Metamorphic reaction
Jump to navigation
Jump to search
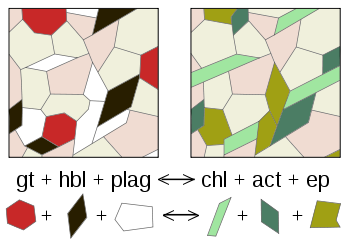
A metamorphic reaction is a chemical reaction that takes place during the geological process of metamorphism wherein one assemblage of minerals is transformed into a second assemblage which is stable under the new temperature/pressure conditions resulting in the final stable state of the observed metamorphic rock.[1]
Examples include the production of talc under varied metamorphic conditions:
- serpentine + carbon dioxide → talc + magnesite + water
- chlorite + quartz → kyanite + talc + water

Polymorphic Transformations
This section is empty. You can help by adding to it. (June 2019) |
Exsolution Reactions
This section is empty. You can help by adding to it. (June 2019) |
Devolatilization Reactions
This section is empty. You can help by adding to it. (July 2019) |
Continuous Reactions
This section is empty. You can help by adding to it. (June 2019) |
Ion Exchange Reactions
This section is empty. You can help by adding to it. (June 2019) |
Oxidation/Reduction Reactions
This section is empty. You can help by adding to it. (June 2019) |
Reactions Involving Dissolved Species
This section is empty. You can help by adding to it. (June 2019) |
Chemographics
This section is empty. You can help by adding to it. (June 2019) |
Petrogenetic Grids
This section is empty. You can help by adding to it. (June 2019) |
Schreinemakers Method
This section is empty. You can help by adding to it. (June 2019) |
Reaction Mechanisms
This section is empty. You can help by adding to it. (June 2019) |
See also
Notes
- ^ "Types of Metamorphic Reactions". Tulane University. Retrieved 2007-06-22.
Categories:
- Articles to be expanded from June 2019
- All articles to be expanded
- Articles with empty sections from June 2019
- All articles with empty sections
- Articles using small message boxes
- Articles to be expanded from July 2019
- Articles with empty sections from July 2019
- Metamorphic petrology
- Geochemical processes
- Reaction mechanisms