Neonatal jaundice
| Neonatal jaundice | |
|---|---|
| Other names: Neonatal hyperbilirubinemia, neonatal icterus, jaundice in newborns | |
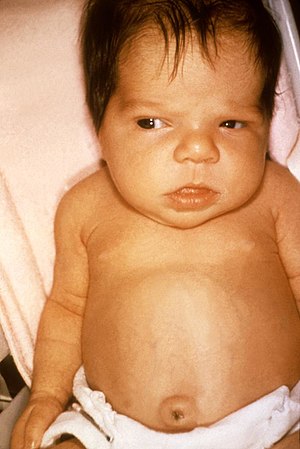 | |
| Jaundice in a newborn | |
| Specialty | Pediatrics |
| Symptoms | Yellowish discoloration of the skin and white part of the eyes[1] |
| Complications | Seizures, cerebral palsy, kernicterus[1] |
| Usual onset | Newborns[1] |
| Types | Physiologic, pathologic[1] |
| Causes | Red blood cell breakdown, liver disease, infection, hypothyroidism, metabolic disorders[2][1] |
| Diagnostic method | Based on symptoms, confirmed by bilirubin[1] |
| Treatment | More frequent feeding, phototherapy, exchange transfusions[1] |
| Frequency | >50% of babies[1] |
Neonatal jaundice is a yellowish discoloration of the white part of the eyes and skin in a newborn baby due to high bilirubin levels.[1] Other symptoms may include excess sleepiness or poor feeding.[1] Complications may include seizures, cerebral palsy, or kernicterus.[1]
In many cases there is no specific underlying disorder (physiologic).[2] In other cases it results from red blood cell breakdown, liver disease, infection, hypothyroidism, or metabolic disorders (pathologic).[2][1] A bilirubin level more than 34 μmol/l (2 mg/dL) may be visible.[1] Concerns, in otherwise healthy babies, occur when levels are greater than 308 μmol/L (18 mg/dL), jaundice is noticed in the first day of life, there is a rapid rise in levels, jaundice lasts more than two weeks, or the baby appears unwell.[1] In those with concerning findings further investigations to determine the underlying cause are recommended.[1]
The need for treatment depends on bilirubin levels, the age of the child, and the underlying cause.[1][3] Treatments may include more frequent feeding, phototherapy, or exchange transfusions.[1] In those who are born early more aggressive treatment tends to be required.[1] Physiologic jaundice generally lasts less than seven days.[1] The condition affects over half of babies in the first week of life.[1] Of babies that are born early about 80% are affected.[2] Globally over 100,000 late-preterm and term babies die each year as a result of jaundice.[4]
Signs and symptoms
The primary symptom is yellowish discoloration of the white part of the eyes and skin in a newborn baby.[1] Other symptoms may include excess sleepiness or poor feeding.[1]
A bilirubin level more than 34 μmol/l (2 mg/dL) may be visible.[1] For the feet to be affected level generally must be over 255 μmol/l (15 mg/dL).[1]
Complications
Prolonged hyperbilirubinemia (severe jaundice) can result in chronic bilirubin encephalopathy (kernicterus).[5][6] Quick and accurate treatment of neonatal jaundice helps to reduce the risk of neonates developing kernicterus.[7]
Infants with kernicterus may have a fever[8] or seizures.[9] High pitched crying is an effect of kernicterus.
Exchange transfusions performed to lower high bilirubin levels are an aggressive treatment.[10]
Causes
In newborns, jaundice tends to develop because of two factors—the breakdown of fetal hemoglobin as it is replaced with adult hemoglobin and the relatively immature metabolic pathways of the liver, which are unable to conjugate and so excrete bilirubin as quickly as an adult. This causes an accumulation of bilirubin in the blood (hyperbilirubinemia), leading to the symptoms of jaundice.
If the neonatal jaundice is not resolved with simple phototherapy, other causes such as biliary atresia, Progressive familial intrahepatic cholestasis, bile duct paucity, Alagille syndrome, alpha 1-antitrypsin deficiency, and other pediatric liver diseases should be considered. The evaluation for these will include blood work and a variety of diagnostic tests. Prolonged neonatal jaundice is serious and should be followed up promptly.
Severe neonatal jaundice may indicate the presence of other conditions contributing to the elevated bilirubin levels, of which there are a large variety of possibilities (see below). These should be detected or excluded as part of the differential diagnosis to prevent the development of complications. They can be grouped into the following categories:
| Neonatal jaundice | |||||||||||||||||||||||||||||||||||||||||||||||
| Unconjugated bilirubin | Conjugated bilirubin | ||||||||||||||||||||||||||||||||||||||||||||||
| Pathologic | Physiological jaundice of neonates | Liver | Post-liver | ||||||||||||||||||||||||||||||||||||||||||||
| Hemolytic | Non-hemolytic | ||||||||||||||||||||||||||||||||||||||||||||||
| Intrinsic causes | Extrinsic causes | ||||||||||||||||||||||||||||||||||||||||||||||
Unconjugated
Hemolytic
Intrinsic causes of hemolysis
- Membrane conditions
- Enzyme conditions
- Glucose-6-phosphate dehydrogenase deficiency (also called G6PD deficiency)
- Pyruvate kinase deficiency
- Globin synthesis defect
- Sickle cell disease
- Alpha-thalassemia, e.g. HbH disease
Extrinsic causes of hemolysis
- Systemic conditions
- Alloimmunity (The neonatal or cord blood gives a positive direct Coombs test and the maternal blood gives a positive indirect Coombs test)
Non-hemolytic causes
- Breastfeeding jaundice
- Breast milk jaundice
- Cephalohematoma
- Polycythemia
- Urinary tract infection
- Sepsis
- Hypothyroidism
- Gilbert's syndrome
- Crigler–Najjar syndrome
- High GI obstruction (Pyloric stenosis, Bowel obstruction)
Conjugated (direct)
Liver causes
- Infections
- Metabolic
- Galactosemia
- Alpha 1-antitrypsin deficiency, which is commonly missed, and must be considered in DDx
- Cystic fibrosis
- Dubin–Johnson syndrome
- Rotor syndrome
- Drugs
- Total parenteral nutrition
- Idiopathic
Post-liver
- Biliary atresia or bile duct obstruction
Non-organic causes
Breastfeeding jaundice
Breastfeeding jaundice (or "lack of breastfeeding jaundice") is caused by insufficient breast milk intake,[12] resulting in inadequate quantities of bowel movements to remove bilirubin from the body. This leads to increased enterohepatic circulation, resulting in increased reabsorption of bilirubin from the intestines.[13] Usually occurring in the first week of life, most cases can be ameliorated by frequent breastfeeding sessions of sufficient duration to stimulate adequate milk production.[citation needed]
Breast milk jaundice
Whereas breastfeeding jaundice is a mechanical problem, breast milk jaundice is a biochemical occurrence and the higher bilirubin possibly acts as an antioxidant. Breast milk jaundice occurs later in the newborn period, with the bilirubin level usually peaking in the sixth to 14th days of life. This late-onset jaundice may develop in up to one third of healthy breastfed infants.[14]
- First, the gut is sterile at birth and normal gut flora takes time to establish. The bacteria in the adult gut convert conjugated bilirubin to stercobilinogen which is then oxidized to stercobilin and excreted in the stool. In the absence of sufficient bacteria, the bilirubin is de-conjugated by brush border β-glucuronidase and reabsorbed. This process of re-absorption is called enterohepatic circulation. It has been suggested that bilirubin uptake in the gut (enterohepatic circulation) is increased in breast fed babies, possibly as the result of increased levels of epidermal growth factor (EGF) in breast milk.[15] Breast milk also contains glucoronidase which will increase deconjugation and enterohepatic recirculation of bilirubin.
- Second, the breast-milk of some women contains a metabolite of progesterone called 3-alpha-20-beta pregnanediol. This substance inhibits the action of the enzyme uridine diphosphoglucuronic acid (UDPGA) glucuronyl transferase responsible for conjugation and subsequent excretion of bilirubin. In the newborn liver, activity of glucuronyl transferase is only at 0.1-1% of adult levels, so conjugation of bilirubin is already reduced. Further inhibition of bilirubin conjugation leads to increased levels of bilirubin in the blood.[16] However, these results have not been supported by subsequent studies.[17]
- Third, an enzyme in breast milk called lipoprotein lipase produces increased concentration of nonesterified free fatty acids that inhibit hepatic glucuronyl transferase, which again leads to decreased conjugation and subsequent excretion of bilirubin.[18]
Physiological jaundice
Most infants develop visible jaundice due to elevation of unconjugated bilirubin concentration during their first week. This is called physiological jaundice. This pattern of hyperbilirubinemia has been classified into two functionally distinct periods.[citation needed]
- Phase one
- Term infants - jaundice lasts for about 10 days with a rapid rise of serum bilirubin up to 204 μmol/l (12 mg/dL).
- Preterm infants - jaundice lasts for about two weeks, with a rapid rise of serum bilirubin up to 255 μmol/l (15 mg/dL).
- Phase two - bilirubin levels decline to about 34 μmol/l (2 mg/dL) for two weeks, eventually mimicking adult values.
- Preterm infants - phase two can last more than one month.
- Exclusively breastfed infants - phase two can last more than one month.
Mechanisms involved in physiological jaundice include:
- Relatively low activity of the enzyme glucuronosyltransferase which normally converts unconjugated bilirubin to conjugated bilirubin that can be excreted into the gastrointestinal tract.[19] Before birth, this enzyme is actively down-regulated, since bilirubin needs to remain unconjugated in order to cross the placenta to avoid being accumulated in the fetus.[20] After birth, it takes some time for this enzyme to gain function.
- Shorter life span of fetal red blood cells,[19] being approximately 80 to 90 days in a full term infant,[21] compared to 100 to 120 days in adults.
- Relatively low conversion of bilirubin to urobilinogen by the intestinal flora, resulting in relatively high absorption of bilirubin back into the circulation.[19]
Diagnosis
Diagnosis is often by measuring the serum bilirubin level in the blood.[3] In those who are born after 35 weeks and are more than a day old transcutaneous bilirubinometer may also be used.[3] The use of an icterometer, a piece of transparent plastic painted in five transverse strips of graded yellow lines, is not recommended.[3]
Transcutaneous bilirubinometer
This is hand held, portable and rechargeable but expensive. When pressure is applied to the photoprobe, a xenon tube generates a strobe light, and this light passes through the subcutaneous tissue. The reflected light returns through the second fiber optic bundle to the spectrophotometric module. The intensity of the yellow color in this light, after correcting for the hemoglobin, is measured and instantly displayed in arbitrary units.[citation needed]
Pathological jaundice
Any of the following features suggests pathological jaundice:
- Clinical jaundice appearing in the first 24 hours or greater than 14 days of life.
- Increases in the level of total bilirubin by more than 8.5 μmol/l (0.5 mg/dL) per hour or (85 μmol/l) 5 mg/dL per 24 hours.
- Total bilirubin more than 331.5 μmol/l (19.5 mg/dL) (hyperbilirubinemia).
- Direct bilirubin more than 34 μmol/l (2.0 mg/dL).
The signs which help detect pathological jaundice are the presence of intrauterine growth restriction, stigma of intrauterine infections (e.g. cataracts, small head, and enlargement of the liver and spleen), cephalohematoma, bruising, signs of bleeding in the brain's ventricles. History of illness is noteworthy. Family history of jaundice and anemia, family history of neonatal or early infant death due to liver disease, maternal illness suggestive of viral infection (fever, rash or lymphadenopathy), maternal drugs (e.g. sulphonamides, anti-malarials causing red blood cell destruction in G6PD deficiency) are suggestive of pathological jaundice in neonates.[citation needed]
Treatment
The bilirubin levels for initiative of phototherapy varies depends on the age and health status of the newborn. However, any newborn with a total serum bilirubin greater than 359 μmol/l ( 21 mg/dL) should receive phototherapy.[22]
Phototherapy
Babies with neonatal jaundice may be treated with colored light called phototherapy, which works by changing trans-bilirubin into the water-soluble cis-bilirubin isomer.[23][24][25]: 2533
The phototherapy involved is not ultraviolet light therapy but rather a specific frequency of blue light. The light can be applied with overhead lamps, which means that the baby's eyes need to be covered, or with a device called a biliblanket, which sits under the baby's clothing close to its skin.[24]
The use of phototherapy was first discovered, accidentally, at Rochford Hospital in Essex, England, when a nurse, Sister Jean Ward, noticed that babies exposed to sunlight had reduced jaundice, and a pathologist, Dr. Perryman, who noticed that a vial of blood left in the sun had turned green. Drs Cremer, Richards and Dobbs put together these observations,[26] leading to a landmark randomized clinical trial which was published in Pediatrics in 1968; it took another ten years for the practice to become established.[24][27] Massage therapy could be useful in addition to phototherapy in order to reduce the phototherapy duration. However, it does not appear to reduce the requirement for phototherapy in the treatment of neonatal jaundice.[28]
There is currently no reliable evidence about whether home-based or hospital-based phototherapy is more effective for full term infants with jaundice.[29]
Exchange transfusions
Much like with phototherapy the level at which exchange transfusion should occur depends on the health status and age of the newborn. It should however be used for any newborn with a total serum bilirubin of greater than 428 μmol/l ( 25 mg/dL ).[22][25]: 2533
Alternative therapy
Homeopathy, acupuncture, and traditional Chinese medicine do not work and should not be used.[3]
Research
Penicillamine was studied in the 1970s in hyperbilirubinemia due to ABO hemolytic disease.[30] While tin mesoporphyrin IX may decrease bilirubin such use is not recommended in babies.[30] Preclinical studies have looked at minocycline to help prevent neurotoxicity.[30] Clofibrate may decrease the duration of phototherapy.[30] Evidence as of 2012 however is insufficient to recommend its use.[31]
References
- ↑ 1.00 1.01 1.02 1.03 1.04 1.05 1.06 1.07 1.08 1.09 1.10 1.11 1.12 1.13 1.14 1.15 1.16 1.17 1.18 1.19 1.20 1.21 1.22 1.23 "Neonatal Hyperbilirubinemia". Merck Manuals Professional Edition. August 2015. Archived from the original on 2 December 2016. Retrieved 11 December 2017.
- ↑ 2.0 2.1 2.2 2.3 "Jaundice in newborn babies under 28 days | Guidance and guidelines". NICE. October 2016. Archived from the original on 20 August 2019. Retrieved 11 December 2017.
- ↑ 3.0 3.1 3.2 3.3 3.4 "Jaundice in newborn babies under 28 days". NICE. October 2016. Archived from the original on 22 August 2019. Retrieved 11 December 2017.
- ↑ Olusanya, BO; Teeple, S; Kassebaum, NJ (February 2018). "The Contribution of Neonatal Jaundice to Global Child Mortality: Findings From the GBD 2016 Study". Pediatrics. 141 (2): e20171471. doi:10.1542/peds.2017-1471. PMID 29305393.
- ↑ Juetschke, L.J. (2005, Mar/Apr). Kernicterus: still a concern. Neonatal Network, 24(2), 7-19, 59-62
- ↑ Colletti, JE; Kothari, S; Kothori, S; Jackson, DM; Kilgore, KP; Barringer, K (November 2007). "An emergency medicine approach to neonatal hyperbilirubinemia". Emerg. Med. Clin. North Am. 25 (4): 1117–35, vii. doi:10.1016/j.emc.2007.07.007. PMID 17950138.
- ↑ Watchko, JF (December 2006). "Hyperbilirubinemia and bilirubin toxicity in the late preterm infant" (PDF). Clin Perinatol. 33 (4): 839–52, abstract ix. doi:10.1016/j.clp.2006.09.002. PMID 17148008. Archived from the original (PDF) on 2017-08-08. Retrieved 2019-09-26.
- ↑ Shah, Z; Chawla, A; Patkar, D; Pungaonkar, S (March 2003). "MRI in kernicterus". Australas Radiol. 47 (1): 55–7. doi:10.1046/j.1440-1673.2003.00973.x. PMID 12581055.
- ↑ Malik, BA; Butt, MA; Shamoon, M; Tehseen, Z; Fatima, A; Hashmat, N (December 2005). "Seizures etiology in the newborn period". Journal of the College of Physicians and Surgeons--Pakistan. 15 (12): 786–90. PMID 16398972.
- ↑ Gómez, M; Bielza, C; Fernández del Pozo, JA; Ríos-Insua, S (2007). "A graphical decision-theoretic model for neonatal jaundice". Med Decis Making. 27 (3): 250–65. doi:10.1177/0272989X07300605. PMID 17545496. S2CID 14514900.
- ↑ 11.0 11.1 Click, R; Dahl-Smith, J; Fowler, L; DuBose, J; Deneau-Saxton, M; Herbert, J (January 2013). "An osteopathic approach to reduction of readmissions for neonatal jaundice". Osteopathic Family Physician. 5 (1): 17–23. doi:10.1016/j.osfp.2012.09.005. Archived from the original on 2013-04-15.
- ↑ Lynn C. Garfunkel; Jeffrey; Cynthia Christy (2002). Mosby's pediatric clinical advisor: instant diagnosis and treatment. Elsevier Health Sciences. pp. 200–. ISBN 978-0-323-01049-8. Archived from the original on 22 August 2019. Retrieved 14 June 2010.
- ↑ Leung, A. K.; Sauve, R. S. (1989-12-01). "Breastfeeding and breast milk jaundice". Journal of the Royal Society of Health. 109 (6): 213–217. doi:10.1177/146642408910900615. ISSN 0264-0325. PMID 2513410.
- ↑ Dennis, Maj Beth L.; Porter, Meredith L. (2002-02-15). "Hyperbilirubinemia in the Term Newborn". American Family Physician. 65 (4): 599–606. PMID 11871676. Archived from the original on 2021-02-24. Retrieved 2014-03-04.
- ↑ Kumral, A; Ozkan H; Duman N; et al. (2009). "Breast milk jaundice correlates with high levels of epidermal growth factor". Pediatr Res. 66 (2): 218–21. doi:10.1203/pdr.0b013e3181ac4a30. PMID 19617811.
- ↑ Arias, IM; Gartner LM; Seifter S; Furman M (1964). "Prolonged neonatal unconjugated hyperbilirubinemia associated with breast feeding and a steroid, pregnane-3(alpha), 20(beta)-diol in maternal milk that inhibits glucuronide formation in vitro". J Clin Invest. 43 (11): 2037–47. doi:10.1172/jci105078. PMC 441992. PMID 14228539.
- ↑ Murphy, J F; Hughes I; Verrier Jones ER; Gaskell S; Pike AW (1981). "Pregnanediols and breast-milk jaundice". Arch Dis Child. 56 (6): 474–76. doi:10.1136/adc.56.6.474. PMC 1627473. PMID 7259280.
- ↑ Poland, R L; Schultz GE; Gayatri G (1980). "High milk lipase activity associated with breastmilk jaundice". Pediatr Res. 14 (12): 1328–31. doi:10.1203/00006450-198012000-00011. PMID 6782543.
- ↑ 19.0 19.1 19.2 Page 45 in: Obstetrics & Gynaecology, by B. Jain, 2002. ISBN 8180562107, 9788180562105
- ↑ McDonagh, A. F. (2007). "Movement of Bilirubin and Bilirubin Conjugates Across the Placenta". Pediatrics. 119 (5): 1032–1033, author 1033 1033. doi:10.1542/peds.2006-3669. PMID 17473108. S2CID 21269991.
- ↑ Harrison, K. L. (1979). "Fetal Erythrocyte Lifespan". Journal of Paediatrics and Child Health. 15 (2): 96–97. doi:10.1111/j.1440-1754.1979.tb01197.x. PMID 485998.
- ↑ 22.0 22.1 American Academy of Pediatrics Subcommittee on Hyperbilirubinemia (July 2004). "Management of hyperbilirubinemia in the newborn infant 35 or more weeks of gestation". Pediatrics. 114 (1): 297–316. doi:10.1542/peds.114.1.297. PMID 15231951. Archived from the original on 2021-05-31. Retrieved 2008-12-13.
- ↑ Stokowski LA (December 2006). "Fundamentals of phototherapy for neonatal jaundice". Adv Neonatal Care. 6 (6): 303–12. doi:10.1016/j.adnc.2006.08.004. PMID 17208161. S2CID 31233601.
- ↑ 24.0 24.1 24.2 Jones, Clay (9 May 2014). "Separating Fact from Fiction in the Not-So-Normal Newborn Nursery: Newborn Jaundice". Science-Based Medicine. Archived from the original on 7 March 2021. Retrieved 9 September 2017.
- ↑ 25.0 25.1 Wolkoff, Allan W. (2012). "Chapter 303: The Hyperbilirubinemias". In Longo, Dan L.; Kasper, Dennis L. (eds.). Harrison's principles of internal medicine (18th ed.). New York: McGraw-Hill. ISBN 978-0071748896.
- ↑ CREMER, RJ; PERRYMAN, PW; RICHARDS, DH (24 May 1958). "Influence of light on the hyperbilirubinaemia of infants". Lancet. 1 (7030): 1094–7. doi:10.1016/s0140-6736(58)91849-x. PMID 13550936.
- ↑ Lucey, J; Ferriero, M; Hewitt, J (June 1968). "Prevention of hyperbilirubinemia of prematurity by phototherapy". Pediatrics. 41 (6): 1047–54. PMID 5652916.
- ↑ Abdellatif, Mohammed (February 2020). "Massage therapy for the treatment of neonatal jaundice: A systematic review and network meta-analysis". Journal of Neonatal Nursing. 26 (1): 17–24. doi:10.1016/j.jnn.2019.09.002.
- ↑ Malwade, US; Jardine, LA (10 June 2014). "Home- versus hospital-based phototherapy for the treatment of non-haemolytic jaundice in infants at more than 37 weeks' gestation". The Cochrane Database of Systematic Reviews (6): CD010212. doi:10.1002/14651858.CD010212.pub2. PMID 24913724.
- ↑ 30.0 30.1 30.2 30.3 Mancuso, Cesare (May 15, 2017). "Bilirubin and brain: A pharmacological approach". Neuropharmacology. 118: 113–123. doi:10.1016/j.neuropharm.2017.03.013. PMID 28315352. S2CID 21001248.
- ↑ Gholitabar, M; McGuire, H; Rennie, J; Manning, D; Lai, R (12 December 2012). "Clofibrate in combination with phototherapy for unconjugated neonatal hyperbilirubinaemia". The Cochrane Database of Systematic Reviews. 12: CD009017. doi:10.1002/14651858.CD009017.pub2. PMC 6426433. PMID 23235669.
External links
- American Academy of Pediatrics has issued guidelines for managing this disease, which can be obtained for free.
- National Institute for Health and Care Excellence (NICE) has issued guidelines for the recognition and treatment of neonatal jaundice in the United Kingdom.
| Classification | |
|---|---|
| External resources |
- Pages with script errors
- All articles with unsourced statements
- Articles with unsourced statements from July 2020
- Articles with invalid date parameter in template
- Articles with unsourced statements from June 2020
- Articles with unsourced statements from November 2019
- Neonatology
- Hepatology
- Haemorrhagic and haematological disorders of fetus and newborn
- RTT