Shwachman–Diamond syndrome
| Shwachman–Diamond syndrome | |
|---|---|
| Other names: Shwachman–Bodian–Diamond syndrome | |
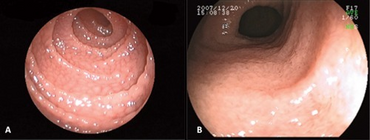 | |
| Endoscopic view: diffusely-scattered granular mucosa in (A) duodenum and (B) terminal ileum. | |
| Prognosis | five-year survival rate 64% |
| Frequency | 122,600 |
| Deaths | 21,650 |
Shwachman–Diamond syndrome (SDS), or Shwachman–Bodian–Diamond syndrome, is a rare congenital disorder characterized by exocrine pancreatic insufficiency, bone marrow dysfunction, skeletal abnormalities and short stature. After cystic fibrosis (CF), it is the second most common cause of exocrine pancreatic insufficiency in children.
Signs and symptoms
The syndrome shows a wide range of abnormalities and symptoms. The main characteristics of the syndrome are exocrine pancreatic dysfunction, hematologic abnormalities and growth retardation. Only the first two of these are included in the clinical diagnostic criteria.[1]
- Hematologic abnormalities: neutropenia may be intermittent or persistent and is the most common hematological finding. Low neutrophil counts leave patients at risk of developing severe recurrent infections that may be life-threatening. Anemia (low red blood cell counts) and thrombocytopenia (low platelet counts) may also occur. Bone marrow is typically hypocellular, with maturation arrest in the myeloid lineages that give rise to neutrophils, macrophages, platelets and red blood cells. Patients may also develop progressive marrow failure or transform to acute myelogenous leukemia.
- Exocrine pancreatic dysfunction: Pancreatic exocrine insufficiency arises due to a lack of acinar cells that produce digestive enzymes. These are extensively depleted and replaced by fat. A lack of pancreatic digestive enzymes leaves patients unable to digest and absorb fat. However, pancreatic status may improve with age in some patients.
- Growth retardation: More than 50% of patients are below the third percentile for height, and short stature appears to be unrelated to nutritional status. Other skeletal abnormalities include metaphyseal dysostosis (45% of patients), thoracic dystrophy (rib cage abnormalities in 46% of patients) and costochondral thickening (shortened ribs with flared ends in 32% of patients). Skeletal problems are one of the most variable components of SDS, with 50% affected siblings from the same family discordant for clinical presentation or type of abnormality. Despite this, a careful review of radiographs from 15 patients indicated that all of them had at least one skeletal anomaly, though many were subclinical.
- Other features include metaphysial dysostosis, mild hepatic dysfunction, increased frequency of infections.
Genetics

Shwachman–Diamond syndrome is characterized by an autosomal recessive mode of inheritance. The gene that is mutated in this syndrome, SBDS,[2][3][4] lies on the long arm of chromosome 7 at cytogenetic position 7q11.[5][6] It is composed of five exons and has an associated mRNA transcript that is 1.6 kilobase pairs in length. The SBDS gene resides in a block of genomic sequence that is locally duplicated on the chromosome. The second copy contains a non-functional version of the SBDS gene that is 97% identical to the original gene, but has accumulated inactivating mutations over time. It is considered to be a pseudogene. In a study of 158 SDS families, 75% of disease-associated mutations appeared to be the result of gene conversion, while 89% of patients harbored at least one such mutation. Gene conversion occurs when the intact SBDS gene and its pseudogene copy aberrantly recombine at meiosis, leading to an incorporation of pseudogene-like sequences into the otherwise functional copy of the SBDS gene, thereby inactivating it.[citation needed]
Two gene conversion mutations predominate in SDS patients. One is a splice site mutation affecting the 5' splice site of intron two, while the second is an exon two nonsense mutation. The marked absence of patients homozygous for the otherwise common nonsense mutation suggested that the SBDS gene is essential. Consistent with this, knockout of the mouse gene leads to early embryonic lethality.[7] This, in turn, suggests that the common splice site mutation seen in patients may be hypomorphic, i.e. that it results in only a partial loss of function.
Mechanisms
The SBDS gene is expressed in all tissues and encodes a protein of 250 amino acid residues. A great deal of indirect evidence suggested that the SBDS protein may be involved in an aspect of cellular RNA metabolism or ribosome assembly or function. The wide occurrence of the gene in all archaea and eukaryotes supported a role for this protein in a very fundamental and evolutionarily conserved aspect of cellular biology.[8] The homologous genes in archaea also tend to be present in conserved cluster enriched for RNA processing and ribosomal genes. A specific function for SBDS in RNA metabolism or ribosome assembly or function is further supported by its localization to the nucleolus, the nuclear subdomain where these processes occur. In line with this, the yeast homologue, SdoI, has been shown to be critical for maturation of pre-60S ribosomes, by effecting release and recycling of the nucleolar shuttling factor Tif6.[9] This is required for 60S maturation and translational activation of ribosomes. It has also been shown that the Dictyostelium discoideum homologue catalyzes the removal of eukaryotic initiation factor 6 (eIF6), which is required for the translational activation of ribosomes.[10] Cells from SDS patients were shown to have a defect in assembly of ribosome subunits.[citation needed]
At present, it is not obvious how disruption of the basic cellular process of translation leads to the tissue- and organ-specific manifestations seen in SDS. However, unusual and combinations of tissues and organs are also affected in Diamond–Blackfan anemia, X-linked dyskeratosis congenita, and cartilage–hair hypoplasia—three diseases that may also be linked to defective ribosome function. Pleiotropic disease features may be the result of cell-specific effects of reduced levels of SBDS activity provided by hypomorphic mutations.[citation needed]
Diagnosis
Initially, the clinical presentation of SDS may appear similar to cystic fibrosis. However, CF can be excluded with a normal chloride in sweat test but faecal elastase as a marker of pancreatic function will be reduced. The variation, intermittent nature, and potential for long-term improvement of some clinical features make this syndrome difficult to diagnose. SDS may present with either malabsorption, or hematological problems. Rarely, SDS may present with skeletal defects, including severe rib cage abnormalities that lead to difficulty in breathing. Diagnosis is generally based on evidence of exocrine pancreatic dysfunction and neutropenia. Skeletal abnormalities and short stature are characteristics that can be used to support the diagnosis. The gene responsible for the disease has been identified and genetic testing is now available. Though useful in diagnostics, a genetic test does not surmount the need for careful clinical assessment and monitoring of all patients.[citation needed]
Management
Pancreatic exocrine insufficiency may be treated through pancreatic enzyme supplementation,[11] while severe skeletal abnormalities may require surgical intervention. Neutropenia may be treated with granulocyte-colony stimulating factor (GCSF) to boost peripheral neutrophil counts. However, there is ongoing and unresolved concern that this drug could contribute to the development of leukemia. Signs of progressive marrow failure may warrant bone marrow transplantation (BMT). This has been used successfully to treat hematological aspects of disease. However, SDS patients have an elevated occurrence of BMT-related adverse events, including graft-versus-host disease (GVHD) and toxicity relating to the pre-transplant conditioning regimen. In the long run, study of the gene that is mutated in SDS should improve understanding of the molecular basis of disease. This, in turn, may lead to novel therapeutic strategies, including gene therapy and other gene- or protein-based approaches.[citation needed]
==Research A major goal of curative therapy for SDS is to reduce the risk of bone marrow failure and halt the progression of malignant transformation toward myelodysplastic syndrome (MDS) and acute myeloid leukemia (AML), the most detrimental complications of SDS. Currently, there is no such therapy. However, several emerging therapeutic strategies, including gene therapy and antisense oligonucleotides (ASOs), could potentially slow or prevent malignant transformation, at least in theory. These new therapies have been proven effective for several rare diseases, including metachromatic leukodystrophy and spinal muscular atrophy. Several SDS patient groups are advocating for better therapies for SDS,[12][13][14][15] and one organization is focused on driving the therapy development efforts.[16] The challenge is whether the SDS community can come together to support the required research,[17] and whether the organizations can successfully execute a strategy that coordinates the efforts for new therapy development.[18]
Epidemiology
It is thought to have an estimated incidence of 1 in 75,000 people.[19]
History
The disease was first described as a coherent clinical entity in May 1964 by Bodian, Sheldon, and Lightwood.[20] It was subsequently described by Shwachman, Diamond, Oski, and Khaw in November of the same year.[21] In 2001, linkage analysis in SDS families indicated that affected gene mapped to a large region of human chromosome seven.[22] In 2002, this interval was refined to a region on the long arm of the chromosome next to the centromere.[23]
In 2003, a team of researchers led by Johanna Rommens at the Hospital of Sick Children (SickKids) in Toronto, Canada, discovered mutations in the SBDS gene (Shwachman–Bodian–Diamond syndrome) were associated with disease.[24]
Eponym
Shwachman–Diamond syndrome, less commonly known as Shwachman–Bodian–Diamond syndrome, is named for Harry Shwachman (1910 – September 12, 1986), an American physician, Martin Bodian (1912 – May 12, 1994), a British ophthalmologist who worked in New York City, and Louis Klein Diamond (May 11, 1902 – June 14, 1999), an American pediatrician.
References
- ↑ Orkin, Stuart H.; Nathan, David G.; Ginsburg, David; A. Thomas Look (2009). Nathan and Oski's hematology of infancy and childhood. Elsevier Health Sciences. pp. 344–. ISBN 978-1-4160-3430-8. Archived from the original on 5 October 2021. Retrieved 8 August 2011.
- ↑ Shammas C, Menne TF, Hilcenko C, Michell SR, Goyenechea B, Boocock GR, Durie PR, Rommens JM, Warren AJ (2005). "Structural and mutational analysis of the SBDS protein family. Insight into the leukemia-associated Shwachman–Diamond Syndrome". J Biol Chem. 280 (19): 19221–9. doi:10.1074/jbc.M414656200. PMID 15701631. S2CID 8011896.
- ↑ Austin KM, Leary RJ, Shimamura A (2005). "The Shwachman–Diamond SBDS protein localizes to the nucleolus". Blood. 106 (4): 1253–8. doi:10.1182/blood-2005-02-0807. PMC 1895203. PMID 15860664.
- ↑ Makitie O, Ellis L, Durie PR, Morrison JA, Sochett EB, Rommens JM, Cole WG (2004). "Skeletal phenotype in patients with Shwachman–Diamond syndrome and mutations in SBDS". Clin Genet. 65 (2): 101–12. doi:10.1111/j.0009-9163.2004.00198.x. PMID 14984468. S2CID 34964226.
- ↑ Goobie, S; Popovic, M; Morrison, J; Ellis, L; Ginzberg, H; Boocock, GR; Ehtesham, N; Bétard, C; Brewer, CG; Roslin, NM; Hudson, TJ; Morgan, K; Fujiwara, TM; Durie, PR; Rommens, JM (April 2001). "Shwachman-Diamond syndrome with exocrine pancreatic dysfunction and bone marrow failure maps to the centromeric region of chromosome 7". American Journal of Human Genetics. 68 (4): 1048–54. doi:10.1086/319505. PMC 1275624. PMID 11254457.
- ↑ Popovic, M; Goobie, S; Morrison, J; Ellis, L; Ehtesham, N; Richards, N; Boocock, G; Durie, PR; Rommens, JM (April 2002). "Fine mapping of the locus for Shwachman-Diamond syndrome at 7q11, identification of shared disease haplotypes, and exclusion of TPST1 as a candidate gene". European Journal of Human Genetics. 10 (4): 250–8. doi:10.1038/sj.ejhg.5200798. PMID 12032733. S2CID 12667823.
- ↑ Zhang S, Shi M, Hui CC, Rommens, JM (2006). "Loss of the mouse ortholog of the shwachman-diamond syndrome gene (Sbds) results in early embryonic lethality". Mol Cell Biol. 26 (17): 6656–63. doi:10.1128/MCB.00091-06. PMC 1592835. PMID 16914746.
- ↑ Boocock, GR, Marit, MR, Rommens, JM (2006). "Phylogeny, sequence conservation, and functional complementation of the SBDS protein family". Genomics. 87 (6): 758–71. doi:10.1016/j.ygeno.2006.01.010. PMID 16529906.
- ↑ Menne TF, Goyenechea B, Sánchez-Puig N, Wong CC, Tonkin LM, Ancliff PJ, Brost RL, Costanzo M, Boone C, Warren AJ (2007). "The Shwachman-Bodian-Diamond syndrome protein mediates translational activation of ribosomes in yeast". Nat Genet. 39 (4): 486–95. doi:10.1038/ng1994. PMID 17353896. S2CID 8076230.
- ↑ Wong CC, Traynor D, Basse N, Kay RR, Warren AJ (2011). "Defective ribosome assembly in Shwachman-Diamond syndrome". Blood. 118 (16): 4305–12. doi:10.1182/blood-2011-06-353938. PMID 21803848.
- ↑ Disorders, National Organization for Rare (2003). NORD guide to rare disorders. Lippincott Williams & Wilkins. pp. 417–. ISBN 978-0-7817-3063-1. Archived from the original on 5 October 2021. Retrieved 8 August 2011.
- ↑ "Home". Shwachman (in Nederlands). Archived from the original on 2021-01-24. Retrieved 2021-01-25.
- ↑ "AISS". www.shwachman.it. Archived from the original on 2020-09-29. Retrieved 2021-01-25.
- ↑ "Home". Shwachman-Diamond Syndrome. Archived from the original on 2021-01-21. Retrieved 2021-01-25.
- ↑ "Archive copy". www.sdsdeutschland.de. Archived from the original on 2021-01-21. Retrieved 2021-01-25.
{{cite web}}: CS1 maint: archived copy as title (link) - ↑ "Shwachman-Diamond Syndrome Alliance - SDS Alliance Foundation". Shwachman-Diamond Syndrome Alliance. Archived from the original on 2021-01-18. Retrieved 2021-01-25.
- ↑ "How Nonprofit Foundations Can Sustainably Fund Disease Research". Harvard Business Review. 2020-09-30. ISSN 0017-8012. Archived from the original on 2021-01-21. Retrieved 2021-01-25.
- ↑ "How Medical Nonprofits Set Winning Strategy". Harvard Business Review. 2020-03-06. ISSN 0017-8012. Archived from the original on 2021-01-26. Retrieved 2021-01-25.
- ↑ Hassan, Fauziya; Byersdorfer, Craig; Nasr, Samya (1 January 2010). "Severe Shwachman-Diamond syndrome and associated CF carrier mutations". Respiratory Medicine CME. 3 (1): 18–19. doi:10.1016/j.rmedc.2009.02.001.
- ↑ Bodian M, Sheldon W, Lightwood R (1964). "Congenital hypoplasia of the exocrine pancreas". Acta Paediatr. 53 (3): 282–93. doi:10.1111/j.1651-2227.1964.tb07237.x. PMID 14158482. S2CID 34362201.
- ↑ Shwachman H, Diamond LK, Oski FA, Khaw KT (1964). "The syndrome of pancreatic insufficiency and bone marrow dysfunction". J Pediatr. 65 (5): 645–63. doi:10.1016/S0022-3476(64)80150-5. PMID 14221166.
- ↑ Goobie S, Popovic M, Morrison J, Ellis L, Ginzberg H, Boocock GR, Ehtesham N, Betard C, Brewer CG, Roslin NM, Hudson TJ, Morgon K, Fujiwara TM, Durie PR, Rommens JM (2001). "Shwachman–Diamond syndrome with exocrine pancreatic dysfunction and bone marrow failure maps to the centromeric region of chromosome 7". Am J Hum Genet. 68 (4): 1048–54. doi:10.1086/319505. PMC 1275624. PMID 11254457.
- ↑ Popovic M, Goobie S, Morrison J, Ellis L, Ehtesham N, Richards N, Boocock G, Durie PR, Rommens JM (2002). "Fine mapping of the locus for Shwachman–Diamond syndrome at 7q11, identification of shared disease haplotypes, and exclusion of TPST1 as a candidate gene". Eur J Hum Genet. 10 (4): 250–8. doi:10.1038/sj.ejhg.5200798. PMID 12032733. S2CID 12667823.
- ↑ Boocock GR, Morrison JA, Popovic M, Richards N, Ellis L, Durie PR, Rommens JM (2003). "Mutations in SBDS are associated with Shwachman–Diamond syndrome". Nat Genet. 33 (1): 97–101. doi:10.1038/ng1062. PMID 12496757. S2CID 5091627.
Further reading
- Foerster (2014). "Ch 37. Inherited Aplastic Anemia Syndromes". In John P. Greer; Daniel A. Arber; Bertil Glader; Alan F. List; Robert T. Means Jr.; Frixos Paraskevas; George M. Rodgers; John Foerster (eds.). Wintrobe's clinical hematology (13th ed.). p. Shwachman-Diamond Syndrome. ISBN 978-1451172683. Archived from the original on 2021-10-05. Retrieved 2021-05-17.
- GeneReviews/NCBI/NIH/UW entry on Shwachman–Diamond Syndrome Archived 2010-02-07 at the Wayback Machine
External links
| Classification | |
|---|---|
| External resources |
- Pages with script errors
- CS1 Nederlands-language sources (nl)
- CS1 maint: archived copy as title
- All articles with unsourced statements
- Articles with unsourced statements from July 2020
- Articles with invalid date parameter in template
- Articles with unsourced statements from September 2020
- Webarchive template wayback links
- Autosomal recessive disorders
- Pancreas disorders
- Blood disorders
- Noninfectious immunodeficiency-related cutaneous conditions
- Congenital defects of phagocyte number, function, or both
- Syndromes