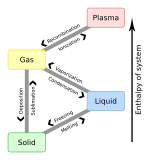
Triple point
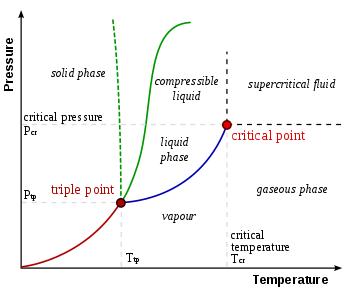
In thermodynamics, the triple point of a substance is the temperature and pressure at which the three phases (gas, liquid, and solid) of that substance coexist in thermodynamic equilibrium.[1] It is that temperature and pressure at which the sublimation, fusion, and vaporisation curves meet. For example, the triple point of mercury occurs at a temperature of −38.8 °C (−37.8 °F) and a pressure of 0.165 mPa.
In addition to the triple point for solid, liquid, and gas phases, a triple point may involve more than one solid phase, for substances with multiple polymorphs. Helium-4 is unusual in that it has no sublimation/deposition curve and therefore no triple points where its solid phase meets its gas phase. Instead, it has a vapor-liquid-superfluid point, a solid-liquid-superfluid point, a solid-solid-liquid point, and a solid-solid-superfluid point. None of these should be confused with the Lambda Point, which is not any kind of triple point.
The triple point of water was used to define the kelvin, the base unit of thermodynamic temperature in the International System of Units (SI).[2] The value of the triple point of water was fixed by definition, rather than by measurement, but that changed with the 2019 redefinition of SI base units. The triple points of several substances are used to define points in the ITS-90 international temperature scale, ranging from the triple point of hydrogen (13.8033 K) to the triple point of water (273.16 K, 0.01 °C, or 32.018 °F).
The term "triple point" was coined in 1873 by James Thomson, brother of Lord Kelvin.[3]
Triple point of water
Gas–liquid–solid triple point

Following the 2019 redefinition of the SI base units, the value of the triple point of water is no longer used as a defining point. However, its empirical value remains important: the unique combination of pressure and temperature at which liquid water, solid ice, and water vapor coexist in a stable equilibrium is approximately 273.16±0.0001 K[4] and a vapor pressure of 611.657 pascals (6.11657 mbar; 0.00603659 atm).[5][6]
Liquid water can only exist at pressures equal to or greater than the triple point. Below this, in the vacuum of outer space, solid ice sublimates, transitioning directly into water vapor when heated at a constant pressure. Conversely, above the triple point, solid ice first melts into liquid water upon heating at a constant pressure, then evaporates or boils to form vapor at a higher temperature.
For most substances, the gas–liquid–solid triple point is the minimum temperature where the liquid can exist. For water, this is not the case. The melting point of ordinary ice decreases with pressure, as shown by the phase diagram's dashed green line. Just below the triple point, compression at a constant temperature transforms water vapor first to solid and then to liquid.
Historically, during the Mariner 9 mission to Mars, the triple point pressure of water was used to define "sea level." Now, laser altimetry and gravitational measurements are preferred to define Martian elevation.[7]
High-pressure phases
At high pressures, water has a complex phase diagram with 15 known phases of ice and several triple points, including 10 whose coordinates are shown in the diagram. For example, the triple point at 251 K (−22 °C) and 210 MPa (2070 atm) corresponds to the conditions for the coexistence of ice Ih (ordinary ice), ice III and liquid water, all at equilibrium. There are also triple points for the coexistence of three solid phases, for example ice II, ice V and ice VI at 218 K (−55 °C) and 620 MPa (6120 atm).
For those high-pressure forms of ice which can exist in equilibrium with liquid, the diagram shows that melting points increase with pressure. At temperatures above 273 K (0 °C), increasing the pressure on water vapor results first in liquid water and then a high-pressure form of ice. In the range 251–273 K, ice I is formed first, followed by liquid water and then ice III or ice V, followed by other still denser high-pressure forms.

| Phases in stable equilibrium | Pressure | Temperature |
|---|---|---|
| liquid water, ice Ih, and water vapor | 611.657 Pa[8] | 273.16 K (0.01 °C) |
| liquid water, ice Ih, and ice III | 209.9 MPa | 251 K (−22 °C) |
| liquid water, ice III, and ice V | 350.1 MPa | −17.0 °C |
| liquid water, ice V, and ice VI | 632.4 MPa | 0.16 °C |
| ice Ih, Ice II, and ice III | 213 MPa | −35 °C |
| ice II, ice III, and ice V | 344 MPa | −24 °C |
| ice II, ice V, and ice VI | 626 MPa | −70 °C |
Triple-point cells
Triple-point cells are used in the calibration of thermometers. For exacting work, triple-point cells are typically filled with a highly pure chemical substance such as hydrogen, argon, mercury, or water (depending on the desired temperature). The purity of these substances can be such that only one part in a million is a contaminant, called "six nines" because it is 99.9999% pure. A specific isotopic composition (for water, VSMOW) is used because variations in isotopic composition cause small changes in the triple point. Triple-point cells are so effective at achieving highly precise, reproducible temperatures, that an international calibration standard for thermometers called ITS–90 relies upon triple-point cells of hydrogen, neon, oxygen, argon, mercury, and water for delineating six of its defined temperature points.
Table of triple points
This table lists the gas–liquid–solid triple points of several substances. Unless otherwise noted, the data come from the U.S. National Bureau of Standards (now NIST, National Institute of Standards and Technology).[9]
| Substance | T [K] (°C) | p [kPa]* (atm) |
|---|---|---|
| Acetylene | 192.4 K (−80.7 °C) | 120 kPa (1.2 atm) |
| Ammonia | 195.40 K (−77.75 °C) | 6.060 kPa (0.05981 atm) |
| Argon | 83.8058 K (−189.3442 °C) | 68.9 kPa (0.680 atm) |
| Arsenic | 1,090 K (820 °C) | 3,628 kPa (35.81 atm) |
| Butane[10] | 134.6 K (−138.6 °C) | 7×10−4 kPa (6.9×10−6 atm) |
| Carbon (graphite) | 4,765 K (4,492 °C) | 10,132 kPa (100.00 atm) |
| Carbon dioxide | 216.55 K (−56.60 °C) | 517 kPa (5.10 atm) |
| Carbon monoxide | 68.10 K (−205.05 °C) | 15.37 kPa (0.1517 atm) |
| Chloroform | 175.43 K (−97.72 °C)[citation needed] | 0.870 kPa (0.00859 atm)[citation needed] |
| Deuterium | 18.63 K (−254.52 °C) | 17.1 kPa (0.169 atm) |
| Ethane | 89.89 K (−183.26 °C) | 1.1×10−3 kPa (1.1×10−5 atm) |
| Ethanol[11] | 150 K (−123 °C) | 4.3×10−7 kPa (4.2×10−9 atm) |
| Ethylene | 104.0 K (−169.2 °C) | 0.12 kPa (0.0012 atm) |
| Formic acid[12] | 281.40 K (8.25 °C) | 2.2 kPa (0.022 atm) |
| Helium-4 (vapor−He-I−He-II)[13] | 2.1768 K (−270.9732 °C) | 5.048 kPa (0.04982 atm) |
| Helium-4 (hcp−bcc−He-II)[14] | 1.463 K (−271.687 °C) | 26.036 kPa (0.25696 atm) |
| Helium-4 (bcc−He-I−He-II)[14] | 1.762 K (−271.388 °C) | 29.725 kPa (0.29336 atm) |
| Helium-4 (hcp−bcc−He-I)[14] | 1.772 K (−271.378 °C) | 30.016 kPa (0.29623 atm) |
| Hexafluoroethane[15] | 173.08 K (−100.07 °C) | 26.60 kPa (0.2625 atm) |
| Hydrogen | 13.8033 K (−259.3467 °C) | 7.04 kPa (0.0695 atm) |
| Hydrogen-1 (Protium)[16] | 13.96 K (−259.19 °C) | 7.18 kPa (0.0709 atm) |
| Hydrogen chloride | 158.96 K (−114.19 °C) | 13.9 kPa (0.137 atm) |
| Iodine[17] | 386.65 K (113.50 °C) | 12.07 kPa (0.1191 atm) |
| Isobutane[18] | 113.55 K (−159.60 °C) | 1.9481×10−5 kPa (1.9226×10−7 atm) |
| Krypton | 115.76 K (−157.39 °C) | 74.12 kPa (0.7315 atm) |
| Mercury | 234.3156 K (−38.8344 °C) | 1.65×10−7 kPa (1.63×10−9 atm) |
| Methane | 90.68 K (−182.47 °C) | 11.7 kPa (0.115 atm) |
| Neon | 24.5561 K (−248.5939 °C) | 43.332 kPa (0.42765 atm) |
| Nitric oxide | 109.50 K (−163.65 °C) | 21.92 kPa (0.2163 atm) |
| Nitrogen | 63.18 K (−209.97 °C) | 12.6 kPa (0.124 atm) |
| Nitrous oxide | 182.34 K (−90.81 °C) | 87.85 kPa (0.8670 atm) |
| Oxygen | 54.3584 K (−218.7916 °C) | 0.14625 kPa (0.0014434 atm) |
| Palladium | 1,825 K (1,552 °C) | 3.5×10−3 kPa (3.5×10−5 atm) |
| Platinum | 2,045 K (1,772 °C) | 2×10−4 kPa (2.0×10−6 atm) |
| Radon | 202 K (−71 °C) | 70 kPa (0.69 atm) |
| (mono)Silane[19] | 88.48 K (−184.67 °C) | 0.019644 kPa (0.00019387 atm) |
| Sulfur dioxide | 197.69 K (−75.46 °C) | 1.67 kPa (0.0165 atm) |
| Titanium | 1,941 K (1,668 °C) | 5.3×10−3 kPa (5.2×10−5 atm) |
| Uranium hexafluoride | 337.17 K (64.02 °C) | 151.7 kPa (1.497 atm) |
| Water[5][6] | 273.16 K (0.01 °C) | 0.611657 kPa (0.00603659 atm) |
| Xenon | 161.3 K (−111.8 °C) | 81.5 kPa (0.804 atm) |
| Zinc | 692.65 K (419.50 °C) | 0.065 kPa (0.00064 atm) |
Notes:
- For comparison, typical atmospheric pressure is 101.325 kPa (1 atm).
- Before the new definition of SI units, water's triple point, 273.16 K, was an exact number.
See also
References
- ^ IUPAC, Compendium of Chemical Terminology, 2nd ed. (the "Gold Book") (1997). Online corrected version: (1994) "Triple point". doi:10.1351/goldbook.T06502.
- ^ Definition of the kelvin at BIPM.
- ^ James Thomson (1873) "A quantitative investigation of certain relations between the gaseous, the liquid, and the solid states of water-substance", Proceedings of the Royal Society, 22 : 27–36. From a footnote on page 28: " … the three curves would meet or cross each other in one point, which I have called the triple point".
- ^ "SI Brochure: The International System of Units (SI) – 9th edition". BIPM. Retrieved 21 February 2022.
- ^ a b International Equations for the Pressure along the Melting and along the Sublimation Curve of Ordinary Water Substance. W. Wagner, A. Saul and A. Pruss (1994), J. Phys. Chem. Ref. Data, 23, 515.
- ^ a b Murphy, D. M. (2005). "Review of the vapour pressures of ice and supercooled water for atmospheric applications". Quarterly Journal of the Royal Meteorological Society. 131 (608): 1539–1565. Bibcode:2005QJRMS.131.1539M. doi:10.1256/qj.04.94. S2CID 122365938.
- ^ Carr, Michael H. (2007). The Surface of Mars. Cambridge University Press. p. 5. ISBN 978-0-521-87201-0.
- ^ Murphy, D. M. (2005). "Review of the vapour pressures of ice and supercooled water for atmospheric applications". Quarterly Journal of the Royal Meteorological Society. 131 (608): 1539–1565. Bibcode:2005QJRMS.131.1539M. doi:10.1256/qj.04.94. S2CID 122365938.
- ^ Cengel, Yunus A.; Turner, Robert H. (2004). Fundamentals of thermal-fluid sciences. Boston: McGraw-Hill. p. 78. ISBN 0-07-297675-6.
- ^ See Butane (data page)
- ^ See Ethanol (data page)
- ^ See Formic acid (data page)
- ^ Donnelly, Russell J.; Barenghi, Carlo F. (1998). "The Observed Properties of Liquid Helium at the Saturated Vapor Pressure". Journal of Physical and Chemical Reference Data. 27 (6): 1217–1274. Bibcode:1998JPCRD..27.1217D. doi:10.1063/1.556028.
- ^ a b c Hoffer, J. K.; Gardner, W. R.; Waterfield, C. G.; Phillips, N. E. (April 1976). "Thermodynamic properties of 4He. II. The bcc phase and the P-T and VT phase diagrams below 2 K". Journal of Low Temperature Physics. 23 (1): 63–102. Bibcode:1976JLTP...23...63H. doi:10.1007/BF00117245. S2CID 120473493.
- ^ See Hexafluoroethane (data page)
- ^ "Protium | isotope | Britannica". 27 January 2024.
- ^ Walas, S. M. (1990). Chemical Process Equipment – Selection and Design. Amsterdam: Elsevier. p. 639. ISBN 0-7506-7510-1.
- ^ See Isobutane (data page)
- ^ "Silane-Gas Encyclopedia". Gas Encyclopedia. Air Liquide.
External links
 Media related to Triple point at Wikimedia Commons
Media related to Triple point at Wikimedia Commons