Homocitric acid
Jump to navigation
Jump to search

| |
| Names | |
|---|---|
| Preferred IUPAC name
2-Hydroxybutane-1,2,4-tricarboxylic acid | |
| Other names
Homocitric acid
Homocitrate | |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChemSpider | |
| KEGG | |
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
| C7H10O7 | |
| Molar mass | 206.150 g·mol−1 |
| Appearance | Colorless solid |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Homocitric acid is an organic compound with the formula HOC(CO2H)(CH2CO2H)(C2H4CO2H). This tricarboxylic acid occurs naturally as a component of the iron-molybdenum cofactor of certain nitrogenase proteins.[1] Biochemists often refer to this cofactor as homocitrate, which is the conjugate bases that predominate in neutral aqueous solutions of this species.
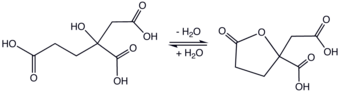
The molecule is related to citric acid by the addition of one methylene unit, hence the prefix "homo." Unlike citric acid, homocitric acid is chiral. The acid exists in equilibrium with the lactone.
See also
References
- ^ Rees, Douglas C. (2002). "Great Metalloclusters in Enzymology" (PDF). Annual Review of Biochemistry. 71: 221–46. doi:10.1146/annurev.biochem.71.110601.135406. PMID 12045096.
Categories:
- Articles with changed EBI identifier
- Articles with changed ChemSpider identifier
- Articles with changed KEGG identifier
- Articles with changed InChI identifier
- Articles containing unverified chemical infoboxes
- Chembox image size set
- Articles with short description
- Short description matches Wikidata
- Alpha hydroxy acids
- Chelating agents
- Tricarboxylic acids
- All stub articles
- Biochemistry stubs
- Chemical compound stubs