumatic brain injuries and found changes in the cells' cytoskeletons, which they suggested might be due to damage to cerebral blood vessels.[15]
Increased exposure to concussions and sub-concussive blows is regarded as the most important risk factor, which can depend on the total number of fights, number of knockout losses, the duration of career, fight frequency, age of retirement, and boxing style.[16]
Diagnosis
Diagnosis of CTE cannot be made in living individuals. A clear diagnosis is possible during an autopsy.[17] Though there are signs and symptoms some researchers associate with CTE, there is no definitive test to prove the existence in a living person. Signs are also very similar to that of other neurological conditions such as Alzheimer's.
The lack of distinct biomarkers is the reason CTE cannot typically be diagnosed while a person is alive. Concussions are non-structural injuries and do not result in brain bleeding, which is why most concussions cannot be seen on routine neuroimaging tests such as CT or MRI.[18] Acute concussion symptoms (those that occur shortly after an injury) should not be confused with CTE. Differentiating between prolonged post-concussion syndrome (PCS, where symptoms begin shortly after a concussion and last for weeks, months, and sometimes even years) and CTE symptoms can be difficult. Research studies are currently examining whether neuroimaging can detect subtle changes in axonal integrity and structural lesions that can occur in CTE.[5] Recently, more progress in in-vivo diagnostic techniques for CTE has been made, using DTI, fMRI, MRI, and MRS imaging; however, more research needs to be done before any such techniques can be validated.[10]
PET tracers that bind specifically to tau protein are desired to aid diagnosis of CTE in living individuals. One candidate is the tracer [18F]FDDNP, which is retained in the brain in individuals with a number of dementing disorders such as Alzheimer's disease, Down syndrome, progressive supranuclear palsy, corticobasal degeneration, familial frontotemporal dementia, and Creutzfeldt–Jakob disease.[19] In a small study of 5 retired NFL players with cognitive and mood symptoms, the PET scans revealed accumulation of the tracer in their brains.[20] However, [18F]FDDNP binds to beta-amyloid and other proteins as well. Moreover, the sites in the brain where the tracer was retained were not consistent with the known neuropathology of CTE.[21] A more promising candidate is the tracer [18F]-T807, which binds only to tau. It is being tested in several clinical trials.[21]
A putative biomarker for CTE is the presence in serum of autoantibodies against the brain. The autoantibodies were detected in football players who experienced a large number of head hits but no concussions, suggesting that even sub-concussive episodes may be damaging to the brain. The autoantibodies may enter the brain by means of a disrupted blood-brain barrier, and attack neuronal cells which are normally protected from an immune onslaught.[22] Given the large numbers of neurons present in the brain (86 billion), and considering the poor penetration of antibodies across a normal blood-brain barrier, there is an extended period of time between the initial events (head hits) and the development of any signs or symptoms. Nevertheless, autoimmune changes in blood of players may consist the earliest measurable event predicting CTE.[23]
Imaging
Although the diagnosis of CTE cannot be determined by imaging, the effects of head trauma may be seen with the use of structural imaging.[24] Imaging techniques include the use of magnetic resonance imaging, nuclear magnetic resonance spectroscopy, CT scan, single-photon emission computed tomography, Diffusion MRI, and Positron emission tomography (PET).[24] One specific use of imaging is the use of a PET scan is to evaluate for tau deposition, which has been conducted on retired NFL players.[25]
Prevention
Prevention of CTE in sport is not an idealistic goal because repetitive concussions increase the risk for this condition.[26] Prevention techniques are also difficult because diagnosis of the condition can only be during a postmortem autopsy.[24] The initial onset of this condition can not yet be determined, and therefore creating techniques for prevention pose a struggle.
Some common preventative methods have been the use of helmets and mouth-guards; though neither has significant research to support its use, both have been shown to reduce direct head trauma.[26] Although there is no significant research to support the use of helmets to reduce the risk of concussions, there is evidence to support that helmet use reduces impact forces. Mouth guards have been shown to decrease dental injuries, but again have not shown significant evidence to reduce concussions.[24] A growing area of practice is improved recognition and treatment for concussions and other head trauma, since repeated impacts are thought to increase the likelihood of CTE development, removal from sport during these traumatic incidences is essential.[24] Proper return-to-play protocol after possible brain injuries is also important in decreasing the significance of future impacts.[24]
Another factor that has been implemented and continues to be an area of debate is changing the rules of many contact sports to make them effectively safer.[24] Examples of these rules are the evolution of tackling technique rules in American football, such as the banning of helmet-first tackles, and the addition of rules to protect defenseless players. Likewise, another growing area of debate is better implementation of rules already in place to protect athletes.[24]
Because of the concern that boxing may cause CTE, there is a movement among medical professionals to ban the sport.[7] Medical professionals have called for such a ban as early as the 1950s.[6]
Management
No cure currently exists for CTE.[27] Treatment is supportive as with other forms of dementia.[28] Those with CTE-related symptoms may receive medication and non-medication related treatments.[29]
Epidemiology
Rates of disease have been found to be about 30% among those with a history of multiple head injuries.[1] Population rates, however, are unclear.[2]
Professional level athletes are the largest group with CTE, due to frequent concussions and sub-concussive impacts from play in contact sport.[30] These contact-sports include American football, Australian rules football,[31] ice hockey, rugby,[32] boxing, mixed martial arts, association football,[33][32] and wrestling.[34] In association football, only prolific headers are known to have developed CTE.[33]
Other individuals diagnosed with CTE were those involved in military service, had a previous history of chronic seizures, were domestically abused, or were involved in activities resulting in repetitive head collisions.[35][36][37]
History
CTE was originally studied in boxers in the 1920s as dementia pugilistica. DP was first described in 1928 by a forensic pathologist, Dr. Harrison Stanford Martland, who was the chief medical examiner of Essex County in Newark, New Jersey in a Journal of the American Medical Association article, in which he noted the tremors, slowed movement, confusion and speech problems typical of the condition.[38] The initial diagnosis of dementia pugilistica was derived from the Latin word for boxer pugil (akin to pugnus ‘fist’, pugnāre ‘to fight’).[39][40]
Other terms for the condition have included chronic boxer's encephalopathy, traumatic boxer's encephalopathy, boxer's dementia, pugilistic dementia, chronic traumatic brain injury associated with boxing (CTBI-B), and punch-drunk syndrome.[3]
The seminal work on the disease came from British neurologist Macdonald Critchley, who in 1949 wrote a paper titled "Punch-drunk syndromes: the chronic traumatic encephalopathy of boxers."[41] CTE was first recognized as affecting individuals who took considerable blows to the head, but was believed to be confined to boxers and not other athletes. As evidence pertaining to the clinical and neuropathological consequences of repeated mild head trauma grew, it became clear that this pattern of neurodegeneration was not restricted to boxers, and the term chronic traumatic encephalopathy became most widely used.[42][43] In the early 2000s, Nigerian-American neuropathologist Bennet Omalu worked on the case of American football player Mike Webster, who died following unusual and unexplained behavior. In 2005 Omalu, along with colleagues in the Department of Pathology at the University of Pittsburgh, published his findings in the journal Neurosurgery in a paper which he titled "Chronic Traumatic Encephalopathy in a National Football League Player". This was followed by a paper on a second case in 2006 describing similar pathology.
In 2008, the Sports Legacy Institute joined with the Boston University School of Medicine (BUSM) to form the Center for the Study of Traumatic Encephalopathy (CSTE).[44] Brain Injury Research Institute (BIRI) also studies the impact of concussions.[45][46]
Research
In 2005, forensic pathologist Bennet Omalu, along with colleagues in the Department of Pathology at the University of Pittsburgh, published a paper, "Chronic Traumatic Encephalopathy in a National Football League Player", in the journal Neurosurgery, based on analysis of the brain of deceased former NFL center Mike Webster. This was then followed by a paper on a second case in 2006 describing similar pathology, based on findings in the brain of former NFL player Terry Long.
In 2008, the Center for the Study of Traumatic Encephalopathy (CSTE) at Boston University at the BU School of Medicine started the CSTE brain bank at the Bedford Veterans Administration Hospital to analyze the effects of CTE and other neurodegenerative diseases on the brain and spinal cord of athletes, military veterans, and civilians[9] To date, the CSTE Brain Bank is the largest CTE tissue repository in the world.[10] On 21 December 2009, the National Football League Players Association announced that it would collaborate with the CSTE at the Boston University School of Medicine to support the Center's study of repetitive brain trauma in athletes.[47] Additionally, in 2010 the National Football League gave the CSTE a $1 million gift with no strings attached.[48][49] In 2008, twelve living athletes (active and retired), including hockey players Pat LaFontaine and Noah Welch as well as former NFL star Ted Johnson, committed to donate their brains to CSTE after their deaths.[44][50] In 2009, NFL Pro Bowlers Matt Birk, Lofa Tatupu, and Sean Morey pledged to donate their brains to the CSTE.[51] In 2010, 20 more NFL players and former players pledged to join the CSTE Brain Donation Registry, including Chicago Bears linebacker Hunter Hillenmeyer, Hall of Famer Mike Haynes, Pro Bowlers Zach Thomas, Kyle Turley, and Conrad Dobler, Super Bowl Champion Don Hasselbeck and former pro players Lew Carpenter, and Todd Hendricks. In 2010, professional wrestlers Mick Foley, Booker T and Matt Morgan also agreed to donate their brains upon their deaths. Also in 2010, MLS player Taylor Twellman, who had to retire from the New England Revolution because of post-concussion symptoms, agreed to donate his brain upon his death. As of 2010, the CSTE Brain Donation Registry consists of over 250 current and former athletes.[52]
In related research, the Center for the Study of Retired Athletes, which is part of the Department of Exercise and Sport Science at the University of North Carolina at Chapel Hill, is conducting research funded by National Football League Charities to "study former football players, a population with a high prevalence of exposure to prior Mild Traumatic Brain Injury (MTBI) and sub-concussive impacts, in order to investigate the association between increased football exposure and recurrent MTBI and neurodegenerative disorders such as cognitive impairment and Alzheimer's disease (AD)".[53]
In February 2011, Dave Duerson committed suicide,[54] leaving text messages to loved ones asking that his brain be donated to research for CTE.[55] The family got in touch with representatives of the Boston University center studying the condition, said Robert Stern, the co-director of the research group. Stern said Duerson's gift was the first time of which he was aware that such a request had been made by someone who had committed suicide that was potentially linked to CTE.[56] Stern and his colleagues found high levels of the protein tau in Duerson's brain. These elevated levels, which were abnormally clumped and pooled along the brain sulci,[9] are indicative of CTE.[57]
In July 2010, NHL enforcer Bob Probert died of heart failure. Before his death, he asked his wife to donate his brain to CTE research because it was noticed that Probert experienced a mental decline in his 40s. In March 2011, researchers at Boston University concluded that Probert had CTE upon analysis of the brain tissue he donated. He is the second NHL player from the program at the Center for the Study of Traumatic Encephalopathy to be diagnosed with CTE postmortem.[58]
BUSM has also found indications of links between amyotrophic lateral sclerosis (ALS) and CTE in athletes who have participated in contact sports. Tissue for the study was donated by twelve athletes and their families to the CSTE Brain Bank at the Bedford, Massachusetts VA Medical Center.[59]
In 2013, President Barack Obama announced the creation of the Chronic Effects of Neurotrauma Consortium or CENC, a federally funded research project devised to address the long-term effects of mild traumatic brain injury in military service personnel (SM's) and Veterans.[60][61][62] The CENC is a multi-center collaboration linking premiere basic science, translational, and clinical neuroscience researchers from the DoD, VA, academic universities, and private research institutes to effectively address the scientific, diagnostic, and therapeutic ramifications of mild TBI and its long-term effects.[63][64][65][66][67] Nearly 20% of the more than 2.5 million U.S. Service Members (SMs) deployed since 2003 to Operation Enduring Freedom (OEF) and Operation Iraqi Freedom (OIF) have sustained at least one traumatic brain injury (TBI), predominantly mild TBI (mTBI),[68][69] and almost 8% of all OEF/OIF Veterans demonstrate persistent post-TBI symptoms more than six months post-injury.[70][71] Unlike those head injuries incurred in most sporting events, recent military head injuries are most often the result of blast wave exposure.[citation needed] After a competitive application process, a consortium led by Virginia Commonwealth University was awarded funding.[63][64][65][66][72][73] The project principal investigator for the CENC is David Cifu, Chairman and Herman J. Flax professor[74] of the Department of Physical Medicine and Rehabilitation (PM&R) at Virginia Commonwealth University (VCU) in Richmond, Virginia, with co-principal investigators Ramon Diaz-Arrastia, Professor of Neurology, Uniformed Services University of the Health Sciences,[66] and Rick L. Williams, statistician at RTI International. In 2017, Aaron Hernandez, a former professional football player and convicted murderer, committed suicide while in prison. His family donated his brain to BU's CTE Center. Ann McKee, the head of Center, concluded that "Hernandez had Stage 3 CTE, which researchers had never seen in a brain younger than 46 years old".[75]
See also
- Acquired brain injury
- Brain damage
- Traumatic brain injury
References
- ↑ 1.00 1.01 1.02 1.03 1.04 1.05 1.06 1.07 1.08 1.09 1.10 1.11 1.12 1.13 1.14 1.15 1.16 1.17 1.18 Asken, BM; Sullan, MJ; DeKosky, ST; Jaffee, MS; Bauer, RM (1 October 2017). "Research Gaps and Controversies in Chronic Traumatic Encephalopathy: A Review". JAMA Neurology. 74 (10): 1255–1262. doi:10.1001/jamaneurol.2017.2396. PMID 28975240.
- ↑ 2.0 2.1 2.2 2.3 2.4 2.5 2.6 Stein, TD; Alvarez, VE; McKee, AC (2014). "Chronic traumatic encephalopathy: a spectrum of neuropathological changes following repetitive brain trauma in athletes and military personnel". Alzheimer's Research & Therapy. 6 (1): 4. doi:10.1186/alzrt234. PMC 3979082. PMID 24423082.
- ↑ 3.0 3.1 3.2 3.3 3.4 3.5 "Alzheimer's & Dementia". Alzheimer's Association. alz.org. Archived from the original on 17 May 2018. Retrieved 21 September 2017.
- ↑ 4.0 4.1 Maroon, Joseph C; Winkelman, Robert; Bost, Jeffrey; Amos, Austin C; Mathyssek, Christina; Miele, Vincent (2015). "Chronic Traumatic Encephalopathy in Contact Sports: A Systematic Review of All Reported Pathological Cases". PLOS One. 10 (2): e0117338. Bibcode:2015PLoSO..1017338M. doi:10.1371/journal.pone.0117338. PMC 4324991. PMID 25671598.
- ↑ 5.0 5.1 5.2 McKee AC, Cantu RC, Nowinski CJ, Hedley-Whyte ET, Gavett BE, Budson AE, Santini VE, Lee HS, Kubilus CA, Stern RA (2009). "Chronic traumatic encephalopathy in athletes: progressive tauopathy after repetitive head injury". J Neuropathol Exp Neurol. 68 (7): 709–35. doi:10.1097/NEN.0b013e3181a9d503. PMC 2945234. PMID 19535999.
- ↑ 6.0 6.1 Corsellis; et al. (1973). "The Aftermath of Boxing". Psychological Medicine. 3 (3): 270–303. doi:10.1017/S0033291700049588. PMID 4729191.
- ↑ 7.0 7.1 Mendez MF (1995). "The neuropsychiatric aspects of boxing". International Journal of Psychiatry in Medicine. 25 (3): 249–262. doi:10.2190/CUMK-THT1-X98M-WB4C. PMID 8567192.
- ↑ Nitrini, R (2017). "Soccer (Football Association) and chronic traumatic encephalopathy: A short review and recommendation". Dementia & Neuropsychologia. 11 (3): 218–220. doi:10.1590/1980-57642016dn11-030002. PMC 5674664. PMID 29213517.
- ↑ 9.0 9.1 9.2 9.3 McKee AC, Stern RA, Nowinski CJ, Stein TD, Alvarez VE, Daneshvar DH, Lee HS, Wojtowicz SM, Hall G, Baugh CM, Riley DO, Kubilus CA, Cormier KA, Jacobs MA, Martin BR, Abraham CR, Ikezu T, Reichard RR, Wolozin BL, Budson AE, Goldstein LE, Kowall NW, Cantu RC (2013). "The spectrum of disease in chronic traumatic encephalopathy". Brain. 136 (Pt 1): 43–64. doi:10.1093/brain/aws307. PMC 3624697. PMID 23208308.
- ↑ 10.0 10.1 10.2 10.3 10.4 Baugh CM, Stamm JM, Riley DO, Gavett BE, Shenton ME, Lin A, Nowinski CJ, Cantu RC, McKee AC, Stern RA (2012). "Chronic traumatic encephalopathy: neurodegeneration following repetitive concussive and subconcussive brain trauma". Brain Imaging Behavior. 6 (2): 244–54. doi:10.1007/s11682-012-9164-5. PMID 22552850.
- ↑ Jancin, Bruce (1 June 2011). "Chronic traumatic encephalopathy test sought". Internal Medicine News. Archived from the original on 28 August 2021. Retrieved 15 December 2013.
- ↑ Taylor DD, Gercel-Taylor C (2014). "Exosome platform for diagnosis and monitoring of traumatic brain injury". Philosophical Transactions of the Royal Society of London. Series B, Biological Sciences. 369 (1652): 20130503. doi:10.1098/rstb.2013.0503. PMC 4142024. PMID 25135964.
- ↑ Graham DI and Gennareli TA. Chapter 5, "Pathology of Brain Damage After Head Injury" In, Cooper P and Golfinos G. 2000. Head Injury, 4th Ed. Morgan Hill, New York.
- ↑ Hof PR, Bouras C, Buée L, Delacourte A, Perl DP, Morrison JH (1992). "Differential Distribution of Neurofibrillary Tangles in the Cerebral Cortex of Dementia Pugilistica and Alzheimer's Disease Cases". Acta Neuropathologica. 85 (1): 23–30. doi:10.1007/BF00304630. PMID 1285493.
- ↑ Geddes JF, Vowles GH, Nicoll JA, Révész T (1999). "Neuronal Cytoskeletal Changes are an Early Consequence of Repetitive Head Injury". Acta Neuropathologica. 98 (2): 171–178. doi:10.1007/s004010051066. PMID 10442557.
- ↑ Jordan, B. D. (2009). Brain injury in boxing. Clinics in Sports Medicine, 28(4), 561–78, vi.
- ↑ Concannon, Leah (2014). "Counseling Athletes on the Risk on Chronic Traumatic Encephalopathy". Sports Health. 6 (5): 396–401. doi:10.1177/1941738114530958. PMC 4137675. PMID 25177414.
- ↑ Poirier MP (2003). "Concussions: Assessment, management, and recommendations for return to activity". Clinical Pediatric Emergency Medicine. 4 (3): 179–85. doi:10.1016/S1522-8401(03)00061-2.
- ↑ Villemagne VL, Fodero-Tavoletti MT, Masters CL, Rowe CC (2015). "Tau imaging: early progress and future directions". The Lancet. Neurology. 14 (1): 114–24. doi:10.1016/S1474-4422(14)70252-2. PMID 25496902.
- ↑ Small GW, Kepe V, Siddarth P, Ercoli LM, Merrill DA, Donoghue N, Bookheimer SY, Martinez J, Omalu B, Bailes J, Barrio JR (2013). "PET scanning of brain tau in retired national football league players: preliminary findings". Am J Geriatr Psychiatry. 21 (2): 138–44. CiteSeerX 10.1.1.372.2960. doi:10.1016/j.jagp.2012.11.019. PMID 23343487.
- ↑ 21.0 21.1 Montenigro PH, Corp DT, Stein TD, Cantu RC, Stern RA (2015). "Chronic traumatic encephalopathy: historical origins and current perspective". Annual Review of Clinical Psychology. 11: 309–30. doi:10.1146/annurev-clinpsy-032814-112814. PMID 25581233.
- ↑ John Mangels, Cleveland Plain Dealer Archived 6 November 2018 at the Wayback Machine, 2013/03.
- ↑ Marchi N, Bazarian JJ, Puvenna V, Janigro M, Ghosh C, Zhong J, Zhu T, Blackman E, Stewart D, Ellis J, Butler R, Janigro D (2013). "Consequences of repeated blood-brain barrier disruption in football players". PLOS One. 8 (3): e56805. Bibcode:2013PLoSO...856805M. doi:10.1371/journal.pone.0056805. PMC 3590196. PMID 23483891.
- ↑ 24.0 24.1 24.2 24.3 24.4 24.5 24.6 24.7 Concannon, Leah (October 2014). "Counseling Athletes on the Risk of Chronic Traumatic Encephalopathy". Sports Health. 6 (5): 396–401. doi:10.1177/1941738114530958. PMC 4137675. PMID 25177414.
- ↑ Stern, Robert A.; Adler, Charles H.; Chen, Kewei; Navitsky, Michael; Luo, Ji; Dodick, David W.; Alosco, Michael L.; Tripodis, Yorghos; Goradia, Dhruman D.; Martin, Brett; Mastroeni, Diego; Fritts, Nathan G.; Jarnagin, Johnny; Devous, Michael D.; Mintun, Mark A.; Pontecorvo, Michael J.; Shenton, Martha E.; Reiman, Eric M. (2019). "Tau Positron-Emission Tomography in Former National Football League Players". New England Journal of Medicine. 380 (18): 1716–1725. doi:10.1056/NEJMoa1900757. ISSN 0028-4793. PMC 6636818. PMID 30969506.
- ↑ 26.0 26.1 Saffary, Roya (2012). "From Concussion to Chronic Traumatic Encephalopathy: A Review". Journal of Clinical Sport Psychology: 315–362.
- ↑ "Alzheimer's & Dementia". Alzheimer's Association. alz.org. Archived from the original on 17 May 2018. Retrieved 21 September 2017.
- ↑ "Treating CTE". NHS Choices. GOV.UK. 1 October 2017. Archived from the original on 15 February 2018. Retrieved 14 February 2018.
- ↑ Cantu, Robert; Budson, Andrew (October 2019). "Management of chronic traumatic encephalopathy". Expert Review of Neurotherapeutics. 19 (10): 1015–1023. doi:10.1080/14737175.2019.1633916. PMID 31215252.
- ↑ Saulle M, Greenwald BD (2012). "Chronic traumatic encephalopathy: a review" (PDF). Rehabil Res Pract. 2012: 1–9. doi:10.1155/2012/816069. PMC 3337491. PMID 22567320. Archived (PDF) from the original on 25 July 2018. Retrieved 5 August 2020.
- ↑ "CTE discovered in Polly Farmer's brain in AFL-first". Archived from the original on 18 September 2020.
- ↑ 32.0 32.1 Stone, Paul (18 March 2014). "First Soccer and Rugby Players Diagnosed With CTE". Neurologic Rehabilitation Institute at Brookhaven Hospital. Archived from the original on 2 April 2016. Retrieved 21 March 2016.
- ↑ 33.0 33.1 Ling, Helen; Morris, Huw R.; Neal, James W.; Lees, Andrew J.; Hardy, John; Holton, Janice L.; Revesz, Tamas; Williams, David D.R. (March 2017). "Mixed pathologies including chronic traumatic encephalopathy account for dementia in retired association football (soccer) players". Acta Neuropathologica. 133 (3): 337–352. doi:10.1007/s00401-017-1680-3. PMC 5325836. PMID 28205009.
- ↑ Daneshvar DH, Nowinski CJ, McKee AC, Cantu RC (2011). "The epidemiology of sport-related concussion". Clin Sports Med. 30 (1): 1–17, vii. doi:10.1016/j.csm.2010.08.006. PMC 2987636. PMID 21074078.
- ↑ Daneshvar DH, Riley DO, Nowinski CJ, McKee AC, Stern RA, Cantu RC (2011). "Long-term consequences: effects on normal development profile after concussion". Phys Med Rehabil Clin N Am. 22 (4): 683–700, ix. doi:10.1016/j.pmr.2011.08.009. PMC 3208826. PMID 22050943.
- ↑ Concannon, Leah (October 2014). "Counseling Athletes on the Risk of Chronic Traumatic Encephalopathy". Sports Health. 6 (5): 396–401. doi:10.1177/1941738114530958. PMC 4137675. PMID 25177414.
- ↑ Shetty, Teena (2016). "Imaging in Chronic Traumatic Encephalopathy and Traumatic Brain Injury". Sports Health. 8 (1): 26–36. doi:10.1177/1941738115588745. PMC 4702153. PMID 26733590.
- ↑ Martland HS (1928). "Punch Drunk". Journal of the American Medical Association. 91 (15): 1103–1107. doi:10.1001/jama.1928.02700150029009.
- ↑ Pugilism (origin) Archived 27 February 2016 at the Wayback Machine, retrieved on 2 February 2013.
- ↑ NCERx. 2005. Brain Trauma, Subdural Hematoma and Dementia Pugilistica Archived 27 May 2007 at the Wayback Machine. About-dementia.com. Retrieved on 19 December 2007.
- ↑ "'Concussion' Subject Bennet Omalu Exaggerated His Role, Researchers Say" Archived 30 March 2019 at the Wayback Machine. CBS New York. 17 December 2015.
- ↑ Martland H (1928). "Punch Drunk". The Journal of the American Medical Association. 91 (15): 1103–07. doi:10.1001/jama.1928.02700150029009.
- ↑ Gavett BE, Stern RA, McKee AC (2011). "Chronic traumatic encephalopathy: a potential late effect of sport-related concussive and subconcussive head trauma". Clin Sports Med. 30 (1): 179–88, xi. doi:10.1016/j.csm.2010.09.007. PMC 2995699. PMID 21074091.
- ↑ 44.0 44.1 "New pathology findings show significant brain degeneration in professional athletes with history of repetitive concussions" Archived 22 June 2013 at the Wayback Machine, Center for the Study of Traumatic Encephalopathy, 25 September 2008.
- ↑ "Seau family revisiting brain decision". ESPN. 6 May 2012. Archived from the original on 6 May 2012.
- ↑ "Our Team". Brain Injury Research Institute. Archived from the original on 7 June 2011.
- ↑ Staff. "NFL Players Association to Support Brain Trauma Research at Boston University" Archived 13 December 2013 at the Wayback Machine, Center for the Study of Traumatic Encephalopathy press release dated 21 December 2009. Accessed 17 August 2010.
- ↑ Support and Funding Archived 15 July 2010 at the Wayback Machine, Center for the Study of Traumatic Encephalopathy. Accessed 17 August 2010.
- ↑ Schwarz, Alan. "N.F.L. Donates $1 Million for Brain Studies" Archived 19 July 2018 at the Wayback Machine, The New York Times, 20 April 2010. Accessed 17 August 2010.
- ↑ "Welch to donate brain for concussion study". Edmonton Journal. Archived from the original on 6 October 2010. Retrieved 18 December 2008.
- ↑ Staff. "Three active NFL Pro Bowl players to donate brains to research" Archived 19 February 2014 at the Wayback Machine, Center for the Study of Traumatic Encephalopathy press release dated 14 September 2009. Accessed 17 August 2010.
- ↑ Staff. "20 more NFL stars to donate brains to research" Archived 22 June 2013 at the Wayback Machine, Center for the Study of Traumatic Encephalopathy press release dated 1 February 2010. Accessed 17 August 2010.
- ↑ "A Study on the Association Between Football Exposure and Dementia in Retired Football Players". UNC College of Arts and Sciences. Archived from the original on 11 August 2012. Retrieved 1 August 2012.
- ↑ Smith, Michael David, "Boston researchers request Junior Seau's brain" Archived 19 May 2018 at the Wayback Machine. NBC Sports Pro Football Talk, 3 May 2012. Retrieved 3 May 2012.
- ↑ Kusinski, Peggy (1 February 2011). "Dave Duerson Committed Suicide: Medical Examiner". NBC Chicago. Archived from the original on 19 May 2018. Retrieved 20 February 2011.
- ↑ Schwarz, Alan (20 February 2011). "Before Suicide, Duerson Asked for Brain Study". The New York Times. Archived from the original on 4 May 2020. Retrieved 5 August 2020.
- ↑ Deardorff, Julie (2 May 2011). "Study: Duerson had brain damage at time of suicide". Los Angeles Times. Retrieved 2 May 2011.[dead link]
- ↑ Schwarz, Alan (2 March 2011). "Hockey Brawler Paid Price, With Brain Trauma". The New York Times. Archived from the original on 17 March 2011. Retrieved 14 March 2011.
- ↑ "Researchers Discover Brain Trauma in Sports May Cause a New Disease That Mimics ALS" Archived 3 May 2012 at the Wayback Machine, BUSM press release, 17 August, 2010 3:41 pm. Retrieved 11 September 2011.
- ↑ Jordan, Bryant (12 August 2013). "Obama Introduces New PTSD and Education Programs". military.com. Archived from the original on 2 May 2014. Retrieved 2 May 2014.
- ↑ "Obama administration to research TBI, PTSD in new efforts Read more: Chronic Effects of Neurotrauma Consortium". fiercegovernment.com. Archived from the original on 2 May 2014. Retrieved 2 May 2014.
- ↑ "DoD, VA Establish Two Multi-Institutional Consortia to Research PTSD and TBI". va.gov. Archived from the original on 2 May 2014. Retrieved 2 May 2014.
- ↑ 63.0 63.1 "Fact Sheet: Largest federal grant in VCU's history". spectrum.vcu.edu. Archived from the original on 2 May 2014. Retrieved 2 May 2014.
- ↑ 64.0 64.1 "VCU to lead major study of concussions". grpva.com. Archived from the original on 3 May 2014. Retrieved 2 May 2014.
- ↑ 65.0 65.1 "Brain trust – the US consortia tacking military PTSD and brain injury". army-technology.com. 9 March 2014. Archived from the original on 2 May 2014. Retrieved 2 May 2014.[unreliable source?]
- ↑ 66.0 66.1 66.2 "DOD partners to combat brain injury". army.mil. Archived from the original on 10 April 2014. Retrieved 2 May 2014.
- ↑ "RTI to research mild traumatic brain injury effects in US soldiers". army-technology.com. 2 August 2013. Archived from the original on 2 May 2014. Retrieved 2 May 2014.[unreliable source?]
- ↑ Warden D. Military TBI during the Iraq and Afghanistan wars. J Head Trauma Rehabil. 2006; 21 (5): 398–402.
- ↑ "DoD Worldwide Numbers for TBI". dvbic.dcoe.mil. Archived from the original on 17 January 2014. Retrieved 4 February 2013.
- ↑ Scholten JD, Sayer NA, Vanderploeg RD, Bidelspach DE, Cifu DX (2012). "Analysis of US Veterans Health Administration comprehensive evaluations for traumatic brain injury in Operation Enduring Freedom and Operation Iraqi Freedom Veterans". Brain Inj. 26 (10): 1177–1184. doi:10.3109/02699052.2012.661914. PMID 22646489.
- ↑ Taylor BC, Hagel EM, Carlson KF, Cifu DX, Cutting A, Bidelspach DE, Sayer NA (2012). "Prevalence and costs of co-occurring traumatic brain injury with and without psychiatric disturbance and pain among Afghanistan and Iraq War Veteran V.A. users". Med Care. 50 (4): 342–346. doi:10.1097/MLR.0b013e318245a558. PMID 22228249. Archived from the original on 10 August 2020. Retrieved 5 August 2020.
- ↑ "Fact Sheet: The Obama Administration's Work to Honor Our Military Families and Veterans". whitehouse.gov. 1 August 2013. Archived from the original on 26 February 2015. Retrieved 2 May 2014.
- ↑ "Fact Sheet: VCU will lead $62 million study of traumatic brain injuries in military personnel". news.vcu.edu. Archived from the original on 12 April 2014. Retrieved 2 May 2014.
- ↑ About Us Archived 22 December 2015 at the Wayback Machine, Department of Physical Medicine and Rehabilitation, Virginia Commonwealth University. Retrieved 21 December 2015.
- ↑ Kilgore, Adam (9 November 2017). "Aaron Hernandez suffered from most severe CTE ever found in a person his age". Washington Post. Archived from the original on 3 October 2020. Retrieved 12 July 2020.
External links
- League of Denial – FRONTLINE Archived 18 June 2020 at the Wayback Machine
| Classification |
|---|
- Pages with script errors
- Webarchive template wayback links
- All articles with dead external links
- Articles with dead external links from October 2016
- Articles with invalid date parameter in template
- All articles lacking reliable references
- Articles lacking reliable references from June 2016
- Use dmy dates from July 2020
- Articles with hatnote templates targeting a nonexistent page
- All articles with unsourced statements
- Articles with unsourced statements from December 2016
- Motor neuron diseases
- Neurotrauma
- Overuse injuries
- Sports injuries
- Sports controversies
- RTT
- National Football League controversies
- Professional wrestling controversies