Topical steroid
 | |
| Names | |
|---|---|
| Other names | Topical corticosteroids |
Topical steroids are steroid creams that are applied to skin, generally used to treat inflammation of the skin such as eczema, particularly when simple moisturisers do not work.[1] Topical steroids are the most commonly prescribed topical medications for the treatment of rash, eczema, and dermatitis. Topical steroids have anti-inflammatory properties, and are classified based on their skin vasoconstrictive abilities.[2] There are numerous topical steroid products. All the preparations in each class have the same anti-inflammatory properties, but essentially differ in base and price.
Side effects may occur from long-term topical steroid use.[3]
Medical uses
Weaker topical steroids are utilized for thin-skinned and sensitive areas, especially areas under occlusion, such as the armpit, groin, buttock crease, and breast folds. Weaker steroids are used on the face, eyelids, diaper area, perianal skin, and intertrigo of the groin or body folds. Moderate steroids are used for atopic dermatitis, nummular eczema, xerotic eczema, lichen sclerosis et atrophicus of the vulva, scabies (after scabiecide) and severe dermatitis. Strong steroids are used for psoriasis, lichen planus, discoid lupus, chapped feet, lichen simplex chronicus, severe poison ivy exposure, alopecia areata, nummular eczema, and severe atopic dermatitis in adults.[2]
To prevent tachyphylaxis, a topical steroid is often prescribed to be used on a week on, week off routine. Some recommend using the topical steroid for 3 consecutive days on, followed by 4 consecutive days off.[4] Long-term use of topical steroids can lead to secondary infection with fungus or bacteria (see tinea incognito), skin atrophy, telangiectasia (prominent blood vessels), skin bruising and fragility.[5]
The use of the finger tip unit may be helpful in guiding how much topical steroid is required to cover different areas of the body.
Side effects
- Hypothalamic–pituitary–adrenal axis (HPA) suppression[6]
- Cushing's syndrome
- Diabetes mellitus[7]
- Osteoporosis
- Topical steroid addiction
- Allergic contact dermatitis (see steroid allergy)
- Steroid atrophy
- Perioral dermatitis: This is a rash that occurs around the mouth and the eye region that has been associated with topical steroids.
- Ocular effects: Topical steroid drops are frequently used after eye surgery but can also raise intraocular pressure (IOP) and increase the risk of glaucoma, cataract, retinopathy as well as systemic adverse effects.[8]
- Tachyphylaxis: The acute development of tolerance to the action of a drug after repeated doses.[9] Significant tachyphylaxis can occur by day 4 of therapy. Recovery usually occurs after 3 to 4 days' rest. This has led to therapies such as 3 days on, 4 days off; or one week on therapy, and one week off therapy.
- Delivery-related adverse effects
- Other local adverse effects: These include facial hypertrichosis, folliculitis, miliaria, genital ulcers, and granuloma gluteale infantum. Long-term use has resulted in Norwegian scabies, Kaposi's sarcoma, and other unusual dermatosis.[10]
-
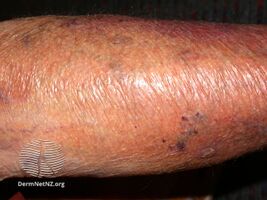
Skin thinning due to topical steroids
-
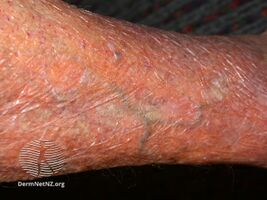
Skin thinning due to topical steroids
Safety in pregnancy
A 2015 meta-analysis of observational studies of pregnancies found no association between mothers' use of topical steroids and type of delivery, APGAR score, birth defects, or prematurity.[11]
Classification systems
USA system
The USA system utilizes 7 classes, which are classified by their ability to constrict capillaries and cause skin blanching. Class I is the strongest, or superpotent. Class VII is the weakest and mildest.[12]
Class I
Very potent: up to 600 times stronger than hydrocortisone
- Clobetasol propionate 0.05% (Dermovate)
- Betamethasone dipropionate 0.25% (Diprolene)
- Halobetasol propionate 0.05% (Ultravate, Halox)
- Diflorasone diacetate 0.05% (Psorcon)
Class II
- Fluocinonide 0.05% (Lidex)
- Halcinonide 0.05% (Halog)
- Amcinonide 0.05% (Cyclocort)
- Desoximetasone 0.25% (Topicort)
Class III
- Triamcinolone acetonide 0.5% (Kenalog, Aristocort cream)
- Mometasone furoate 0.1% (Elocon, Elocom ointment)
- Fluticasone propionate 0.005% (Cutivate)
- Betamethasone dipropionate 0.05% (Diprosone)
- Halometasone 0.05%
Class IV
- Fluocinolone acetonide 0.01-0.2% (Synalar, Synemol, Fluonid)
- Hydrocortisone valerate 0.2% (Westcort)
- Hydrocortisone butyrate 0.1% (Locoid)
- Flurandrenolide 0.05% (Cordran)
- Triamcinolone acetonide 0.1% (Kenalog, Aristocort A ointment)
- Mometasone furoate 0.1% (Elocon cream, lotion)
Class V
- Fluticasone propionate 0.05% (Cutivate cream)
- Desonide 0.05% (Tridesilon, DesOwen ointment)
- Fluocinolone acetonide 0.025% (Synalar, Synemol cream)
- Hydrocortisone valerate 0.2% (Westcort cream)
Class VI
- Alclometasone dipropionate 0.05% (Aclovate cream, ointment)
- Triamcinolone acetonide 0.025% (Aristocort A cream, Kenalog lotion)
- Fluocinolone acetonide 0.01% (Capex shampoo, Dermasmooth)
- Desonide 0.05% (DesOwen cream, lotion)
Class VII
The weakest class of topical steroids. Has poor lipid permeability, and can not penetrate mucous membranes well.
- Hydrocortisone 2.5% (Hytone cream, lotion, ointment)
- Hydrocortisone 1% (Many over-the-counter brands)
Other countries
Most other countries, such as the United Kingdom, Germany, the Netherlands, New Zealand, recognize only 4 classes.[13] In the United Kingdom and New Zealand I is the strongest, while in Continental Europe, class IV is regarded as the strongest.
Class IV (UK/NZ: class I)
Very potent (up to 600 times as potent as hydrocortisone)
- Clobetasol propionate (Dermovate Cream/Ointment, Exel Cream)
- Betamethasone dipropionate (Diprosone OV Cream/Ointment, Diprovate Cream)
Class III (UK/NZ: class II)
Potent (50–100 times as potent as hydrocortisone)
- Betamethasone valerate (Beta Cream/Ointment/Scalp Application, Betnovate Lotion/C Cream/C Ointment, Fucicort)
- Betamethasone dipropionate (Diprosone Cream/Ointment, Diprovate Cream, Daivobet 50/500 Ointment)
- Diflucortolone valerate (Nerisone C/Cream/Fatty Ointment/Ointment)
- Hydrocortisone 17-butyrate (Locoid C/Cream/Crelo Topical Emulsion/Lipocream/Ointment/Scalp Lotion)
- Mometasone furoate (Elocon Cream/Lotion/Ointment)
- Methylprednisolone aceponate (Advantan Cream/Ointment)
- Halometasone 0.05%
Class II (UK/NZ: class III)
Moderate (2–25 times as potent as hydrocortisone)
- Clobetasone butyrate (Eumovate Cream)
- Triamcinolone acetonide (Aristocort Cream/Ointment, Viaderm KC Cream/Ointment, Kenacomb Ointment)
Class I (UK/NZ: class IV)
Mild
- Hydrocortisone 0.5–2.5% (DermAid Cream/Soft Cream, DP Lotion-HC 1%, Skincalm, Lemnis Fatty Cream HC, Pimafucort Cream/Ointment)
Japan classification
Japan rates topical steroids from 1 to 5, with 1 being strongest.
Allergy associations
The highlighted steroids are often used in the screening of allergies to topical steroid and systemic steroids.[14] When one is allergic to one group, one is allergic to all steroids in that group.
Group A
Hydrocortisone, hydrocortisone acetate, cortisone acetate, tixocortol pivalate, prednisolone, methylprednisolone, and prednisone
Group B
Triamcinolone acetonide, triamcinolone alcohol, amcinonide, budesonide, desonide, fluocinonide, fluocinolone acetonide, and halcinonide
Group C
Betamethasone, betamethasone sodium phosphate, dexamethasone, dexamethasone sodium phosphate, and fluocortolone
Group D
Hydrocortisone 17-butyrate, hydrocortisone-17-valerate, alclometasone dipropionate, betamethasone valerate, betamethasone dipropionate, prednicarbate, clobetasone-17-butyrate, Clobetasol-17 propionate, fluocortolone caproate, fluocortolone pivalate, fluprednidene acetate, and mometasone furoate
History
Topical steroids were first used successfully in 1952.[15]
See also
References
- ↑ Hitchings, Andrew; Lonsdale, Dagan; Burrage, Daniel; Baker, Emma (2019). The Top 100 Drugs: Clinical Pharmacology and Practical Prescribing (2nd ed.). Elsevier. pp. 112–113. ISBN 978-0-7020-7442-4. Archived from the original on 2021-05-22. Retrieved 2021-11-09.
- ↑ 2.0 2.1 Habif, Thomas P. (1990). Clinical dermatology: a color guide to diagnosis and therapy (2nd ed.). St. Louis: Mosby. p. 27. ISBN 0-8016-2465-7.
- ↑ Coondoo, A; Phiske, M; Verma, S; Lahiri, K (2014). "Side effects of topical steroids: A long overdue revisit". Indian Dermatol Online J. 5 (4): 416–25. doi:10.4103/2229-5178.142483. PMC 4228634. PMID 25396122.
- ↑ Recommendations from New Zealand Dermatological Society Incorporated on corticosteroids Archived 2016-07-08 at the Wayback Machine
- ↑ Habif, Thomas P. (1990). Clinical dermatology: a color guide to diagnosis and therapy (2nd ed.). St. Louis: Mosby. pp. 27–30. ISBN 0-8016-2465-7.
- ↑ Fisher, DA (1995). "Adverse effects of topical corticosteroid use". West. J. Med. 162 (2): 123–6. PMC 1022645. PMID 7794369.
- ↑ van der Linden MW, Penning-van Beest FJ, Nijsten T, Herings RM (2009). "Topical corticosteroids and the risk of diabetes mellitus: a nested case-control study in the Netherlands". Drug Saf. 32 (6): 527–37. doi:10.2165/00002018-200932060-00008. PMID 19459719. Archived from the original on 2021-08-29. Retrieved 2020-02-11.
- ↑ Lebreton, O.; Weber, M. (2011). "Complications ophtalmologiques des corticoïdes systémiques". La Revue de Médecine Interne. 32 (8): 506–512. doi:10.1016/j.revmed.2011.01.003. PMID 21330017.
- ↑ Wolverton, Stephen E. (2001). Comprehensive Dermatologic Drug Therapy. Philadelphia, PA: W.B. Saunders Company. pp. 562–3. ISBN 0-7216-7728-2.
- ↑ Wolverton, Stephen E. (2001). Comprehensive Dermatologic Drug Therapy. Philadelphia, PA: W.B. Saunders Company. p. 563. ISBN 0-7216-7728-2.
- ↑ Chi, Ching-Chi; Wang, Shu-Hui; Wojnarowska, Fenella; Kirtschig, Gudula; Davies, Emily; Bennett, Cathy (2015-10-26). "Safety of topical corticosteroids in pregnancy". Cochrane Database of Systematic Reviews. doi:10.1002/14651858.CD007346.pub3. ISSN 1465-1858. Archived from the original on 2020-08-15. Retrieved 2020-02-11.
- ↑ Habif, Thomas P. (1990). Clinical dermatology: a color guide to diagnosis and therapy (2nd ed.). St. Louis: Mosby. p. Inside front cover. ISBN 0-8016-2465-7.
- ↑ "Archive copy". Archived from the original on 2016-07-25. Retrieved 2020-02-11.
{{cite web}}: CS1 maint: archived copy as title (link) - ↑ Wolverton, Stephen E. (2001). Comprehensive Dermatologic Drug Therapy. Philadelphia, PA: W.B. Saunders Company. p. 562. ISBN 0-7216-7728-2.
- ↑ Mukhopadhyay, Sandip; Kwatra, Gangandeep (2018). "1. Evolution and development of topical corticosteroids". In Lahiri, Koushik (ed.). A Treatise on Topical Corticosteroids in Dermatology: Use, Misuse and Abuse. Singapore: Springer. p. 1. ISBN 978-981-10-4608-7. Archived from the original on 2021-08-29. Retrieved 2021-06-13.
- Pages using duplicate arguments in template calls
- Webarchive template wayback links
- CS1 maint: archived copy as title
- Chemical articles with unknown parameter in Infobox drug
- Infobox drug articles with non-default infobox title
- Chemical articles without CAS registry number
- Articles without EBI source
- Chemical pages without ChemSpiderID
- Chemical pages without DrugBank identifier
- Articles without KEGG source
- Articles without InChI source
- Articles without UNII source
- Drugs missing an ATC code
- Drugs with no legal status
- Articles containing unverified chemical infoboxes
- Articles with hatnote templates targeting a nonexistent page
- Corticosteroids