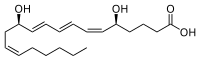
Leukotriene
Leukotrienes are a family of eicosanoid inflammatory mediators produced in leukocytes by the oxidation of arachidonic acid (AA) and the essential fatty acid eicosapentaenoic acid (EPA) by the enzyme arachidonate 5-lipoxygenase.[1][2][3]
Leukotrienes use lipid signaling to convey information to either the cell producing them (autocrine signaling) or neighboring cells (paracrine signaling) in order to regulate immune responses. The production of leukotrienes is usually accompanied by the production of histamine and prostaglandins, which also act as inflammatory mediators.[4]
One of their roles (specifically, leukotriene D4) is to trigger contractions in the smooth muscles lining the bronchioles; their overproduction is a major cause of inflammation in asthma and allergic rhinitis.[5] Leukotriene antagonists are used to treat these disorders by inhibiting the production or activity of leukotrienes.[6]
History
The name leukotriene, introduced by Swedish biochemist Bengt Samuelsson in 1979, comes from the words leukocyte and triene (indicating the compound's three conjugated double bonds). What would be later named leukotriene C, "slow reaction smooth muscle-stimulating substance" (SRS) was originally described between 1938 and 1940 by Feldberg and Kellaway.[7][8][9] The researchers isolated SRS from lung tissue after a prolonged period following exposure to snake venom and histamine.[9]
Types
Cysteinyl leukotrienes
LTC4, LTD4, LTE4 and LTF4 are often called cysteinyl leukotrienes due to the presence of the amino acid cysteine in their structure. The cysteinyl leukotrienes make up the slow-reacting substance of anaphylaxis (SRS-A). LTF4, like LTD4, is a metabolite of LTC4, but, unlike LTD4, which lacks the glutamic residue of glutathione, LTF4 lacks the glycine residue of glutathione.[10]
LTB4
LTB4 is synthesized in vivo from LTA4 by the enzyme LTA4 hydrolase. Its primary function is to recruit neutrophils to areas of tissue damage, though it also helps promote the production of inflammatory cytokines by various immune cells. Drugs that block the actions of LTB4 have shown some efficacy in slowing the progression of neutrophil-mediated diseases.[11]
LTG4
There has also been postulated the existence of LTG4, a metabolite of LTE4 in which the cysteinyl moiety has been oxidized to an alpha-keto-acid (i.e.—the cysteine has been replaced by a pyruvate). Very little is known about this putative leukotriene.[citation needed]
LTB5
Leukotrienes originating from the omega-3 class eicosapentanoic acid (EPA) have diminished inflammatory effects. In human subjects whose diets have been supplemented with eicosapentaenoic acid, leukotrine B5, along with leukotrine B4, is produced by neutrophils.[12] LTB5 induces aggregation of rat neutrophils, chemokinesis of human polymorphonuclear neutrophils (PMN), lysosomal enzyme release from human PMN and potentiation of bradykinin-induced plasma exudation, although compared to LTB4, it has at least 30 times less potency.[13]
Biochemistry
Synthesis
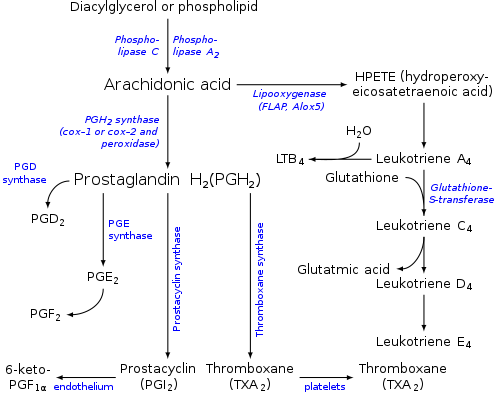
Leukotrienes are synthesized in the cell from arachidonic acid by arachidonate 5-lipoxygenase. The catalytic mechanism involves the insertion of an oxygen moiety at a specific position in the arachidonic acid backbone.[citation needed]
The lipoxygenase pathway is active in leukocytes and other immunocompetent cells, including mast cells, eosinophils, neutrophils, monocytes, and basophils. When such cells are activated, arachidonic acid is liberated from cell membrane phospholipids by phospholipase A2, and donated by the 5-lipoxygenase-activating protein (FLAP) to 5-lipoxygenase.[citation needed]
5-Lipoxygenase (5-LO) uses FLAP to convert arachidonic acid into 5-hydroperoxyeicosatetraenoic acid (5-HPETE), which spontaneously reduces to 5-hydroxyeicosatetraenoic acid (5-HETE). The enzyme 5-LO acts again on 5-HETE to convert it into leukotriene A4 (LTA4), an unstable epoxide. 5-HETE can be further metabolized to 5-oxo-ETE and 5-oxo-15-hydroxy-ETE, all of which have pro-inflammatory actions similar but not identical to those of LTB4 and mediated not by LTB4 receptors but rather by the OXE receptor (see 5-Hydroxyeicosatetraenoic acid and 5-Oxo-eicosatetraenoic acid).[14][15]
In cells equipped with LTA hydrolase, such as neutrophils and monocytes, LTA4 is converted to the dihydroxy acid leukotriene LTB4, which is a powerful chemoattractant for neutrophils acting at BLT1 and BLT2 receptors on the plasma membrane of these cells.[citation needed]
In cells that express LTC4 synthase, such as mast cells and eosinophils, LTA4 is conjugated with the tripeptide glutathione to form the first of the cysteinyl-leukotrienes, LTC4. Outside the cell, LTC4 can be converted by ubiquitous enzymes to form successively LTD4 and LTE4, which retain biological activity.[citation needed]
The cysteinyl-leukotrienes act at their cell-surface receptors CysLT1 and CysLT2 on target cells to contract bronchial and vascular smooth muscle, to increase permeability of small blood vessels, to enhance secretion of mucus in the airway and gut, and to recruit leukocytes to sites of inflammation.[citation needed]
Both LTB4 and the cysteinyl-leukotrienes (LTC4, LTD4, LTE4) are partly degraded in local tissues, and ultimately become inactive metabolites in the liver.[citation needed]
Function
Leukotrienes act principally on a subfamily of G protein-coupled receptors. They may also act upon peroxisome proliferator-activated receptors. Leukotrienes are involved in asthmatic and allergic reactions and act to sustain inflammatory reactions. Several leukotriene receptor antagonists such as montelukast and zafirlukast are used to treat asthma. Recent research points to a role of 5-lipoxygenase in cardiovascular and neuropsychiatric illnesses.[16]
Leukotrienes are very important agents in the inflammatory response. Some such as LTB4 have a chemotactic effect on migrating neutrophils, and as such help to bring the necessary cells to the tissue. Leukotrienes also have a powerful effect in bronchoconstriction and increase vascular permeability.[17]
Medicine

Leukotrienes in asthma
Leukotrienes contribute to the pathophysiology of asthma, especially in patients with aspirin-exacerbated respiratory disease (AERD), and cause or potentiate the following symptoms:[19]
- Airflow obstruction
- Increased secretion of mucus
- Mucosal accumulation
- Bronchoconstriction
- Infiltration of inflammatory cells in the airway wall
Role of cysteinyl leukotrienes
Cysteinyl leukotriene receptors CYSLTR1 and CYSLTR2 are present on mast cells, eosinophil, and endothelial cells. During cysteinyl leukotriene interaction, they can stimulate proinflammatory activities such as endothelial cell adherence and chemokine production by mast cells. As well as mediating inflammation, they induce asthma and other inflammatory disorders, thereby reducing the airflow to the alveoli. The levels of cysteinyl leukotrienes, along with 8-isoprostane, have been reported to be increased in the EBC of patients with asthma, correlating with disease severity.[20] Cysteinyl leukotrienes may also play a role in adverse drug reactions in general and in contrast medium induced adverse reactions in particular.[21]
In excess, the cysteinyl leukotrienes can induce anaphylactic shock.[22]
Leukotrienes in dementia
Leukotrienes are found to play an important role in the later stages of Alzheimer's disease and related dementias in studies with animals. In tau transgenic mice, which develop tau pathology, "zileuton, a drug that inhibits leukotriene formation by blocking the 5-lipoxygenase enzyme" was found to reverse memory loss.[23]
See also
- A chemical synthesis of Leukotriene A methyl ester
- Eoxins (14,15-leukotrienes)
References
- ↑ Loick, H.; Theissen, J. (1994). "Die Eicosanoide als Mediatoren beim ARDS" [Eicosanoids as mediators in ARDS]. Anästhesiologie, Intensivmedizin, Notfallmedizin, Schmerztherapie (in Deutsch). 29 (1): 3–9. doi:10.1055/s-2007-996677. PMID 8142566.
- ↑ Salmon, John A; Higgs, Gerald A (1987). "Prostaglandins and leukotrienes as inflammatory mediators". British Medical Bulletin. 43 (2): 285–96. doi:10.1093/oxfordjournals.bmb.a072183. PMID 2825898.
- ↑ O'Byrne, Paul M.; Israel, Elliot; Drazen, Jeffrey M. (1997). "Antileukotrienes in the treatment of asthma". Annals of Internal Medicine. 127 (6): 472–80. doi:10.7326/0003-4819-127-6-199709150-00009. PMID 9313005. S2CID 21480605.
- ↑ White, Martha (1999). "Mediators of inflammation and the inflammatory process". The Journal of Allergy and Clinical Immunology. 103 (3 Pt 2): S378-81. doi:10.1016/S0091-6749(99)70215-0. PMID 10069896. Retrieved 8 June 2019.
- ↑ Nelson, David L.; Cox, Michael M. (2008). "Leukotrienes". Lehninger Principles of Biochemistry (5th ed.). Macmillan. p. 359. ISBN 978-0-7167-7108-1.
- ↑ Scott JP, Peters-Golden M (September 2018). "Antileukotriene agents for the treatment of lung disease". Am. J. Respir. Crit. Care Med. 188 (5): 538–544. doi:10.1164/rccm.201301-0023PP. PMID 23822826.
- ↑ Feldberg, W.; Kellaway, C. H. (1938). "Liberation of histamine and formation of lysocithin-like substances by cobra venom". The Journal of Physiology. 94 (2): 187–226. doi:10.1113/jphysiol.1938.sp003674. PMC 1393616. PMID 16995038.
- ↑ Feldberg, W.; Holden, H. F.; Kellaway, C. H. (1938). "The formation of lysocithin and of a muscle-stimulating substance by snake venoms". The Journal of Physiology. 94 (2): 232–48. doi:10.1113/jphysiol.1938.sp003676. PMC 1393612. PMID 16995040.
- ↑ 9.0 9.1 Kellaway, C.H.; Trethewie, E.R. (1940). "The Liberation of a Slow-Reacting Smooth Muscle-Stimulating Substance in Anaphylaxis". Quarterly Journal of Experimental Physiology and Cognate Medical Sciences. 30 (2): 121–145. doi:10.1113/expphysiol.1940.sp000825. ISSN 1469-445X.
- ↑ internet Archived 2015-02-26 at the Wayback Machine checked April 24, 2012[full citation needed]
- ↑ Crooks, S.W; Stockley, R.A (1998). "Leukotriene B4". The International Journal of Biochemistry & Cell Biology. 30 (2): 173–8. doi:10.1016/S1357-2725(97)00123-4. PMID 9608670. S2CID 45983006.
- ↑ von Schacky, C; Fahrer, C; Fischer, S (October 1990). "Catabolism of leukotriene B5 in humans". Journal of Lipid Research. 31 (10): 1831–1838. doi:10.1016/S0022-2275(20)42326-0. PMID 1964169. Archived from the original on 16 January 2023. Retrieved 16 January 2023.
- ↑ Terano, Takashi; Salmon, John A.; Moncada, Salvador (1984). "Biosynthesis and biological activity of leukotriene B5". Prostaglandins. 27 (2): 217–32. doi:10.1016/0090-6980(84)90075-3. PMID 6326200.
- ↑ O'Flaherty, Joseph T.; Taylor, Jennifer S.; Thomas, Michael J. (1998). "Receptors for the 5-oxo class of eicosanoids in neutrophils". The Journal of Biological Chemistry. 273 (49): 32535–41. doi:10.1074/jbc.273.49.32535. PMID 9829988.
- ↑ Powell, William S.; Rokach, Joshua (2013). "The eosinophil chemoattractant 5-oxo-ETE and the OXE receptor". Progress in Lipid Research. 52 (4): 651–65. doi:10.1016/j.plipres.2013.09.001. PMC 5710732. PMID 24056189.
- ↑ Manev, Radmila; Manev, Hari (2004). "5-Lipoxygenase as a Putative Link Between Cardiovascular and Psychiatric Disorders". Critical Reviews in Neurobiology. 16 (1–2): 181–6. doi:10.1615/CritRevNeurobiol.v16.i12.190. PMID 15581413.
- ↑ Dahlén, Sven-Erik; Björk, Jakob; Hedqvist, Per; Arfors, Karl-E.; Hammarström, Sven; Lindgren, Jan-Åke; Samuelsson, Bengt (1981). "Leukotrienes promote plasma leakage and leukocyte adhesion in postcapillary venules: in vivo effects with relevance to the acute inflammatory response". Proceedings of the National Academy of Sciences. 78 (6): 3887–91. Bibcode:1981PNAS...78.3887D. doi:10.1073/pnas.78.6.3887. JSTOR 10943. PMC 319678. PMID 6267608.
- ↑ "Mechanism of action of leukotriene modifiers". PharmGKB.
- ↑ Berger, A. (1999). "Science commentary: What are leukotrienes and how do they work in asthma?". BMJ. 319 (7202): 90. doi:10.1136/bmj.319.7202.90. PMC 1116241. PMID 10398630.
- ↑ Samitas, Konstantinos; Chorianopoulos, Dimitrios; Vittorakis, Stelios; Zervas, Eleftherios; Economidou, Erasmia; Papatheodorou, George; Loukides, Stelios; Gaga, Mina (2009). "Exhaled cysteinyl-leukotrienes and 8-isoprostane in patients with asthma and their relation to clinical severity". Respiratory Medicine. 103 (5): 750–6. doi:10.1016/j.rmed.2008.11.009. PMID 19110408.
- ↑ Böhm, Ingrid; Speck, Ulrich; Schild, Hans (2005). "A possible role for cysteinyl-leukotrienes in non-ionic contrast media induced adverse reactions". European Journal of Radiology. 55 (3): 431–6. doi:10.1016/j.ejrad.2005.01.007. PMID 16129253.
- ↑ Brocklehurst, W. E. (1960). "The release of histamine and formation of a slow-reacting substance (SRS-A) during anaphylactic shock". The Journal of Physiology. 151 (3): 416–35. doi:10.1113/jphysiol.1960.sp006449. PMC 1363273. PMID 13804592.
- ↑ "Temple researchers reverse cognitive impairments in mice with dementia". Eurekalart!. June 8, 2018. Archived from the original on November 9, 2020. Retrieved March 30, 2024.
Further reading
- Bailey, J. Martyn (1985) Prostaglandins, leukotrienes, and lipoxins: biochemistry, mechanism of action, and clinical applications Plenum Press, New York, ISBN 0-306-41980-7
- Lipkowitz, Myron A. and Navarra, Tova (2001) The Encyclopedia of Allergies (2nd ed.) Facts on File, New York, p. 167, ISBN 0-8160-4404-X
- Samuelsson, Bengt (ed.) (2001) Advances in prostaglandin and leukotriene research: basic science and new clinical applications: 11th International Conference on Advances in Prostaglandin and Leukotriene Research: Basic Science and New Clinical Applications, Florence, Italy, June 4–8, 2000 Kluwer Academic Publishers, Dordrecht, ISBN 1-4020-0146-0
External links
- Leukotrienes at the US National Library of Medicine Medical Subject Headings (MeSH)
- CS1 Deutsch-language sources (de)
- Webarchive template wayback links
- All articles with incomplete citations
- Articles with incomplete citations from April 2016
- Articles with invalid date parameter in template
- Articles with hatnote templates targeting a nonexistent page
- All articles with unsourced statements
- Articles with unsourced statements from April 2016
- Eicosanoids
- Immunology
- Polyenes