Iohexol
 | |
| Names | |
|---|---|
| Trade names | Omnipaque, Hexopaque, others |
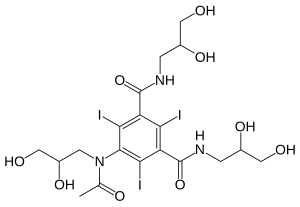
| Other names | 5-[N-(2,3-Dihydroxypropyl)acetamido]-2,4,6-triiodo-N,N'-bis(2,3-dihydroxypropyl)isophthalamide |
| |
| Clinical data | |
| Pregnancy category |
|
| Routes of use | intrathecal, intravascular, by mouth, intracavital, rectal |
| Defined daily dose | not established[1] |
| External links | |
| AHFS/Drugs.com | Micromedex Detailed Consumer Information |
| Legal | |
| License data | |
| Legal status |
|
| Pharmacokinetics | |
| Protein binding | Low |
| Metabolism | Nil |
| Elimination half-life | Variable |
| Excretion | Kidney, unchanged |
| Chemical and physical data | |
| Formula | C19H26I3N3O9 |
| Molar mass | 821.142 g·mol−1 |
| 3D model (JSmol) | |
| Melting point | 174 to 180 °C (345 to 356 °F) |
| |
| |
Iohexol, sold under the trade name Omnipaque among others, is a contrast agent used during X-rays.[2] This includes when visualizing arteries, veins, ventricles of the brain, the urinary system, and joints, as well as during computer tomography (CT scan).[2] It is given by mouth, injection into a vein, or into a body cavity.[3]
Side effects include vomiting, skin flushing, headache, itchiness, kidney problems, and low blood pressure.[2] Less commonly allergic reactions or seizures may occur.[2] Allergies to povidone-iodine or shellfish do not affect the risk of side effects more than other allergies.[4] Use in the later part of pregnancy may cause hypothyroidism in the baby.[5] Iohexol is an iodinated non-ionic radiocontrast agent.[2] It is in the low osmolar family.[6]
Iohexol was approved for medical use in 1985.[7] It is on the World Health Organization's List of Essential Medicines.[8] The wholesale cost in the developing world is about US$10.99 per 50 ml vial.[9] In the United States a dose costs US$50–100.[3]
Medical uses
Dosage
The defined daily dose is not established.[1]
Chemistry
The osmolality of iohexol ranges from 322 mOsm/kg—approximately 1.1 times that of blood plasma—to 844 mOsm/kg, almost three times that of blood.[10] Despite this difference, iohexol is still considered a low-osmolality contrast agent; the osmolality of older agents, such as diatrizoate, may be more than twice as high.[11]
Society and culture
Names
It is sold under the brand names Omnipaque[12] and Hexopaque. It is also sold as a density gradient medium under the names Accudenz, Histodenz and Nycodenz.[13][14]
Formulations
It is available in various concentrations, from 140 to 350 milligrams of iodine per milliliter.
References
- ↑ 1.0 1.1 "WHOCC - ATC/DDD Index". www.whocc.no. Archived from the original on 22 January 2021. Retrieved 15 September 2020.
- ↑ 2.0 2.1 2.2 2.3 2.4 World Health Organization (2009). Stuart MC, Kouimtzi M, Hill SR (eds.). WHO Model Formulary 2008. World Health Organization. pp. 317–8. hdl:10665/44053. ISBN 9789241547659.
- ↑ 3.0 3.1 Hamilton, Richart (2015). Tarascon Pocket Pharmacopoeia 2015 Deluxe Lab-Coat Edition. Jones & Bartlett Learning. p. 171. ISBN 9781284057560.
- ↑ ACR Manual on Contrast Media v10.3. 2017 (PDF). American College of Radiology. 2017. p. 6. ISBN 9781559030120. Archived (PDF) from the original on 1 January 2018. Retrieved 1 January 2018.
- ↑ Briggs, Gerald G.; Freeman, Roger K.; Yaffe, Sumner J. (2011). Drugs in Pregnancy and Lactation: A Reference Guide to Fetal and Neonatal Risk. Lippincott Williams & Wilkins. p. 761. ISBN 9781608317080. Archived from the original on 1 January 2017.
- ↑ Sutton, David; Young, Jeremy W. R. (2012). A Short Textbook of Clinical Imaging. Springer Science & Business Media. p. 235. ISBN 9781447117551. Archived from the original on 1 January 2017.
- ↑ Broe, Marc E. de; Porter, George A.; Bennett, William M.; Verpooten, G. A. (2013). Clinical Nephrotoxins: Renal Injury from Drugs and Chemicals. Springer Science & Business Media. p. 325. ISBN 9789401590884. Archived from the original on 1 January 2017.
- ↑ World Health Organization (2019). World Health Organization model list of essential medicines: 21st list 2019. Geneva: World Health Organization. hdl:10665/325771. WHO/MVP/EMP/IAU/2019.06. License: CC BY-NC-SA 3.0 IGO.
- ↑ "Iohexol". International Drug Price Indicator Guide. Archived from the original on 22 January 2018. Retrieved 8 December 2016.
- ↑ GE Healthcare (May 2006). "Omnipaque (Iohexol) injection. Product label". DailyMed. U.S. National Library of Medicine. Archived from the original on 7 July 2012. Retrieved 28 March 2007.
- ↑ Amersham Health (April 2006). "Hypaque (Diatrizoate Meglumine and Diatrizoate Sodium) injection, solution. Product label". DailyMed. U.S. National Library of Medicine. Archived from the original on 23 May 2011. Retrieved 29 March 2007.[dead link]
- ↑ "Omnipaque" (PDF). Ireland: Health Products Regulatory Authority. January 2018. Archived from the original on 28 August 2021. Retrieved 31 July 2020.
- ↑ "HistoDenz (D2158)" Archived 2015-11-20 at the Wayback Machine, product information sheet, Sigma-Aldrich. Accessed on line 19 November 2015.
- ↑ "Nycodenz®: A universal density gradient medium" Archived 2015-02-26 at the Wayback Machine, Axis-Shield Density Gradient Media. Accessed 19 November 2015.
External links
| External sites: | |
|---|---|
| Identifiers: |
|
- "Iohexol Injection, Oral, Rectal Advanced Patient Information". Drugs.com. 13 January 2019. Archived from the original on 3 February 2020. Retrieved 3 February 2020.
- Pages using duplicate arguments in template calls
- All articles with dead external links
- Articles with dead external links from February 2020
- Articles with invalid date parameter in template
- Webarchive template wayback links
- Use dmy dates from February 2020
- Drugs with non-standard legal status
- Chemical articles with unknown parameter in Infobox drug
- Chemical articles without CAS registry number
- Articles without EBI source
- Chemical pages without ChemSpiderID
- Chemical pages without DrugBank identifier
- Articles without KEGG source
- Articles without UNII source
- Drugs missing an ATC code
- Drugboxes which contain changes to verified fields
- Articles with changed EBI identifier
- Benzamides
- Iodoarenes
- Radiocontrast agents
- World Health Organization essential medicines
- RTT