Lithium chlorate
Jump to navigation
Jump to search
| |
| Names | |
|---|---|
| Other names
Chloric acid, lithium salt
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.033.288 |
PubChem CID
|
|
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
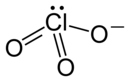
| LiClO3 | |
| Molar mass | 90.39 g/mol |
| Melting point | 127.6 to 129 °C (261.7 to 264.2 °F; 400.8 to 402.1 K)[2][3][4] |
| 241 g/100 mL (0 °C) 459 g/100 mL (25 °C) 777 g/100 mL (60 °C) 2226 g/100 mL (100 °C)[1] | |
| −28.8·10−6 cm3/mol | |
| Related compounds | |
Other anions
|
Lithium chloride Lithium hypochlorite Lithium perchlorate |
Other cations
|
Sodium chlorate Potassium chlorate Caesium chlorate |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Lithium chlorate is the inorganic chemical compound with the formula LiClO3. Like all chlorates, it is an oxidizer and may become unstable and possibly explosive if mixed with organic materials, reactive metal powders, or sulfur.
It can be manufactured by the reaction of hot, concentrated lithium hydroxide with chlorine:
- 3 Cl2 + 6 LiOH → 5 LiCl + LiClO3 + 3 H2O
Lithium chlorate has one of the highest solubilities in water for a chemical compound. It is also a six-electron oxidant. Its electrochemical reduction is facilitated by acid, electrocatalysts and redox mediators. These properties make lithium chlorate a useful oxidant for high energy density flow batteries.[5] Lithium chlorate has a very low melting point for an inorganic ionic salt.
References
- ^ John Rumble (June 18, 2018). CRC Handbook of Chemistry and Physics (99 ed.). CRC Press. pp. 4–47. ISBN 978-1138561632.
- ^ Wang, Su-Chee Simon (1983). "The Electrochemistry of Molten Lithium Chlorate and its Possible Use with Lithium in a Battery". Journal of the Electrochemical Society. 130 (4): 741–747. Bibcode:1983JElS..130..741W. doi:10.1149/1.2119796.
- ^ A. N. Campbell, E. M. Kartzmark, W. B. Maryk (1966). "The Systems Sodium Chlorate - Water - Dioxane and Lithium Chlorate - Water - Dioxane, at 25°". Can. J. Chem. 44 (8): 935–937. doi:10.1139/v66-136. S2CID 97413079.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ http://scitation.aip.org/getabs/servlet/GetabsServlet?prog=normal&id=JESOAN000130000004000741000001&idtype=cvips&gifs=yes&ref=no [dead link]
- ^ US 20140170511
Categories:
- CS1 maint: multiple names: authors list
- All articles with dead external links
- Articles with dead external links from January 2019
- Articles without EBI source
- Articles without KEGG source
- Articles without UNII source
- ECHA InfoCard ID from Wikidata
- Articles containing unverified chemical infoboxes
- Articles with short description
- Short description matches Wikidata
- Chlorates
- Lithium salts
- Oxidizing agents