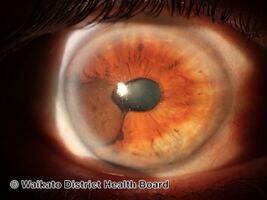
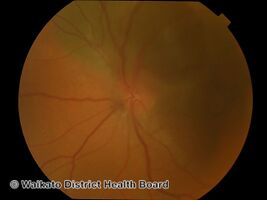
Uveal melanoma
| Uveal melanoma | |
|---|---|
| Other names: Intraocular melanoma, iris melanoma, ciliary body melanoma, choroidal melanoma[1] | |
 | |
| Iris melanoma | |
| Specialty | Oncology |
| Symptoms | Floaters, dark spot on the iris, change in the shape of pupil, poor or blurry vision in one eye, loss of peripheral vision in one eye[1] |
| Complications | Retinal detachment, spread[1] |
| Usual onset | Adults in their 50s[1] |
| Types | Class I (low metastatic risk) and class II (high metastatic risk), or iris, choroid, and ciliary body[2] |
| Risk factors | Spread[1] |
| Diagnostic method | By its appearance, medical imaging[1] |
| Differential diagnosis | For choroid: choroidal tumors, especially choroidal nevus, metastatic tumors, choroidal hemangioma, and osteoma; hemorrhagic conditions like AMD and hemorrhagic choroidal detachment; retinal tumors such as congenital retinal pigment epithelium hypertrophy and retinal pigment epithelium adenocarcinoma; and inflammatory lesions like posterior scleritis. For iris: iris nevus, iris pigment epithelial cyst, iris stromal cyst, metastatic tumor of the iris, melanocytoma, iris atrophy and Cogan-Reese syndrome. For ciliary body: staphyloma, medulloepithelioma and leiomyoma. |
| Prevention | Reduce UV exposure to the eye.[1] |
| Treatment | Brachytherapy, enucleation, proton beam radiotherapy, transpupillary thermotherapy, photocoagulation, photodynamic therapy, local resection.[3] |
| Frequency | 2 million to 8 million cases per year worldwide, males=females[1] |
Uveal melanoma is a type of eye cancer in the uvea of the eye.[2] It is traditionally classed as originating in the iris, choroid, and ciliary body, but can also be divided into class I (low metastatic risk) and class II (high metastatic risk).[2] Symptoms include blurred vision, loss of vision or photopsia, but there may be no symptoms.[3]
It is a melanoma that originates in pigment producing cells that reside within the uvea and give color to the eye. These melanocytes are distinct from the retinal pigment epithelium cells underlying the retina that do not form melanomas. When eye melanoma is spread to distant parts of the body, the five-year survival rate is about 15%.[4] Diagnosis is by its appearance and medical imaging.[5]
It is the commonest type of primary eye cancer.[2] Males and females are affected equally.[1] Worldwide, there are around 2 million to 8 million cases per year.[1] More than 50% spread, mostly to the liver.[1]
Types
Uveal melanoma arises from any of the three parts of the uvea, and are sometimes referred to by their location, choroidal melanoma, ciliary body melanoma, or iris melanoma, but can also be divided into class I (low metastatic risk) and class II (high metastatic risk).[2]
Signs and symptoms
Symptoms and signs include blurred vision, double vision (diplopia), irritation or pain, a perception of flashes of light in the eye (photopsia), a reduction in the total field of vision, loss of vision, a sensation of a foreign body in the field of vision (floaters), redness, bulging or displacement of the eye (proptosis a change in the shape of the pupil, pressure within the eye, and metamorphopsia (a distortion of vision where, when a person looks at a grid of straight lines, the lines appear wavy and parts of the grid appears blank).[6]
Iris melanoma
Uveal tumors can originate from melanocytes residing within the iris. Benign melanocytic tumors, such as iris freckles and moles (nevi), are common and pose no health risks, unless they show signs of malignancy, in which case they are classified as iris melanomas. Though derived from uveal melanocytes, iris melanomas share more in common with cutaneous (skin) melanomas in that they frequently harbor BRAF mutations associated with ultraviolet damage.[7][8] Iris melanomas are much less likely to metastasize than other uveal melanomas, and less likely to impair vision if detected and treated early. Approximately 5% of uveal melanomas involve the iris.[9]
Choroid melanoma
Benign melanocytic tumors of the choroid, such as choroidal freckles and nevi, are very common and pose no health risks, unless they show signs of malignancy, in which case they are considered melanomas.[10][11] Uveal melanoma is distinct from most skin melanomas associated with ultraviolet exposure; however, it shares several similarities with non-sun-exposed melanomas, such as acral melanomas and mucosal melanomas. BRAF mutations are extremely rare in posterior uveal melanomas;[12] instead, uveal melanomas frequently harbor GNAQ/GNA11 mutations, a trait shared with blue nevi, nevus of Ota, and ocular melanosis.[13][14] As seen in BRAF, mutations in GNAQ/GNA11 are early events in tumorigenesis and are not prognostic for tumor stage or later metastatic spread.[15] In contrast, mutations in the gene BAP1 are strongly linked to metastatic spread and patient survival.[16] Incidence of posterior uveal melanoma is highest among people with light skin and blue eyes. Other risk factors, such as blue light exposure and arc welding, have been put forward, but are still debated in the field. Mobile phone use is not a risk factor for uveal melanoma.[17]
-
Variably pigmented, mushroom-shaped choroidal tumor has ruptured the Bruch membrane and grown into the subretinal space.
-
Malignant melanoma of the choroid
Ciliary body melanoma
Ciliary body melanoma symptoms include blurred or partial loss of vision, floaters, photophobia and severe eye pain.[18] It develops from melanocytes in the uveal tract, typically arising from a pre-existing melanocytic nevus.[19] Subtypes of ciliary body melanoma include spindle A, spindle B, and epithelioid types.[19] Risk factors include having a uveal nevus, congenital ocular melanocytosis, xeroderma pigmentosum and having other family members with the condition.[19] It is diagnosed by its appearance.[20] The eye pressure may be found to be low.[20]
It is most common in white people of North European origin and most frequently occurs in the sixth decade of life.[21]
Cause
The cause of uveal melanoma is unclear. Uveal nevi are common (5% of Caucasians),[22] but rarely progress to melanoma.
Metastasis
Because there are no lymphatic channels to the uveal tract, metastasis occurs through local extension and/or blood-borne dissemination.[23] The most common site of metastasis for uveal melanoma is the liver;[24] the liver is the first site of metastasis for 80%-90% of ocular melanoma patients.[25] Other common sites of metastasis include the lung, bones, and just beneath the skin (subcutaneous). Approximately 50 percent of patients will develop metastases within 15 years after treatment of the primary tumor, and the liver will be involved 90% of the time.[26] Metastasis can occur more than 10 years after treatment of the primary tumor, and patients should not be considered cured even after a 10-year interval of monitoring.[27] Molecular features of the tumor, including chromosome 3 status, chromosome 6p status, and chromosome 8q status and gene expression profiling (such as the DecisionDx-UM test), can be used to adjust this likelihood of metastasis for an individual patient.
The average survival time after diagnosis of liver metastases depends on the extent of systemic spread. The disease-free interval, the performance status, the liver substitution by metastases and the serum level of lactic dehydrogenase are the most important prognostic factors for metastatic uveal melanoma.[28] There is currently no cure for metastatic uveal melanoma.
Diagnosis
Diagnosis is by its appearance using an ophthalmoscope, ultrasonography of the eye and sometimes biopsy.[29]
Treatment
The treatment protocol for uveal melanoma has been directed by many clinical studies, the most important being The Collaborative Ocular Melanoma Study (COMS).[30] The treatment varies depending upon many factors, chief among them the size of the tumor and results from testing of biopsied material from the tumor. Primary treatment can involve removal of the affected eye (enucleation); however, this is now reserved for cases of extreme tumor burden or other secondary problems. Advances in radiation therapies have significantly decreased the number of patients treated by enucleation in developed countries. The most common radiation treatment is plaque brachytherapy, in which a small disc-shaped shield (plaque) encasing radioactive seeds (most often iodine-125, though ruthenium-106 and palladium-103 are also used) is attached to the outside surface of the eye, overlying the tumor. The plaque is left in place for a few days and then removed. The risk of metastasis after plaque radiotherapy is the same as that of enucleation, suggesting that micrometastatic spread occurs prior to treatment of the primary tumor. Other modalities of treatment include transpupillary thermotherapy, external beam proton therapy, resection of the tumor, gamma knife stereotactic radiosurgery, or a combination of different modalities. Different surgical resection techniques can include trans-scleral partial choroidectomy and transretinal endoresection. Recent analysis of genomic data led to a new analysis of clinical subtying in uveal melanoma.[31] Ocular melanoma expert Professor Sarah Coupland has recently suggested cautious optimism as new types of targeted therapeutics are tested and approved.[32]
Prognosis
When eye melanoma is spread to distant parts of the body, the five-year survival rate is about 15%.[4]
Several clinical and pathological prognostic factors have been identified that are associated with higher risk of metastasis of uveal melanomas. These include large tumor size, ciliary body involvement, presence of orange pigment overlying the tumor, and older patient age.[33][34] Likewise several histological and cytological factors are associated with higher risk of metastasis, including presence and extent of cells with epithelioid morphology, presence of looping extracellular matrix patterns, increased infiltration of immune cells,[24] and staining with several immunohistochemical markers.[35]
The most important genetic alteration associated with poor prognosis in uveal melanoma is inactivation of BAP1, which most often occurs through mutation of one allele and subsequent loss of an entire copy of chromosome 3 (monosomy 3) to unmask the mutant copy.[16] Because of this function in inactivation of BAP1, monosomy 3 correlates strongly with metastatic spread[36] Where BAP1 mutation status is not available, gains on chromosomes 6 and 8 can be used to refine the predictive value of the monosomy 3 screen, with gain of 6p indicating a better prognosis and gain of 8q indicating a worse prognosis in disomy 3 tumors.[37] In rare instances, monosomy 3 tumors may duplicate the BAP1-mutant copy of the chromosome to return to a disomic state referred to as isodisomy.[38] Thus, isodisomy 3 is prognostically equivalent to monosomy 3, and both can be detected by tests for chromosome 3 loss of heterozygosity.[39] Monosomy 3, along with other chromosomal gains, losses, amplifications, and LOH, can be detected in fresh or paraffin-embedded samples by virtual karyotyping.
The most accurate prognostic factor is molecular classification by gene expression profiling of uveal melanomas. This analysis has been used to identify two subclasses of uveal melanomas: class 1 tumors that have a very low risk of metastasis, and class 2 tumors that have a very high risk of metastasis.[40][41] Gene expression profiling outperforms all of the above-mentioned factors at predicting metastatic spread of the primary tumor, including monosomy 3.[42][43][44]
Surveillance
Currently, there is no consensus regarding type or frequency of scans following diagnosis and treatment of the primary eye tumor. Of the 50% of patients who develop metastatic disease, more than 90% of patients will develop liver metastases. As such, the majority of surveillance techniques are focused on the liver. These include abdominal magnetic resonance imaging (MRI), abdominal ultrasound and liver function tests. The scientific community is currently working to develop guidelines, but until then, each patient must take into consideration their individual clinical situation and discuss appropriate surveillance with their doctors.[45] Some ophthalmologists have also found promise with the use of intravitreal avastin injections in patients suffering from radiation-induced retinopathy, a side effect of plaque brachytherapy treatment, as well as imaging surveillance with SD-OCT.
Epidemiology
Uveal melanomas are the most common primary intraocular tumor in adults.[24] Uveal melanoma is classified as a rare cancer with 5.1 cases per million people per year.[46] The incidence has remained stable for several years.
In 2018, it was reported that two clusters of cases have been identified in Huntersville, North Carolina, and Auburn, Alabama. Cases involved alumni of Auburn University who were acquainted with each other. Health officials investigated and found that the "cluster" is an artifact of social networking.[47][48][49]
There are about 2500 patients with UM diagnosed annually in the US.[50]
History
Uveal melanoma was first described in the literature in 1809-1812 by two Scottish surgeons, Allan Burns and James Wardrop.[51]
See also
References
- ↑ 1.00 1.01 1.02 1.03 1.04 1.05 1.06 1.07 1.08 1.09 1.10 DE, Elder; D, Massi; RA, Scolyer; R, Willemze (2018). "2. Melanocytic tumours: Ocular melanocytic tumours, uveal melanoma". WHO Classification of Skin Tumours. Vol. 11 (4th ed.). Lyon (France): World Health Organization. pp. 137–138. ISBN 978-92-832-2440-2. Archived from the original on 2022-07-11. Retrieved 2022-08-22.
- ↑ 2.0 2.1 2.2 2.3 2.4 Harbour, J. William; Correa, Zelia M. (2021). "1. Molecular basis of uveal melanoma and emerging therapeutic targets". In Bernicker, Eric H. (ed.). Uveal Melanoma: Biology and Management. Switzerland: Springer. pp. 3–12. ISBN 978-3-030-78117-0. Archived from the original on 2022-08-23. Retrieved 2022-08-22.
- ↑ 3.0 3.1 Sadowsky, Dylan; Delijani, Kevin; Lim, John; Cabrera, Matthew (22 July 2022). "Uveal Melanoma". Georgetown Medical Review. doi:10.52504/001c.36973. Archived from the original on 28 July 2022. Retrieved 22 August 2022.
- ↑ 4.0 4.1 Eye Cancer Survival Rates Archived 2021-05-31 at the Wayback Machine, American Cancer Society, Last Medical Review: December 9, 2014 Last Revised: February 5, 2016
- ↑ Davila, Jose R.; Mruthyunjaya, Prithvi (2021). "8. Uveal melanoma: Imaging". In Bernicker, Eric H. (ed.). Uveal Melanoma: Biology and Management. Switzerland: Springer. pp. 89–102. ISBN 978-3-030-78117-0. Archived from the original on 2022-08-23. Retrieved 2022-08-22.
- ↑ "Archive copy". Archived from the original on 2021-05-24. Retrieved 2021-10-25.
{{cite web}}: CS1 maint: archived copy as title (link) - ↑ Henriquez F, Janssen C, Kemp EG, Roberts F (2007). "The T1799A BRAF mutation is present in iris melanoma". Invest Ophthalmol Vis Sci. 48 (11): 4897–900. doi:10.1167/iovs.07-0440. PMID 17962436.
- ↑ Hocker T, Tsao H (2007). "Ultraviolet radiation and melanoma: a systematic review and analysis of reported sequence variants". Hum Mutat. 28 (6): 578–88. doi:10.1002/humu.20481. PMID 17295241. S2CID 36900956.
- ↑ Damato B; Coupland S (2008). "Ocular melanoma". Melanoma Molecular Map Project. Archived from the original on 2012-04-23. Retrieved 2013-02-02.
- ↑ Augsburger JJ (1993). "Is observation really appropriate for small choroidal melanomas". Trans Am Ophthalmol Soc. 91: 147–75. PMC 1298464. PMID 8140689.
- ↑ Shields CL, Demirci H, Materin MA, Marr BP, Mashayekhi A, Shields JA (2004). "Clinical factors in the identification of small choroidal melanoma". Can J Ophthalmol. 39 (4): 351–57. doi:10.1016/s0008-4182(04)80005-x. PMID 15327099.
- ↑ Malaponte G, Libra M, Gangemi P, Bevelacqua V, Mangano K, D'Amico F, Mazzarino MC, Stivala F, McCubrey JA, Travali S (2006). "Detection of BRAF gene mutation in primary choroidal melanoma tissue". Cancer Biol Ther. 5 (2): 225–27. doi:10.4161/cbt.5.2.2429. PMID 16410717.
- ↑ Van Raamsdonk CD, Bezrookove V, Green G, Bauer J, Gaugler L, O'Brien JM, Simpson EM, Barsh GS, Bastian BC (2009). "Frequent somatic mutations of GNAQ in uveal melanoma and blue naevi". Nature. 457 (7229): 599–602. Bibcode:2009Natur.457..599V. doi:10.1038/nature07586. PMC 2696133. PMID 19078957.
- ↑ Van Raamsdonk CD, Griewank KG, Crosby MB, Garrido MC, Vemula S, Wiesner T, Obenauf AC, Wackernagel W, Green G, Bouvier N, Sozen MM, Baimukanova G, Roy R, Heguy A, Dolgalev I, Khanin R, Busam K, Speicher MR, O'Brien J, Bastian BC (2010). "Mutations in GNA11 in uveal melanoma". N Engl J Med. 363 (23): 2191–99. doi:10.1056/NEJMoa1000584. PMC 3107972. PMID 21083380.
- ↑ Onken MD, Worley LA, Long MD, Duan S, Council ML, Bowcock AM, Harbour JW (2008). "Oncogenic mutations in GNAQ occur early in uveal melanoma". Invest Ophthalmol Vis Sci. 49 (12): 5230–34. doi:10.1167/iovs.08-2145. PMC 2634606. PMID 18719078.
- ↑ 16.0 16.1 Harbour JW, Onken MD, Roberson ED, Duan S, Cao L, Worley LA, Council ML, Matatall KA, Helms C, Bowcock AM (2010). "Frequent mutation of BAP1 in metastasizing uveal melanomas". Science. 330 (6009): 1410–13. Bibcode:2010Sci...330.1410H. doi:10.1126/science.1194472. PMC 3087380. PMID 21051595.
- ↑ Stang A, Schmidt-Pokrzywniak A, Lash TL, Lommatzsch PK, Taubert G, Bornfeld N, Jöckel KH (2009). "Mobile phone use and risk of uveal melanoma: results of the risk factors for uveal melanoma case-control study". J Natl Cancer Inst. 101 (2): 120–23. doi:10.1093/jnci/djn441. PMC 2639317. PMID 19141780.
- ↑ Garcia-Valenzuela, Enrique (3 February 2022). "Ciliary Body Melanoma: Patient history". Archived from the original on 2 October 2021. Retrieved 22 August 2022.
- ↑ 19.0 19.1 19.2 Garcia-Valenzuela, Enrique (3 February 2022). "Ciliary Body Melanoma: Etiology". Archived from the original on 2 October 2021. Retrieved 22 August 2022.
- ↑ 20.0 20.1 Garcia-Valenzuela, Enrique (3 February 2022). "Ciliary Body Melanoma: Physical examination". Archived from the original on 2 October 2021. Retrieved 22 August 2022.
- ↑ Garcia-Valenzuela, Enrique (3 February 2022). "Ciliary Body Melanoma: epidemiology". Archived from the original on 2 October 2021. Retrieved 22 August 2022.
- ↑ Chien JL, Sioufi K, Surakiatchanukul T, Shields JA, Shields CL (May 2017). "Choroidal nevus: a review of prevalence, features, genetics, risks, and outcomes". Curr Opin Ophthalmol. 28 (3): 228–37. doi:10.1097/ICU.0000000000000361. PMID 28141766. S2CID 19367181.
- ↑ "Classification and Stage Information for Intraocular (Uveal) Melanoma". National Cancer Institute. 1980-01-01. Archived from the original on 2013-07-07. Retrieved 2013-07-04.
- ↑ 24.0 24.1 24.2 Kumar, Vinay (2009). "Uvea: Neoplasms". Robbins and Cotran Pathologic Basis of Disease, Professional Edition (8th ed.). Philadelphia, PA: Elsevier. ISBN 978-1-4377-0792-2.
- ↑ "Prognostic Indicators". Ocular Melanoma Foundation. 2012. Archived from the original on 2012-03-27. Retrieved 2013-02-02.
- ↑ Spagnolo, Francesco; Graziano Caltabiano; Paola Queirolo (January 2012). "Uveal melanoma". Cancer Treatment Reviews. 38 (5): 549–53. doi:10.1016/j.ctrv.2012.01.002. PMID 22270078. Archived from the original on 8 November 2021. Retrieved 24 November 2013.
- ↑ Kolandjian, NA; Wei C; Patel SP; Richard JL; Dett T; Papadopoulos NE; Bedikian AY (October 2013). "Delayed systemic recurrence of uveal melanoma". American Journal of Clinical Oncology. 36 (5): 443–49. doi:10.1097/COC.0b013e3182546a6b. PMC 4574291. PMID 22706174.
- ↑ Valpione Sara; et al. (Mar 2015). "Development and external validation of a prognostic nomogram for metastatic uveal melanoma". PLOS ONE. 10 (3): e0120181. Bibcode:2015PLoSO..1020181V. doi:10.1371/journal.pone.0120181. PMC 4363319. PMID 25780931.
- ↑ Kivelä, Tero (2012). "Diagnosis of uveal melanoma". Developments in Ophthalmology. 49: 1–15. doi:10.1159/000330613. ISSN 1662-2790. Archived from the original on 8 November 2021. Retrieved 7 November 2021.
- ↑ "The Collaborative Ocular Melanoma Study: An Overview". Medscape. Archived from the original on 27 December 2016. Retrieved 27 December 2016.
- ↑ Robertson AG, Shih J, Yau C, Gibb EA, Oba J, Mungall KL, Hess JM, Uzunangelov V, Walter V, Danilova L, Lichtenberg TM, Kucherlapati M, Kimes PK, Tang M, Penson A, Babur O, Akbani R, Bristow CA, Hoadley KA, Iype L, Chang MT, Cherniack AD, Benz C, Mills GB, Verhaak RG, Griewank KG, Felau I, Zenklusen JC, Gershenwald JE, Schoenfield L, Lazar AJ, Rahman MH, Roman S, Stern MH, Cebulla CM, Williams MD, Jager MJ, Coupland SE, Esmaeli B, Kandoth C, Woodman SE (2017). "Integrative Analysis Identifies Four Molecular and Clinical Subsets in Uveal Melanoma". Cancer Cell. 33 (1): 204–220. doi:10.1016/j.ccell.2017.12.013. PMID 29316429.
- ↑ Sacco JJ, Kalirai H, Kenyani J, Figueiredo CR, Coulson JM, Coupland SE (2018). "Recent breakthroughs in metastatic uveal melanoma: a cause for optimism?". Future Oncology. 14 (14): 1335–1338. doi:10.2217/fon-2018-0116. PMID 29741103.
- ↑ Augsburger JJ, Gamel JW (1990). "Clinical prognostic factors in patients with posterior uveal malignant melanoma". Cancer. 66 (7): 1596–600. doi:10.1002/1097-0142(19901001)66:7<1596::AID-CNCR2820660726>3.0.CO;2-6. PMID 2208011.
- ↑ "General Information About Intraocular (Uveal) Melanoma". National Institutes of Health. 1980-01-01. Archived from the original on 2013-12-03. Retrieved 26 November 2013.
- ↑ Pardo M, Dwek RA, Zitzmann N (2007). "Proteomics in uveal melanoma research: opportunities and challenges in biomarker discovery". Expert Rev Proteomics. 4 (2): 273–86. doi:10.1586/14789450.4.2.273. PMID 17425462. S2CID 7269454.
- ↑ Prescher G, Bornfeld N, Hirche H, Horsthemke B, Jöckel KH, Becher R (1996). "Prognostic implications of monosomy 3 in uveal melanoma". Lancet. 347 (9010): 1222–25. doi:10.1016/S0140-6736(96)90736-9. PMID 8622452. S2CID 44328116.
- ↑ Damato BE, Dopierala J, Klaasen A, van Dijk M, Sibbring J, Coupland S (2009). "Multiplex Ligation-Dependent Probe Amplification of Uveal Melanoma: Correlation with Metastatic Death". Invest Ophthalmol Vis Sci. 50 (7): 3048–55. doi:10.1167/iovs.08-3165. PMID 19182252.
- ↑ White VA, McNeil BK, Horsman DE (1998). "Acquired homozygosity (isodisomy) of chromosome 3 in uveal melanoma". Cancer Genet Cytogenet. 102 (1): 40–45. doi:10.1016/S0165-4608(97)00290-2. PMID 9530338.
- ↑ Onken MD, Worley LA, Person E, Char DH, Bowcock AM, Harbour JW (2007). "Loss of heterozygosity of chromosome 3 detected with single nucleotide polymorphisms is superior to monosomy 3 for predicting metastasis in uveal melanoma". Clin Cancer Res. 13 (10): 2923–37. doi:10.1158/1078-0432.CCR-06-2383. PMID 17504992.
- ↑ Tschentscher F, Hüsing J, Hölter T, Kruse E, Dresen IG, Jöckel KH, Anastassiou G, Schilling H, Bornfeld N, Horsthemke B, Lohmann DR, Zeschnigk M (2003). "Tumor classification based on gene expression profiling shows that uveal melanomas with and without monosomy 3 represent two distinct entities". Cancer Res. 63 (10): 2578–84. PMID 12750282.
- ↑ Onken MD, Worley LA, Ehlers JP, Harbour JW (2004). "Gene expression profiling in uveal melanoma reveals two molecular classes and predicts metastatic death". Cancer Res. 64 (20): 7205–09. doi:10.1158/0008-5472.CAN-04-1750. PMC 5407684. PMID 15492234.
- ↑ Petrausch U, Martus P, Tönnies H, Bechrakis NE, Lenze D, Wansel S, Hummel M, Bornfeld N, Thiel E, Foerster MH, Keilholz U (2008). "Significance of gene expression analysis in uveal melanoma in comparison to standard risk factors for risk assessment of subsequent metastases". Eye. 22 (8): 997–1007. doi:10.1038/sj.eye.6702779. PMID 17384575.
- ↑ van Gils W, Lodder EM, Mensink HW, Kiliç E, Naus NC, Brüggenwirth HT, van Ijcken W, Paridaens D, Luyten GP, de Klein A (2008). "Gene expression profiling in uveal melanoma: two regions on 3p related to prognosis". Invest Ophthalmol Vis Sci. 49 (10): 4254–62. doi:10.1167/iovs.08-2033. PMID 18552379.
- ↑ Worley LA, Onken MD, Person E, Robirds D, Branson J, Char DH, Perry A, Harbour JW (2007). "Transcriptomic versus chromosomal prognostic markers and clinical outcome in uveal melanoma". Clin Cancer Res. 13 (5): 1466–71. doi:10.1158/1078-0432.CCR-06-2401. PMID 17332290.
- ↑ "MRF CURE OM". Melanoma Research Foundation. Archived from the original on 18 April 2012. Retrieved 30 March 2012.
- ↑ Kaliki S, Shields CL (2017). "Uveal melanoma: relatively rare but deadly cancer". Eye (Lond). 31 (2): 241–57. doi:10.1038/eye.2016.275. PMC 5306463. PMID 27911450.
- ↑ "Archive copy". Archived from the original on 2021-07-31. Retrieved 2021-10-25.
{{cite web}}: CS1 maint: archived copy as title (link) - ↑ "What is ocular melanoma? Medical mystery shines light on rare eye cancer". Archived from the original on 2020-11-09. Retrieved 2021-10-25.
- ↑ Rosenberg, Eli (30 April 2018). "A rare eye cancer showed up in three friends. Doctors want to know if the cases are connected". Washington Post. Archived from the original on 16 April 2019. Retrieved 25 October 2021.
- ↑ Masoomian B, Shields JA, Shields CL (2018) Overview of BAP1 cancer predisposition syndrome and the relationship to uveal melanoma. J Curr Ophthalmol 30(2):102-109
- ↑ Kivelä T (Aug 2017). "The first description of the complete natural history of uveal melanoma by two Scottish surgeons, Allan Burns and James Wardrop". Acta Ophthalmol. 95 (2): 203–214. doi:10.1111/aos.13535. PMID 28834323.
External links
| Classification | |
|---|---|
| External resources |
- National Organization for Rare Diseases - Ocular Melanoma Archived 2021-05-24 at the Wayback Machine
- Cancer Research UK Archived 2019-09-21 at the Wayback Machine
- National Cancer Institute - Intraocular (Eye) Melanoma—Patient Version Archived 2021-10-29 at the Wayback Machine
- Ocular Melanoma Foundation Archived 2021-10-21 at the Wayback Machine
- A Cure In Sight Archived 2019-01-22 at the Wayback Machine