Nickel(II) phosphate
(Redirected from Nickel phosphate)
| |

| |
| Names | |
|---|---|
| IUPAC name
Nickel(2+) diphosphate
| |
| Other names
Nickel(III) phosphate, nickel diphosphate
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.030.755 |
| EC Number |
|
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| Ni3(PO4)2 | |
| Molar mass | 366.022924 g/mol |
| Density | 4.38 g/cm3 |
Solubility product (Ksp)
|
4.74×10−32[1] |
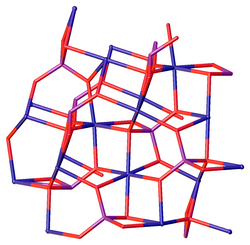
| Structure[2] | |
| Monoclinic, mP26 | |
| P21/c, No. 14 | |
a = 0.58273 nm, b = 0.46964 nm, c = 1.01059 nm α = 90°, β = 91.138°, γ = 90°
| |
| Hazards | |
| GHS labelling: | |
  
| |
| Danger | |
| H317, H334, H372, H410 | |
| P203, P260, P261, P264, P270, P272, P273, P280, P284, P302+P352, P304+P340, P318, P319, P321, P333+P317, P342+P316, P362+P364, P391, P405, P501 | |
| NFPA 704 (fire diamond) | |
| Safety data sheet (SDS) | www.fishersci.com |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Nickel(II) phosphate is an inorganic compound with the formula Ni3(PO4)2. It is a mint green paramagnetic solid that is insoluble in water.[3]
Hydrated nickel(II) phosphate
The hydrate Ni3(PO4)2·8(H2O) is a light green solid, which can be prepared by hydrothermal synthesis[4] and also occurs as the mineral arupite. It features octahedral Ni centers, which are bound to water and phosphate.[5]
References
Wikimedia Commons has media related to Nickel phosphate.
- ^ John Rumble (June 18, 2018). CRC Handbook of Chemistry and Physics (99 ed.). CRC Press. pp. 5–189. ISBN 978-1138561632.
- ^ McMurdie, Howard F.; Morris, Marlene C.; Evans, Eloise H.; Paretzkin, Boris; Wong-Ng, Winnie; Zhang, Yuming; Hubbard, Camden R. (2013). "Standard X-Ray Diffraction Powder Patterns from the JCPDS Research Associateship". Powder Diffraction. 2 (1): 41–52. Bibcode:1987PDiff...2...41M. doi:10.1017/S0885715600012239. S2CID 251057066.
- ^ Calvo, Crispin; Faggiani, Romolo (1975). "Structure of Nickel Orthophosphate". Canadian Journal of Chemistry. 53 (10): 1516–1520. doi:10.1139/v75-210.
- ^ Perry, Dale L. (18 May 2011). Handbook of Inorganic Compounds, Second Edition. CRC Press. p. 292. ISBN 978-1-4398-1462-8.
- ^ Shouwen, Jin; Wang, Daqi; Gao, Xinjun; Wen, Xianhong; Zhou, Jianzhong (2008). "Poly[octaaquadi-μ-phosphato-trinickel(II)]". Acta Crystallographica Section E. 64 (Pt 1): m259. Bibcode:2008AcCrE..64M.259S. doi:10.1107/S1600536807067050. PMC 2915172. PMID 21200596.
- ^ Ni, Bing; Liu, Huiling; Wang, Peng-Peng; He, Jie; Wang, Xun (2015). "General synthesis of inorganic single-walled nanotubes". Nature Communications. 6: 8756. Bibcode:2015NatCo...6.8756N. doi:10.1038/ncomms9756. PMC 4640082. PMID 26510862.
Categories:
- Articles without EBI source
- Articles without KEGG source
- ECHA InfoCard ID from Wikidata
- Chembox having GHS data
- Articles containing unverified chemical infoboxes
- Chembox image size set
- Articles with short description
- Short description matches Wikidata
- Commons category link is locally defined
- Phosphates
- Nickel compounds
