Legg–Calvé–Perthes disease
| Legg–Calvé–Perthes disease | |
|---|---|
| Other names: Legg–Calvé–Perthes syndrome, Perthes disease, Legg–Perthes disease, Calves disease | |
 | |
| Radiograph of a person with Legg–Calvé–Perthes disease | |
| Pronunciation | |
| Specialty | Orthopedics |
| Symptoms | Limp, hip pain[1] |
| Complications | Osteoarthritis[1] |
| Usual onset | 4 to 8 years[1] |
| Risk factors | Genetics, blood clotting problems, exposure to cigarette smoke, extreme motion at the hip[1] |
| Diagnostic method | X-rays[1] |
| Differential diagnosis | Sickle cell disease, lupus, chemotherapy, long-term corticosteroid use, multiple epiphyseal dysplasia[1] |
| Treatment | NSAIDs, decreasing physical activity, physical therapy, surgery[1] |
| Prognosis | Usually good[2] |
| Frequency | 1 in 1200[3] |
Legg–Calvé–Perthes disease (LCPD) is a disorder in children were the head of the femur dies due to a lack of blood flow (avascular necrosis).[1] The most common symptom is a gradual onset of a limp.[1] Ability to move the leg may be deceased.[1] Pain is variably present and may occur in the hip, knee, or bum.[1] Both side are affected in up to 15% of cases.[1] Complications may include osteoarthritis.[1]
The cause is unknown but it is believed to involved multiple factors.[1][2] Proposed risk factors include genetics, hyperactivity, blood clotting problems, exposure to cigarette smoke, and extreme motion at the hip.[1][2] Diagnosis is by X-rays after ruling out other causes of osteonecrosis of the femoral head.[1]
There is no cure.[1] Treatments may include NSAIDs, decreasing physical activity, and physical therapy.[1] If these measures are insufficient surgery may be recommended.[1] Outcomes are usually good.[2]
The condition affects 1 in 1,200 people (1 to 21 per 100,000 people per year).[1][3] Those between the ages of 4 and 8 years are most commonly affected, but it can start at 18 months until growth stops.[1] Males are more commonly affected.[1] The first detailed description was in 1910 by Arthur Legg, Jacques Calvé, and Georg Perthes.[1][2]
Signs and symptoms
The condition is most commonly found in children between the ages of 4-10. Common symptoms include pain in the hip, knee, or groin, exacerbated by hip or leg movement, especially internal hip rotation (with the knee flexed 90°, twisting the lower leg away from the center of the body). The range of motion is reduced, particularly in the abduction and internal rotation, and the patient presents with a limp. Pain is usually mild. Atrophy of thigh muscles may occur from disuse and inequality of leg length. In some cases, some activity can cause severe irritation or inflammation of the damaged area, including standing, walking, running, kneeling, or repeatedly stooping for an extended time. In cases exhibiting severe femoral osteonecrosis, pain is usually a chronic, throbbing sensation exacerbated by activity.[citation needed]
The first signs are complaints of soreness from the child, which are often dismissed as growing pains, and limping or other guarding of the joint, particularly when tired. The pain is usually in the hip, but can also be felt in the knee (referred pain). In some cases, pain is felt in the unaffected hip and leg,[citation needed] due to the children favoring their injured side and placing the majority of their weight on their "good" leg. It is predominantly a disease of boys (4:1 ratio). Perthes is generally diagnosed between 5 and 12 years of age, although it has been diagnosed as early as 18 months. Typically, the disease is only seen in one hip, but bilateral Perthes is seen in about 10% of children diagnosed.[4]
Cause
Perthes disease is a childhood hip disorder initiated by a disruption of blood flow to the head of the femur. Due to the lack of blood flow, the bone dies (osteonecrosis or avascular necrosis) and stops growing.
Legg believed the cause was impairment of blood supply to the femoral epiphysis, Calvé believed rickets, and Perthes deduced an infection possibly causing degenerative arthritis leads to LCP disease. Currently, a number of factors have been implicated, including heredity, trauma, endocrine dysfunction, inflammation, nutrition, and altered circulatory hemodynamics.[5] Risk factors are not limited to impaired and disproportionate growth, low birth weight, delayed skeletal maturity, short stature, systemic hormonal changes, and low economic index.[6] Although no one has identified the cause of Perthes disease, a reduction in blood flow to the joint is known.
The disease is theorized to include the artery of the ligmentum teres femoris being constricted or even blocked too early, not allowing for the time when the medial circumflex femoral artery takes over. The medial circumflex femoral artery is the principal source of blood supply to the femoral head. LCP disease is a restrictive vascular condition of idiopathic nature. Symptoms like femoral head disfigurement, flattening, and collapse occurs between ages four and ten, mostly male children of Caucasian descent. Children affected by LCP disease often display uneven gait and limited range of motion, and they experience mild to severe pain in the groin area.[7] For example, a child maybe six years old chronologically but may have grown only four years old in terms of bone maturity. The child may then engage in activities appropriate for a six-year-old child, but lacking the bone strength of an older child, these activities may lead to flattening or fracture of the hip joint. Genetics do not appear to be a determining factor, but a deficiency of blood factors with anticoagulant property used to disperse blood clots may lead to blockages in the vessels supplying the joint. A deficiency of protein C and protein S, which also act as blood anticoagulants, may also exist; if that were the case, their deficiency could cause clot formation in the ligamentum teres femoris artery and hinder blood supply to the femoral head. However, no evidence of this has been found; over the years, many theories have been published, but none has stood up to professional research.[citation needed]
Pathology
Over time, healing occurs by new blood vessels infiltrating the dead bone and removing the necrotic bone, which leads to a loss of bone mass and a weakening of the femoral head.[8] The bone loss leads to some degree of collapse and deformity of the femoral head and sometimes secondary changes to the shape of the hip socket.
It is also referred to as idiopathic avascular osteonecrosis of the capital femoral epiphysis of the femoral head since the cause of the interruption of the blood supply of the head of the femur in the hip joint is unknown.
Perthes can produce a permanent deformity of the femoral head, which increases the risk of developing osteoarthritis in adults. Perthes is a form of osteochondritis which only affects the hip, although other conditions of osteochondritis can affect elbows, knees, ankles, and feet. Bilateral Perthes, which means both hips are affected, should always be investigated thoroughly to rule out multiple epiphyseal dysplasia.
Diagnosis
X-rays of the hip may suggest or verify the diagnosis. X-rays usually demonstrate a flattened, and later fragmented femoral head. A bone scan or MRI may be useful in making the diagnosis in those cases where X-rays are inconclusive. Usually, plain radiographic changes are delayed six weeks or more from the clinical onset, so bone scintigraphy and MRI are done for early diagnosis. MRI results are more accurate, i.e., 97 to 99% against 88 to 93% in plain radiography. If MRI or bone scans are necessary, a positive diagnosis relies upon patchy areas of vascularity to the capital femoral epiphysis (the developing femoral head).[citation needed]
Treatment
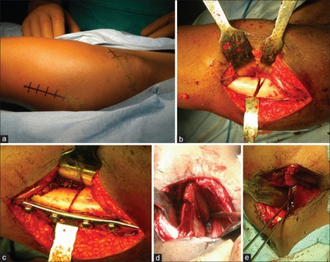
The goals of treatment are to decrease pain, reduce the loss of hip motion, and prevent or minimize permanent femoral head deformity so that the risk of developing a severe degenerative arthritis as adult can be reduced.[9] Assessment by a pediatric orthopaedic surgeon is recommended to evaluate risks and treatment options. Younger children have a better prognosis than older children.[10]
The treatment has historically centered on removing mechanical pressure from the joint until the disease has run its course. Options include traction (to separate the femur from the pelvis and reduce wear), braces (often for several months, with an average of 18 months) to restore range of motion, physiotherapy, and surgical intervention when necessary because of permanent joint damage. Custom orthotics can maintain activities of daily living. Patients can use overnight traction instead of walking devices or in combination.[11] These devices internally rotate the femoral head and abduct the leg(s) at 45°. Orthoses can start as proximal as the lumbar spine and extend the length of the limbs to the floor. Most functional bracing is achieved using a waist belt and thigh cuffs derived from the Scottish-Rite orthosis.[12] These devices are typically prescribed by a physician and implemented by an orthotist. Clinical results of the Scottish Rite orthosis have not been good according to some studies, and its use has gone out of favor. Many children, especially those with the onset of the disease before age 6, need no intervention at all and are asked to refrain from contact sports or games which impact the hip. For older children (onset of Perthes after age 6), the best treatment option remains unclear. Current treatment options for older children over age 8 include prolonged periods without weight-bearing, osteotomy (femoral, pelvic, or shelf), and the hip distraction method using an external fixator which relieves the hip from carrying the body's weight. An external fixator allows room for the top of the femur to regrow.
While running and high-impact sports are not recommended during treatment for Perthes disease, children can remain active through a variety of other activities that limit mechanical stress on the hip joint. Swimming is highly recommended, as it allows the exercise of the hip muscles with a full range of motion while reducing the stress to a minimum. Cycling is another good option as it also keeps stress to a minimum. Physiotherapy generally involves a series of daily exercises, with weekly meetings with a physiotherapist to monitor progress. These exercises focus on improving and maintaining a full range of motion of the femur within the hip socket. Performing these exercises during the healing process is essential to ensure that the femur and hip socket have a perfectly smooth interface. This will minimize the long-term effects of the disease. Use of bisphosphonate such as zoledronate or ibandronate is currently being investigated, but definite recommendations are not yet available.[13][14]
Perthes disease is self-limiting, but if the head of the femur is left deformed, long-term problems can occur. Treatment aims to minimize damage while the disease runs its course, not 'curing' the condition. It is recommended not to use steroids or alcohol as these reduce oxygen in the blood needed in the joint. As sufferers age, problems in the knee and back can arise secondary to abnormal posture and stride adopted to protect the affected joint. The condition is also linked to arthritis of the hip, though this appears not to be an inevitable consequence. Hip replacements are relatively common as the already damaged hip suffers routine wear; this varies by individual, but generally is required any time after age 50.[citation needed]
Prognosis

Children younger than 6 have the best prognosis since they have time for the dead bone to revascularize and remodel, with a good chance that the femoral head will recover and remain spherical after the resolution of the disease.[15] Children who have been diagnosed with Perthes' disease after the age of 10 are at a very high risk of developing osteoarthritis and coxa magna. When an LCP disease diagnosis occurs after age 8, a better outcome results with surgery rather than nonoperative treatments.[16] Shape of the femoral head at the time when Legg-Calvé Perthes disease heals is the most important determinant of risk for degenerative arthritis; hence, the shape of femoral head and congruence of the hip are most useful outcome measures.[7]
Epidemiology
Perthes' disease is one of the most common hip disorders in young children, occurring in roughly 5.5 of 100,000 children per year. The lifetime risk of a child developing the disease is about one per 1,200 individuals. Boys are affected about three to five times more often than girls. New cases of Perthes' disease rarely occur after age 14 years (if diagnosed after 14 years of age, then it is usually old disease from early in childhood or avascular necrosis from an alternative cause).[6] White northern Europeans appear to be affected more frequently than other races, though a paucity of reliable epidemiology exists in the Southern Hemisphere.[17] Children of sufferers of the disease themselves may have a very slightly increased risk, though it is unclear if this is because of a genetic predisposition, or a shared environmental factor. It is most commonly seen in persons aged three to 12 years, with a median of six years of age.[citation needed] The UK incidence rates show an intriguing pattern with low incidence rates in London, and a progressive increase in disease in more northerly areas (maximal in Scotland).[18] Some evidence suggests, at least in developed countries, more socioeconomically deprived communities have a greater risk of disease (a similar trend to diseases such as adult heart disease), though the reason for this remains unknown.[18][19][20][21][22][23] One possible explanation that has been considered is tobacco smoke exposure, though this is significantly confounded by the strong socioeconomic gradient common to both smoking and Perthes' disease.[6][24]
History
- 1897: The disease was first described by Karel Maydl (1853–1903).[25]
- 1909: Henning Waldenström (1877–1972) described the disease, attributing it to tuberculosis.[26]
- 1910: The disease was recognized as being unrelated to tuberculosis by three physicians working independently.[27] Legg–Calvé–Perthes is named after these three doctors:[27] Arthur Legg (1874–1939),[28] Jacques Calvé (1875–1954),[29] and Georg Perthes (1869–1927).[30]
Research
In the UK in 2016, a nationwide study began collecting details of every case of Perthes' disease as part of the British Orthopaedic Surgery Surveillance Study.[31] This aims to get core information about the disease, before starting extensive clinical trials about the best forms of treatment. Every hospital in England, Scotland, and Wales, which treats Perthes' disease, collect new cases. This study is supported by the Perthes' Association (UK). The study is nationally approved by the UK ethics committee and NHS research approval teams in England, Scotland, and Wales. The study is funded by the National Institute of Health Research and the Alder Hey Charity.
Dogs
LCP disease is an avascular necrosis of the femoral head in small-breed dogs, usually those weighing up to 25 lbs. LCP disease was first described in veterinary literature by Tutt in 1935.[32] Tutt described the disease as almost exactly what Waldenstromin (1909) described in humans.[33] Small breeds, particularly Toy Poodles, Yorkshire Terriers, Pugs, Jack Russell Terriers, West Highland White Terriers, and Dachshunds can be affected. No sex predilection is seen in the dog as contrasted to humans, in whom an 80% male incidence of the disease is evident. Like children, however, the condition is usually unilateral, with only about 10% to 15% incidence of bilateral disease. The age of onset varies between 4 and 12 months, with a peak incidence of around seven months.[34] The pathology of avascular necrosis followed by revascularization and bony remodeling of the femoral head in the dog certainly suggests a vascular etiology, though the cause of the condition is not completely understood.[35] Hip pain is usually seen by the age of 6 to 8 months.[36] The disease is bilateral in 12 to 16% of cases.[37] X-rays are necessary to make the diagnosis and show increased opacity and focal lysis in the head of the femur, and later in the disease, collapse, and fracture of the neck of the femur. The recommended treatment is surgical removal of the head of the femur, but conservative treatment (rest, exercise restriction, and pain medication) may be effective in a limited number of cases (less than 25%, according to some studies).[37] In dogs, the prognosis is excellent with surgery.[citation needed] Prednisolone tablets may also be helpful.
References
- ↑ 1.00 1.01 1.02 1.03 1.04 1.05 1.06 1.07 1.08 1.09 1.10 1.11 1.12 1.13 1.14 1.15 1.16 1.17 1.18 1.19 1.20 1.21 1.22 Karkenny, AJ; Tauberg, BM; Otsuka, NY (September 2018). "Pediatric Hip Disorders: Slipped Capital Femoral Epiphysis and Legg-Calvé-Perthes Disease". Pediatrics in review. 39 (9): 454–463. doi:10.1542/pir.2017-0197. PMID 30171056.
- ↑ 2.0 2.1 2.2 2.3 2.4 Leroux, J; Abu Amara, S; Lechevallier, J (February 2018). "Legg-Calvé-Perthes disease". Orthopaedics & traumatology, surgery & research : OTSR. 104 (1S): S107–S112. doi:10.1016/j.otsr.2017.04.012. PMID 29155310.
- ↑ 3.0 3.1 Mills, S; Burroughs, KE (January 2020). "Legg Calve Perthes Disease (Calves Disease)". PMID 30020602.
{{cite journal}}: Cite journal requires|journal=(help) - ↑ Legg-Calvé-Perthes Disease Imaging Archived 2011-05-14 at the Wayback Machine Author: Ali Nawaz Khan. Chief Editor: Felix S Chew. Updated: May 28, 2008
- ↑ Yochum, Rowe (1987). Essentials of Skeletal Radiology. p. 995. ISBN 0-683-09329-0.
- ↑ 6.0 6.1 6.2 Bahmanyar S, Montgomery SM, Weiss RJ, Ekbom A (August 2008). "Maternal smoking during pregnancy, other prenatal and perinatal factors, and the risk of Legg-Calvé-Perthes disease". Pediatrics. 122 (2): e459–64. doi:10.1542/peds.2008-0307. PMID 18625663. S2CID 12645569. Archived from the original on 2019-12-13. Retrieved 2012-02-03.
- ↑ 7.0 7.1 Kim HK (November 2010). "Legg-Calvé-Perthes disease". J Am Acad Orthop Surg. 18 (11): 676–86. doi:10.5435/00124635-201011000-00005. PMID 21041802. Archived from the original on 2019-12-13. Retrieved 2012-02-03.
- ↑ Kim HK (2012). "Pathophysiology and new strategies for the treatment of Legg-Calvé-Perthes disease". J Bone Joint Surg Am. 94 (7): 659–69. doi:10.2106/JBJS.J.01834. PMID 22488623.
- ↑ Kim, HK; JA Herring (July 2011). "Pathophysiology, classifications, and natural history of Perthes disease". Orthop Clin N Am. 42 (2): 285–95. doi:10.1016/J.OCL.2011.04.007. PMID 21742140.
- ↑ Joseph, B (July 2011). "Prognostic factors and outcome measures in Perthes disease". Orthop Clin N Am. 42 (3): 303–15. doi:10.1016/j.ocl.2011.03.004. PMID 21742142.
- ↑ "Archive copy". Archived from the original on 2019-10-02. Retrieved 2022-03-14.
{{cite web}}: CS1 maint: archived copy as title (link) - ↑ Katz, Jacob (1984). Legg–Calvé-Perthes-Disease. New York: Praeger.
- ↑ Young, ML; Little DG; Kim HK (Sep 2012). "Evidence for using bisphosphonate to treat Legg-Calvé-Perthes disease". Clin Orthop Relat Res. 470 (9): 2462–75. doi:10.1007/s11999-011-2240-0. PMC 3830104. PMID 22270467.
- ↑ Johannesen J, Briody J, McQuade M, Little DG, Cowell CT, Munns CF (May 2009). "Systemic effects of zoledronic acid in children with traumatic femoral head avascular necrosis and Legg-Calvé-Perthes disease". Bone. 45 (5): 898–902. doi:10.1016/j.bone.2009.04.255. PMID 19446052.
- ↑ Agabegi, Elizabeth D; Agabegi, Steven S. (2008). Step-Up to Medicine. Step-Up Series. Hagerstwon, MD: Lippincott Williams & Wilkins. p. 83. ISBN 978-0-7817-7153-5.
- ↑ Balasa VV, Gruppo RA, Glueck CJ, et al. (December 2004). "Legg-Calvé-Perthes disease and thrombophilia". J Bone Joint Surg Am. 86-A (12): 2642–7. doi:10.2106/00004623-200412000-00009. PMID 15590848.
{{cite journal}}: CS1 maint: url-status (link) - ↑ Perry, D. C.; Machin, D.M. G.; Pope, D.; Bruce, C. E.; Dangerfield, P.; Platt, M. J.; Hall, A. J. (Feb 1, 2012). "Racial and Geographic Factors in the Incidence of Legg-Calvé-Perthes' Disease: A Systematic Review". Am J Epidemiol. 175 (3): 159–166. doi:10.1093/aje/kwr293. PMID 22223709.
- ↑ 18.0 18.1 Perry, Daniel C.; Bruce, Colin E.; Pope, Daniel; Dangerfield, Peter; Platt, Mary Jane; Hall, Andrew J. (May 2012). "Legg-Calvé-Perthes disease in the UK: Geographic and temporal trends in incidence reflecting differences in degree of deprivation in childhood". Arthritis Rheum. 64 (5): 1673–1679. doi:10.1002/art.34316. PMID 22143958.
- ↑ Perry DC, Bruce CE, Pope D, Dangerfield P, Platt MJ, Hall AJ (2011). "Perthes' disease: deprivation and decline". Arch Dis Child. 96 (12): 1124–28. doi:10.1136/archdischild-2011-300413. PMID 22080458. S2CID 206853890.
- ↑ Barker DJ, Dixon E, Taylor JF (1978). "Perthes' disease of the hip in three regions of England". J Bone Joint Surg Br. 60 (4): 478–80. doi:10.1302/0301-620X.60B4.711792. PMID 711792.
- ↑ Hall AJ, Barker DJ (1989). "Perthes' disease in Yorkshire". J Bone Joint Surg Br. 71 (2): 229–33. doi:10.1302/0301-620X.71B2.2925740. PMID 2925740.
- ↑ Perry DC, Bruce CE, Pope D et al. Perthes' disease of the hip: socioeconomic inequalities and the urban environment. Arch Dis Child 2012;
- ↑ Kealey WD, Moore AJ, Cook S, Cosgrove AP (2000). "Deprivation, urbanisation and Perthes' disease in Northern Ireland". J Bone Joint Surg Br. 82 (2): 167–71. doi:10.1302/0301-620x.82b2.0820167. PMID 10755420. Archived from the original on 2019-08-02. Retrieved 2019-08-02.
- ↑ Daniel AB, Shah H, Kamath A, Guddettu V, Joseph B (Sep 2012). "Environmental tobacco and wood smoke increase the risk of Legg-Calvé-Perthes disease". Clin Orthop Relat Res. 470 (9): 2369–75. doi:10.1007/s11999-011-2180-8. PMC 3830089. PMID 22090357.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ↑ Chou, DTS; Ramachandran, M (2016). "Chapter 7: Legg-Calvé-Perthes' disease: Historical note". In Aresti, NA; Ramachandran, M; Paterson, JMH; Barry, M (eds.). Paediatric Orthopaedics in Clinical Practice. Springer. p. 105. ISBN 978-1-4471-6767-9.
- ↑ Mostofi, SB, ed. (2005). "Henning Waldenström". Who's who in orthopedics. London: Springer. p. 345. ISBN 9781852337865.
- ↑ 27.0 27.1 Wainwright, AM; Catterall, A (2010). "Capter 27: Legg–Calvé–Perthes disease: Its name". In Benson, M; Fixsen, J; MacNicol, M; Parsch, K (eds.). Children's orthopaedics and fractures (3rd ed.). London: Springer. p. 465. ISBN 9781848826106.
- ↑ Mostofi, SB, ed. (2005). "Arthur Thornton Legg". Who's who in orthopedics. London: Springer. p. 189. ISBN 9781852337865.
- ↑ Mostofi, SB, ed. (2005). "Jacques Calvé". Who's who in orthopedics. London: Springer. p. 51. ISBN 9781852337865.
- ↑ Mostofi, SB, ed. (2005). "Georg Perthes". Who's who in orthopedics. London: Springer. p. 267. ISBN 9781852337865.
- ↑ "The BOSS Study: British Orthopaedic Surgery Surveillance Study". www.boss.surgery. Archived from the original on 6 March 2016. Retrieved 28 February 2016.
- ↑ Tutt JFD (1935). "Tuberculosis of the hip joint in a cairn terrier". Vet Rec. 47: 428.
- ↑ Waldenstrom H (1909). "Der oobere Tuberculose Collumherd". Orthop Chir. 24: 487. as tuberculosis of the hip.
- ↑ "Archive copy". Archived from the original on 2011-09-05. Retrieved 2011-12-04.
{{cite web}}: CS1 maint: archived copy as title (link) - ↑ Hulth A, Norberg I, Olsson SE (July 1962). "Coxa plana in the dog". J Bone Joint Surg Am. 44-A (5): 918–30. doi:10.2106/00004623-196244050-00005. PMID 14036446.
{{cite journal}}: CS1 maint: url-status (link) - ↑ Ettinger, Stephen J.; Feldman, Edward C. (2000). Textbook of Veterinary Internal Medicine. Vol. 2 (5th ed.). W.B. Saunders Company. pp. 1873, v. ISBN 0-7216-7258-2.
- ↑ 37.0 37.1 Demko J, McLaughlin R (2005). "Developmental orthopedic disease". Vet Clin N Am Small Animal Pract. 35 (5): 1111–35, v. doi:10.1016/j.cvsm.2005.05.002. PMID 16129135.
- Bibliography
- Nelitz, Manfred; Lippacher, Sabine; Krauspe, Rüdiger; Reichel, Heiko (2009). "Perthes Disease". Dtsch Ärztebl Int. 106 (31–32): 517–523. doi:10.3238/arztebl.2009.0517. ISSN 1866-0452. PMC 2735835. PMID 19730720.
External links
| Classification | |
|---|---|
| External resources |
- Pages with script errors
- CS1 errors: missing periodical
- Webarchive template wayback links
- CS1 maint: archived copy as title
- CS1 maint: url-status
- CS1 maint: multiple names: authors list
- All articles with unsourced statements
- Articles with unsourced statements from August 2013
- Articles with invalid date parameter in template
- Articles with unsourced statements from May 2008
- Articles with unsourced statements from February 2012
- Syndromes affecting bones
- Rare diseases
- Skeletal disorders
- Osteonecrosis
- Syndromes in dogs
- RTT