Treponema denticola
| Treponema denticola | |
|---|---|
| Scientific classification | |
| Domain: | Bacteria |
| Phylum: | Spirochaetota |
| Class: | Spirochaetia |
| Order: | Spirochaetales |
| Family: | Treponemataceae |
| Genus: | Treponema |
| Species: | T. denticola
|
| Binomial name | |
| Treponema denticola (ex Flügge 1886) Chan et al. 1993[1]
| |
Treponema denticola is a Gram-negative, obligate anaerobic, motile and highly proteolytic spirochete bacterium. It is one of four species of oral spirochetes to be reliably cultured, the others being Treponema pectinovorum, Treponema socranskii and Treponema vincentii[2]. T. denticola dwells in a complex and diverse microbial community within the oral cavity and is highly specialized to survive in this environment. T. denticola is associated with the incidence and severity of human periodontal disease. Treponema denticola is one of three bacteria that form the Red Complex, the other two being Porphyromonas gingivalis and Tannerella forsythia. Together they form the major virulent pathogens that cause chronic periodontitis.[3] Having elevated T. denticola levels in the mouth is considered one of the main etiological agents of periodontitis.[4][5] T. denticola is related to the syphilis-causing obligate human pathogen, Treponema pallidum subsp. pallidum. It has also been isolated from women with bacterial vaginosis.[6]
Genome
The first genome of T. denticola to be sequenced was strain 35405 which was initially isolated and designated as the type strain by Chan et al.[7] The 2,843,201-bp genome sequence encodes 2,786 open reading frames (ORFs) as well a 6 rRNAs and 44 tRNAs. This is in stark contrast to the minimal genome of Treponema pallidum which encodes only 1,040 ORFs.[8]
Cell structure
The native cellular architecture of the bacterial cell was revealed using cryo-electron tomography, a technique observing intact plunge-frozen cells.[9] Tightly organized bundles of periplasmic flagella were observed in the periplasm, a characteristic of spirochetes. The presence of the periplasmic flagella is widely regarded as being responsible for the microorganism's ability to rotate and flex, in addition to its translational movement.[2] Spirochetes, including Treponema denticola , are therefore able to navigate through particularly viscous environments, unlike that of other prokaryotes.[2]
The tapering of the cell ends, conserved between cells, encompass a patella-shaped structure observed in the periplasm at the cell tip. This structure might be involved in the polar attachment of the cells.[10] Cytoplasmic filaments, a bacterial intermediate-like filaments composed of the protein CfpA, are adjacent to the inner membrane and run parallel to the tightly organized flagellar filaments.[9][11] Those cytoplasmic filaments are critical for pathogenicity, directly or indirectly as they are also involved in chromosome structure, segregation, or the cell division processes.[12][13] The absence of either flagella or cytoplasmic filaments in deficient mutant increase the mouse peritoneal macrophages in vitro uptake.[14]
Adherence and cytotoxicity
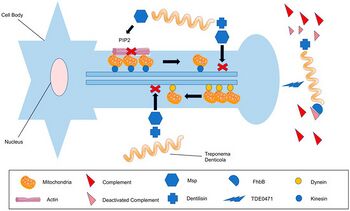
The main site for T. denticola habitation in the oral cavity is the gingival crevice. These spirochetes attach to proteins (including fibronectin and collagen) of local gingival fibroblasts, binding to their plasma membrane. A 53-kDa surface protein on T. denticola is responsible for transporting its components into the host cell, exhibiting a cytotoxic effect. Accumulation of T. denticola in this manner facilitates the disease-causing process, including membrane blebbing and red blood cell lysis.[2]
Disease
Periodontal disease
Periodontal disease is a type of gum disease caused by the accumulation of plaque on the teeth due to poor oral hygiene. Plaque is a sticky substance that contains bacteria which can harden into a substance called calculus, irritating the gums.[15]
[16]Treponema denticola, a subgingival oral spirochete has been associated with many periodontal disease conditions such as: the early stage of periodontitis, acute pericoronitis (infection under the gum tissue covering a partially erupted tooth)3, as well as necrotising ulcerative gingivitis (severe inflammation of the gum more common in immunocompromised patients). It relates to lesions limited to gingival tissue.[17]
Clinical evidence and research shows that periodontal pockets contain large numbers of treponema denticola together with other proteolytic gram negative bacteria, playing an important role in the development of periodontal disease. The toxic products of these bacteria, especially treponema denticola may damage the surface lining periodontal cells making them more prone to damage as well as lysis. Treponema denticola attaches to fibroblasts and epithelial cells as well as to extracellular matrix components which are found in periodontal tissues and release its own bacterial contents. The bacterial components are:[citation needed]
- Outer sheath associated peptidases
- Chymotrypsin proteinases
- Trypsin proteinases
- Hemolytic activities
- Hemagglutinating activities
- outer-sheath protein with pore-forming properties
A number of studies have observed an increase of T. denticola in patients with orthodontic appliances, particularly the fixed type.[18]
Oral cancer
Treponema denticola is a potential etiological bacterial agent for oral cancer.[19] It encourages oncogenesis (process in which healthy cells become cancer cells) and therefore the progression of oral cancer through chronic inflammation advancing invasiveness of the cancer cells. This results in the ceasing of cell apoptosis (inhibition of controlled cell death – a safety mechanism within cells to stop more damage from occurring), resulting in rapid growth and multiplication of cancer cells. This suppresses the immune system stopping the body from recognising the cancerous cells and as a result more cancer-promoting substances are produced.[19] The presence of T. denticola along with other periodontal pathogens and bacterial diversity within the oral cavity are important factors contributing to cancerous cells (including precancerous gastric lesions).[20]
Vascular disease
Atherosclerotic vascular disease is chronic inflammatory disease of large arteries distinguished by invasion, proliferation and accumulation of cells from arterial smooth muscle cells, and the circulating blood in the intimal layer with deposition of connective tissue and lipids, if left to get worse atherosclerotic vascular disease can result in cardiovascular disease. Cardiovascular disease (including angina, heart attack, arrhythmias, heart failure, strokes) is the leading cause of death globally. [citation needed]
Substantial evidence between inflammation from infectious agents and development of atherosclerosis – periodontal pathogens prominent contenders due to chronic inflammation related with periodontal disease.[citation needed]
Treponema denticola bacteria can penetrate gingival tissues and circulate through blood vessels, with opportunity to invade the heart and cardiovascular epithelium in medium to large arteries – including aorta, coronary and carotid arteries.[21]
Pancreatic cancer
It has been recognised that there is an association between oral and intestinal cancer related deaths and chronic periodontitis. This is based on the high amounts of tumor suppressor gene p53 mutations and k-ras arginine mutations found in patients with pancreatic cancer. Peptidylarginine deiminases from oral bacteria are thought to be responsible for these mutations. This means that there could be a causative link between oral bacteria, particularly the red complex, and pancreatic cancer.[3]
See also
References
- ↑ Parte AC. "Treponema". LPSN. Archived from the original on 2022-01-27. Retrieved 2022-02-04.
- ↑ 2.0 2.1 2.2 2.3 Chan EC, McLaughlin R (February 2000). "Taxonomy and virulence of oral spirochetes". Oral Microbiology and Immunology. 15 (1): 1–9. doi:10.1034/j.1399-302x.2000.150101.x. PMID 11155157.
- ↑ 3.0 3.1 Öğrendik M (March 2017). "Periodontal Pathogens in the Etiology of Pancreatic Cancer". Gastrointestinal Tumors. 3 (3–4): 125–127. doi:10.1159/000452708. PMC 5465713. PMID 28611978.
- ↑ Marie-Claude Jobin, Mohsen Amin, Richard P. Ellen (2008). "Chapter 8 - The Molecular Biology of the Survival and Virulence of Treponema denticola". In Anthony H. Rogers (ed.). Molecular Oral Microbiology. Caister Academic Press. p. 177. ISBN 978-1-904455-24-0.
{{cite book}}: CS1 maint: uses authors parameter (link) - ↑ Simonson LG, Goodman CH, Bial JJ, Morton HE (April 1988). "Quantitative relationship of Treponema denticola to severity of periodontal disease". Infection and Immunity. 56 (4): 726–8. doi:10.1128/IAI.56.4.726-728.1988. PMC 259361. PMID 3346072.
- ↑ Africa CW, Nel J, Stemmet M (July 2014). "Anaerobes and bacterial vaginosis in pregnancy: virulence factors contributing to vaginal colonisation". International Journal of Environmental Research and Public Health. 11 (7): 6979–7000. doi:10.3390/ijerph110706979. PMC 4113856. PMID 25014248.
- ↑ Chan EC, Siboo R, Keng T, Psarra N, Hurley R, Cheng SL, Iugovaz I (April 1993). "Treponema denticola (ex Brumpt 1925) sp. nov., nom. rev., and identification of new spirochete isolates from periodontal pockets". International Journal of Systematic Bacteriology. 43 (2): 196–203. doi:10.1099/00207713-43-2-196. PMID 8494734.
- ↑ Seshadri R, Myers GS, Tettelin H, Eisen JA, Heidelberg JF, Dodson RJ, et al. (April 2004). "Comparison of the genome of the oral pathogen Treponema denticola with other spirochete genomes". Proceedings of the National Academy of Sciences of the United States of America. 101 (15): 5646–51. Bibcode:2004PNAS..101.5646S. doi:10.1073/pnas.0307639101. PMC 397461. PMID 15064399.
- ↑ 9.0 9.1 Izard J, Hsieh CE, Limberger RJ, Mannella CA, Marko M (July 2008). "Native cellular architecture of Treponema denticola revealed by cryo-electron tomography". Journal of Structural Biology. 163 (1): 10–7. doi:10.1016/j.jsb.2008.03.009. PMC 2519799. PMID 18468917.
- ↑ Ellen RP, Dawson JR, Yang PF (April 1994). "Treponema denticola as a model for polar adhesion and cytopathogenicity of spirochetes". Trends in Microbiology. 2 (4): 114–9. doi:10.1016/0966-842x(94)90597-5. PMID 8012753.
- ↑ Izard J, McEwen BF, Barnard RM, Portuese T, Samsonoff WA, Limberger RJ (February 2004). "Tomographic reconstruction of treponemal cytoplasmic filaments reveals novel bridging and anchoring components". Molecular Microbiology. 51 (3): 609–18. doi:10.1046/j.1365-2958.2003.03864.x. PMID 14731266. Archived from the original on 2022-05-04. Retrieved 2022-02-04.
- ↑ Izard J, Sasaki H, Kent R (2012). "Pathogenicity of Treponema denticola Wild-Type and Mutant Strain Tested by an Active Mode of Periodontal Infection Using Microinjection". International Journal of Dentistry. 2012: 549169. doi:10.1155/2012/549169. PMC 3398590. PMID 22829826.
- ↑ Izard J, Samsonoff WA, Limberger RJ (February 2001). "Cytoplasmic filament-deficient mutant of Treponema denticola has pleiotropic defects". Journal of Bacteriology. 183 (3): 1078–84. CiteSeerX 10.1.1.488.5178. doi:10.1128/JB.183.3.1078-1084.2001. PMC 94976. PMID 11208807.
- ↑ Gaibani P, Vocale C, Ambretti S, Cavrini F, Izard J, Miragliotta L, et al. (May 2010). "Killing of Treponema denticola by mouse peritoneal macrophages". Journal of Dental Research. 89 (5): 521–6. doi:10.1177/0022034510363105. PMC 2868591. PMID 20200417.
- ↑ "Periodontal Care". SDCEP. Archived from the original on 2019-12-11. Retrieved 2019-12-11.
- ↑ Sela MN (2001). "Role of Treponema denticola in periodontal diseases". Critical Reviews in Oral Biology and Medicine. 12 (5): 399–413. doi:10.1177/10454411010120050301. PMID 12002822.
- ↑ "Management of acute dental problems. Guidance for healthcare professionals" (PDF). Archived (PDF) from the original on 2022-02-04. Retrieved 2022-02-04.
- ↑ Lucchese A, Bondemark L, Marcolina M, Manuelli M (January 2018). "Changes in oral microbiota due to orthodontic appliances: a systematic review". Journal of Oral Microbiology. 10 (1): 1476645. doi:10.1080/20002297.2018.1476645. PMC 6032020. PMID 29988826.
- ↑ 19.0 19.1 Nieminen MT, Listyarifah D, Hagström J, Haglund C, Grenier D, Nordström D, et al. (February 2018). "Treponema denticola chymotrypsin-like proteinase may contribute to orodigestive carcinogenesis through immunomodulation". British Journal of Cancer. 118 (3): 428–434. doi:10.1038/bjc.2017.409. PMC 5808028. PMID 29149107.
- ↑ Zhang WL, Wang SS, Wang HF, Tang YJ, Tang YL, Liang XH (November 2019). "Who is who in oral cancer?". Experimental Cell Research. 384 (2): 111634. doi:10.1016/j.yexcr.2019.111634. PMID 31541617.
- ↑ Chukkapalli SS, Rivera MF, Velsko IM, Lee JY, Chen H, Zheng D, et al. (May 2014). "Invasion of oral and aortic tissues by oral spirochete Treponema denticola in ApoE(-/-) mice causally links periodontal disease and atherosclerosis". Infection and Immunity. 82 (5): 1959–67. doi:10.1128/IAI.01511-14. PMC 3993427. PMID 24566627.
- CS1 maint: uses authors parameter
- Articles with 'species' microformats
- All articles with unsourced statements
- Articles with unsourced statements from January 2021
- Articles with invalid date parameter in template
- Dentistry
- Spirochaetes
- Treponema
- Bacterial vaginosis
- Bacteria described in 1886
- Periodontal disorders