Saline (medicine)
 Saline solution for intravenous infusion. | |
| Clinical data | |
|---|---|
| Main uses | Dry eyes, dehydration, diabetic ketoacidosis[1] |
| Side effects | Fluid overload, swelling, acidosis, high blood sodium[2][1] |
| Routes of use | Intravenous, topical, subcutaneous |
| Defined daily dose | Not established[3] |
| External links | |
| AHFS/Drugs.com | FDA Professional Drug Information |
| Legal | |
| License data | |
| Chemical and physical data | |
| 3D model (JSmol) | |
| |
| |
Saline, also known as saline solution, is a mixture of sodium chloride in water and has a number of uses in medicine.[2] Applied to the affected area it is used to clean wounds, help remove contact lenses, and help with dry eyes.[1] By injection into a vein it is used to treat dehydration such as from gastroenteritis and diabetic ketoacidosis.[1] It is also used to dilute other medications to be given by injection.[2]
Large amounts may result in fluid overload, swelling, acidosis, and high blood sodium.[2][1] In those with long-standing low blood sodium, excessive use may result in osmotic demyelination syndrome.[1] Saline is in the crystalloid family of medications.[4] It is most commonly used as a sterile 9 g of salt per litre (0.9%) solution, known as normal saline.[2] Higher and lower concentrations may also occasionally be used.[5][6] Saline has a pH of 5.5 (mainly due to dissolved carbon dioxide) making it acidic.[7]
The medical use of saline began around 1831.[8] It is on the World Health Organization's List of Essential Medicines.[9] The wholesale cost in the developing world is about US$0.60–4.20 per liter of normal saline.[10] In 2017, sodium salts were the 225th most commonly prescribed medication in the United States, with more than two million prescriptions.[11][12]
Medical use
Normal
For medical purposes, saline is often used to flush wounds and skin abrasions. Normal saline will not burn or sting when applied.
Saline is also used in intravenous therapy, intravenously supplying extra water to rehydrate people or supplying the daily water and salt needs ("maintenance" needs) of a person who is unable to take them by mouth. Because infusing a solution of low osmolality can cause problems such as hemolysis, intravenous solutions with reduced saline concentrations (less than 0.9%) typically have dextrose (glucose) added to maintain a safe osmolality while providing less sodium chloride.
Nose
There is tentative evidence that saline nasal irrigation may help with long term cases of rhinosinusitis.[13] Evidence for use in cases of rhinosinusitis of short duration is unclear.[14] The solution exerts a softening and loosening influence on the mucus to make it easier to wash out and clear the nasal passages in babies.[15] In this case "home-made" saline may be used: this is made by dissolving approximately half a teaspoon of table salt into 240ml (approx. 8 ounces) of clean tap water.[16] In very rare instances, amoeba Naegleria fowleri infection can occur if amoeba enters the body through the nose, therefore water used for nasal irrigation should be sterile.[17]
Dosage
The defined daily dose is not established (by injection).[3]
Hypertonic saline
3% hypertonic saline is used in critical care for acutely increased intracranial pressure and severe hyponatremia (sodium less than 120 mmol/L).[18] [19]
For severe hyponatremia doses of 100 mL over 10 min of 3% hypertonic saline can be given, which can be repeated if needed.[20] Sodium should generally not be corrected more than 6 mEq/L per day.[20]
For increased intracranial pressure a dose of 3 to 5 mls per kg (250 ml for an adult) over 10 to 20 min of 3% saline may be used.[21][22]
There is tentative evidence of benefit for nebulized hypertonic saline in cystic fibrosis.[23]
-
Vial of 23.4% sodium chloride
-
Bag of 3% sodium chloride
Concentrations
Concentrations vary from low to normal to high. High concentrations are used rarely in medicine but frequently in molecular biology.
Normal
Normal saline (NSS, NS or N/S) is the commonly used phrase for a solution of 0.90% w/v of NaCl, 308 mOsm/L or 9.0 g per liter. Less commonly, this solution is referred to as physiological saline or isotonic saline (because it closely approximates isotonic, that is, physiologically normal, solution); although neither of those names is technically accurate (because normal saline is not exactly like blood serum), they convey the practical effect usually seen: good fluid balance with minimal hypotonicity or hypertonicity. NS is used frequently in intravenous drips (IVs) for people who cannot take fluids orally and have developed or are in danger of developing dehydration or hypovolemia. NS is also used for aseptic purpose. NS is typically the first fluid used when hypovolemia is severe enough to threaten the adequacy of blood circulation, and has long been believed to be the safest fluid to give quickly in large volumes. However, it is now known that rapid infusion of NS can cause metabolic acidosis.[24]
The solution is 9 grams of sodium chloride (NaCl) dissolved in water, to a total volume of 1000 ml (weight per unit volume(w/v)). The mass of 1 millilitre of normal saline is 1.0046 gram at 22 °C.[25][26] The molecular weight of sodium chloride is approximately 58.5 grams per mole, so 58.5 grams of sodium chloride equals 1 mole. Since normal saline contains 9 grams of NaCl, the concentration is 9 grams per litre divided by 58.5 grams per mole, or 0.154 mole per litre. Since NaCl dissociates into two ions – sodium and chloride – 1 molar NaCl is 2 osmolar. Thus, NS contains 154 mEq/L of Na+ and the same amount of Cl−. This points to an osmolality of 154 + 154 = 308, which is higher (i.e. more solute per litre) than that of blood (approximately 285).[27] However, if the osmotic coefficient (a correction for non-ideal solutions) is taken into account, then the saline solution is much closer to isotonic. The osmotic coefficient of NaCl is about 0.93,[28] which yields an osmolality of 0.154 × 1000 × 2 × 0.93 = 286.44. Therefore, the osmolarity of normal saline is a close approximation to the osmolarity of blood.
Other
Other concentrations commonly used include:
- Half-normal saline (0.45% NaCl), often with "D5" (5% dextrose), contains 77 mEq/L of Na and Cl and 50 g/L dextrose.
- Quarter-normal saline (0.22% NaCl) has 39 mEq/L of Na and Cl and almost always contains 5% dextrose for osmolality reasons. It can be used alone in Neonatal Intensive Care Units.
- Hypertonic saline may be used in perioperative fluid management protocols to reduce excessive intravenous fluid infusions and lessen pulmonary complications.[29] Hypertonic saline is used in treating hyponatremia and cerebral edema Rapid correction of hyponatremia via hypertonic saline, or via any saline infusion > 40 mmol/L (Na+ having a valence of 1, 40 mmol/L = 40 mEq/L) greatly increases risk of central pontine myelinolysis (CPM), and so requires constant monitoring of the person's response. Water privation combined with diuretic block does not produce as much risk of CPM as saline administration does; however, it does not correct hyponatremia as rapidly as administration of hypertonic saline does. Due to hypertonicity, administration may result in phlebitis and tissue necrosis. As such, concentrations greater than 3% NaCl should normally be administered via a central venous catheter, also known as a 'central line'. Such hypertonic saline is normally available in two strengths, the former of which is more commonly administered:
- 3% NaCl has 513 mEq/L of Na and Cl.
- 5% NaCl has 856 mEq/L of Na and Cl.
- NaCl solutions that are less commonly used are 7% (1200 mEq/L) and 23.4% (approx 4000 mEq/L), both of which are used (also via central line), often in conjunction with supplementary diuretics, in the treatment of traumatic brain injury.[30]
- Dextrose (glucose) 4% in 0.18% saline is used sometimes for maintenance replacement.
Solutions with added ingredients
In medicine, common types of salines include:
- Lactated Ringer's solution
- Acetated Ringer's solution
- Intravenous sugar solutions
- 5% dextrose in normal saline (D5NS)
- 10% dextrose in normal saline (D10NS)
- 5% dextrose in half-normal saline (D5HNS)
- 10% dextrose in half-normal saline (D10HNS)
And in cell biology, in addition to the above the following are used:
- Phosphate buffered saline (PBS) (recipes from Dulbecco = D-PBS, Galfre, Kuchler, Ausubel etc.)
- TRIS-buffered saline (TBS) (recipes from Goldsmith, Ausubel etc.)
- Hank's balanced salt solution (HBSS)
- Earle's balanced salt solution (EBSS)
- Standard saline citrate (SSC)
- HEPES-buffered saline (HBS) (recipes from Dittmar, Liu, Ausubel etc.)
- Gey's balanced salt solution (GBSS)
History
Replacing fluids intravenously was believed to have originated during the 1826–1837 cholera pandemic. After analysing the blood and stool of people with cholera, William Brooke O'Shaughnessy, a recent graduate of Edinburgh Medical School working in India, proposed in an article to The Lancet to inject severely dehydrated cholera affected people with highly oxygenated salts.[31] He found his treatment harmless in dogs, and his proposal was soon adopted by the physician Thomas Aitchison Latta in treating people with cholera to beneficial effect.[32][33] In the following decades, variations and alternatives to Latta's solution were tested and used in treating people with cholera. These solutions contained a range of concentrations of sodium, chloride, potassium, carbonate, phosphate, and hydroxide. The breakthrough in achieving physiological concentrations was accomplished by Sydney Ringer in the early 1880s,[34] when he determined the optimal salt concentrations to maintain the contractility of frog heart muscle tissue. Normal saline is considered a descendant of the pre-Ringer solutions, as Ringer's findings were not adopted and widely used until decades later. The term "normal saline" itself appears to have little historical basis, except for studies done in 1882–83 by Dutch physiologist Hartog Jacob Hamburger; these in vitro studies of red cell lysis suggested incorrectly that 0.9% was the concentration of salt in human blood (rather than 0.6%, the true concentration).[35]
Normal saline has become widely used in modern medicine, but due to the mismatch with real blood, other solutions have proved better. The 2018 publication of a randomized, controlled trial with 15,000 people showed that lactated Ringer's solution reduced mortality risk of people in intensive care unit by 1% compared to normal saline, which given the large number of people is a significant reduction.[36]
Society and culture
Coconut water has been used in place of normal saline in areas without access to normal saline.[37] Its use, however, has not been well studied.[37]
Cost
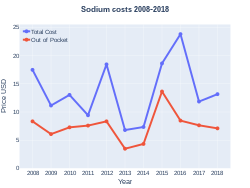
The wholesale cost in the developing world is about US$0.60–4.20 per liter of normal saline.[10] In 2017, sodium salts were the 225th most commonly prescribed medication in the United States, with more than two million prescriptions.[11][12] Although inexpensive, the accompanying equipment required to administer saline can escalate the cost.[38]
-
Sodium salts costs (US)
-
Sodium salt prescriptions (US)
See also
References
- ↑ 1.0 1.1 1.2 1.3 1.4 1.5 British national formulary : BNF 69 (69 ed.). British Medical Association. 2015. pp. 683, 770. ISBN 9780857111562.
- ↑ 2.0 2.1 2.2 2.3 2.4 "Sodium Chloride Injection - FDA prescribing information, side effects and uses". www.drugs.com. Archived from the original on 18 January 2017. Retrieved 14 January 2017.
- ↑ 3.0 3.1 "WHOCC - ATC/DDD Index". www.whocc.no. Archived from the original on 1 July 2021. Retrieved 13 September 2020.
- ↑ Marini, John J.; Wheeler, Arthur P. (2010). Critical Care Medicine: The Essentials. Lippincott Williams & Wilkins. p. 54. ISBN 9780781798396. Archived from the original on 18 September 2017.
- ↑ "Hypertonic Saline - FDA prescribing information, side effects and uses". www.drugs.com. Archived from the original on 18 January 2017. Retrieved 14 January 2017.
- ↑ Pestana, Carlos (2000). Fluids and Electrolytes in the Surgical Patient. Lippincott Williams & Wilkins. p. 11. ISBN 9780781724258. Archived from the original on 18 September 2017.
- ↑ Reddi, BA (2013). "Why is saline so acidic (and does it really matter?)". International Journal of Medical Sciences. 10 (6): 747–50. doi:10.7150/ijms.5868. PMC 3638298. PMID 23630439.
- ↑ Bozzetti, Federico; Staun, Michael; Gossum, Andre van (2014). Home Parenteral Nutrition, 2nd Edition. CABI. p. 4. ISBN 9781780643113. Archived from the original on 18 September 2017.
- ↑ World Health Organization (2019). World Health Organization model list of essential medicines: 21st list 2019. Geneva: World Health Organization. hdl:10665/325771. WHO/MVP/EMP/IAU/2019.06. License: CC BY-NC-SA 3.0 IGO.
- ↑ 10.0 10.1 "Sodium Chloride in Water". International Drug Price Indicator Guide. Archived from the original on 22 January 2018. Retrieved 8 December 2016.
- ↑ 11.0 11.1 "The Top 300 of 2020". ClinCalc. Archived from the original on 18 March 2020. Retrieved 11 April 2020.
- ↑ 12.0 12.1 "Sodium - Drug Usage Statistics". ClinCalc. Archived from the original on 8 July 2020. Retrieved 11 April 2020.
- ↑ Succar, EF; Turner, JH; Chandra, RK (May 2019). "Nasal saline irrigation: a clinical update". International Forum of Allergy & Rhinology. 9 (S1): S4–S8. doi:10.1002/alr.22330. PMID 31087631.
- ↑ Achilles, N; Mösges, R (April 2013). "Nasal saline irrigations for the symptoms of acute and chronic rhinosinusitis". Current Allergy and Asthma Reports. 13 (2): 229–35. doi:10.1007/s11882-013-0339-y. PMID 23354530.
- ↑ Blocked Nose in Babies ('Snuffles') Archived 29 August 2021 at the Wayback Machine at Patient UK
- ↑ Homemade Saline Nose Drops Archived 2012-11-27 at the Wayback Machine at Food.com
- ↑ "CDC - Naegleria - Frequently Asked Questions (FAQs)". Archived from the original on 20 March 2012. Retrieved 9 April 2012.
- ↑ Strandvik GF (2009). "Hypertonic saline in critical care: a review of the literature and guidelines for use in hypotensive states and raised intracranial pressure". Anaesthesia. 64 (9): 990–1003. doi:10.1111/j.1365-2044.2009.05986.x. PMID 19686485.
- ↑ "Hypertonic Saline • LITFL • CCC Pharmacology". Life in the Fast Lane • LITFL. 5 January 2019. Archived from the original on 16 April 2021. Retrieved 11 March 2021.
- ↑ 20.0 20.1 Mason, A; Malik, A; Ginglen, JG (January 2021). "Hypertonic Fluids". PMID 31194351.
{{cite journal}}: Cite journal requires|journal=(help) - ↑ "Use of Hypertonic Saline Injection in Trauma". Medscape. Archived from the original on 18 July 2017. Retrieved 11 March 2021.
- ↑ "Guideline to make up hypertonic saline (3% NaCl)" (PDF). Archived (PDF) from the original on 19 August 2019. Retrieved 11 March 2021.
- ↑ Wark, P; McDonald, VM (27 September 2018). "Nebulised hypertonic saline for cystic fibrosis". The Cochrane database of systematic reviews. 9: CD001506. doi:10.1002/14651858.CD001506.pub4. PMID 30260472.
- ↑ Prough, DS; Bidani, A (1999). "Hyperchloremic metabolic acidosis is a predictable consequence of intraoperative infusion of 0.9% saline". Anesthesiology. 90 (5): 1247–1249. doi:10.1097/00000542-199905000-00003. PMID 10319767.
- ↑ Fluid Density Calculator Archived 2009-09-16 at the Wayback Machine. Earthwardconsulting.com. Retrieved on 2011-02-27.
- ↑ Water Density Calculator Archived 2010-01-22 at the Wayback Machine. Csgnetwork.com. Retrieved on 2011-02-27.
- ↑ Lote, Christopher J. Principles of Renal Physiology, 5th edition. Springer. p. 6.
- ↑ Hamer, Walter J.; Wu, Yung‐Chi (1 October 1972). "Osmotic Coefficients and Mean Activity Coefficients of Uni‐univalent Electrolytes in Water at 25°C". Journal of Physical and Chemical Reference Data. 1 (4): 1047–1100. doi:10.1063/1.3253108.
- ↑ Vivian McAlister, Karen E. A. Burns, Tammy Znajda, and Brian Church. "Hypertonic Saline for Peri-operative Fluid Management" Cochrane Database of Systematic Reviews.1 (2010): CD005576 Available at: "Archived copy". Archived from the original on 6 July 2011. Retrieved 6 February 2011.
{{cite web}}: CS1 maint: archived copy as title (link) - ↑ Access Archived 2010-11-19 at the Wayback Machine. Medscape. Retrieved on 2011-02-27.
- ↑ MacGillivray, Neil (1 August 2017). "Sir William Brooke O'Shaughnessy (1808–1889), MD, FRS, LRCS Ed: Chemical pathologist, pharmacologist and pioneer in electric telegraphy". Journal of Medical Biography. 25 (3): 186–196. doi:10.1177/0967772015596276. ISSN 0967-7720. PMID 26385975. Archived from the original on 29 August 2021. Retrieved 9 April 2021.
- ↑ MacGillivray, Neil (March 2006). "Dr Latta of Leith: pioneer in the treatment of cholera by intravenous saline infusion". The Journal of the Royal College of Physicians of Edinburgh. 36 (1): 80–85. ISSN 1478-2715. PMID 17146955. Archived from the original on 29 August 2021. Retrieved 9 April 2021.
- ↑ Janakan, Gnananandan; Ellis, Harold (May 2013). "Dr Thomas Aitchison Latta (c1796-1833): pioneer of intravenous fluid replacement in the treatment of cholera". Journal of Medical Biography. 21 (2): 70–74. doi:10.1258/jmb.2012.012004. ISSN 1758-1087. PMID 24585745. Archived from the original on 29 August 2021. Retrieved 9 April 2021.
- ↑ Kenneth M Sutin; Marino, Paul L. (2007). "The ICU book" Archived 2017-09-18 at the Wayback Machine. Hagerstwon, MD: Lippincott Williams & Wilkins. ISBN 0-7817-4802-X.
- ↑ Awad, Sherif; Allison Simon P; Lobo Dileep N (2008). "The history of 0.9% saline". Clinical Nutrition. 27 (2): 179–88. doi:10.1016/j.clnu.2008.01.008. PMID 18313809.
- ↑ "Why Did Sterile Salt Water Become The IV Fluid Of Choice?". NPR.org. Archived from the original on 13 May 2018. Retrieved 13 May 2018.
- ↑ 37.0 37.1 Campbell-Falck, D; Thomas, T; Falck, TM; Tutuo, N; Clem, K (January 2000). "The intravenous use of coconut water". The American Journal of Emergency Medicine. 18 (1): 108–11. doi:10.1016/s0735-6757(00)90062-7. PMID 10674546.
- ↑ Hitchings, Andrew; Lonsdale, Dagan; Burrage, Daniel; Baker, Emma (2019). The Top 100 Drugs: Clinical Pharmacology and Practical Prescribing (2nd ed.). Elsevier. pp. 234–235. ISBN 978-0-7020-7442-4. Archived from the original on 22 May 2021. Retrieved 9 November 2021.
External links
| Identifiers: |
|
|---|
- Pages using duplicate arguments in template calls
- Webarchive template wayback links
- CS1 errors: missing periodical
- CS1 maint: archived copy as title
- Use dmy dates from December 2019
- Articles with invalid date parameter in template
- Chemical articles with unknown parameter in Infobox drug
- Infobox drug articles with non-default infobox title
- Chemical articles without CAS registry number
- Articles without EBI source
- Chemical pages without ChemSpiderID
- Chemical pages without DrugBank identifier
- Articles without KEGG source
- Articles without UNII source
- Drugs missing an ATC code
- Drugs with no legal status
- Articles containing unverified chemical infoboxes
- Articles with hatnote templates targeting a nonexistent page
- 1831 introductions
- Intravenous fluids
- World Health Organization essential medicines
- RTT