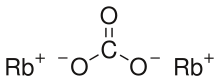
Rubidium carbonate
Jump to navigation
Jump to search
| |

| |
| Names | |
|---|---|
| IUPAC name
Rubidium carbonate
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.008.666 |
PubChem CID
|
|
| RTECS number |
|
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| Rb2CO3 | |
| Molar mass | 230.945 g/mol |
| Appearance | White powder, very hygroscopic |
| Melting point | 837 °C (1,539 °F; 1,110 K)[1] |
| Boiling point | 900 °C (1,650 °F; 1,170 K) (decomposes) |
| Very soluble | |
| −75.4·10−6 cm3/mol | |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards
|
Irritant |
| Flash point | Non-flammable |
| Related compounds | |
Other cations
|
Lithium carbonate Sodium carbonate Potassium carbonate Caesium carbonate |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Rubidium carbonate, Rb2CO3, is a convenient compound of rubidium; it is stable, not particularly reactive, and readily soluble in water, and is the form in which rubidium is usually sold.
Preparation
This salt can be prepared by adding ammonium carbonate to rubidium hydroxide.[2]
Uses
It is used in some kinds of glass-making by enhancing stability and durability as well as reducing its conductivity. It is also used as a part of a catalyst for preparing short-chain alcohols from feed gas.[3]
References
- ^ Pradyot Patnaik. Handbook of Inorganic Chemicals. McGraw-Hill, 2002, ISBN 0-07-049439-8
- ^ Chisholm, Hugh, ed. (1911). . Encyclopædia Britannica. Vol. 23 (11th ed.). Cambridge University Press. p. 809.
- ^ Canada Patents
Categories:
- Wikipedia articles incorporating a citation from the 1911 Encyclopaedia Britannica with Wikisource reference
- Articles without EBI source
- Articles without KEGG source
- Articles without UNII source
- ECHA InfoCard ID from Wikidata
- Articles containing unverified chemical infoboxes
- Articles with short description
- Short description matches Wikidata
- Carbonates
- Rubidium compounds
- All stub articles
- Inorganic compound stubs