Rivaroxaban
 | |
 | |
| Names | |
|---|---|
| Trade names | Xarelto, others |
| Other names | BAY 59-7939 |
| |
| Clinical data | |
| Pregnancy category |
|
| Routes of use | By mouth |
| Defined daily dose | 20 mg[1] |
| External links | |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a611049 |
| Legal | |
| License data |
|
| Legal status | |
| Pharmacokinetics | |
| Bioavailability | 80–100%; Cmax = 2–4 hours (10 mg oral)[2] |
| Metabolism | CYP3A4, CYP2J2 and CYP-independent mechanisms[2] |
| Elimination half-life | 5–9 hours in healthy subjects aged 20 to 45[2][3] |
| Excretion | 2/3 metabolized in liver and 1/3 eliminated unchanged[2] |
| Chemical and physical data | |
| Formula | C19H18ClN3O5S |
| Molar mass | 435.88 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Rivaroxaban, sold under the brand name Xarelto among others, is an anticoagulant medication (blood thinner) used to treat and prevent blood clots.[4] Specifically it is used to treat deep vein thrombosis and pulmonary emboli and prevent blood clots in atrial fibrillation and following hip or knee surgery.[4] It is taken by mouth.[4]
Common side effects include bleeding.[4] Other serious side effects may include spinal hematoma and anaphylaxis.[4] It is unclear if use in pregnancy and breastfeeding is safe.[5] Compared to warfarin it has fewer interactions with other medications.[6] It works by blocking the activity of the clotting protein factor Xa.[4]
Rivaroxaban was patented in 2007 and approved for medical use in the United States in 2011.[7] It is on the World Health Organization's List of Essential Medicines as an alternative to dabigatran.[8] In the United States, it will not be available as a generic medication until 2024.[9][10] A month supply in the United Kingdom costs the NHS about £50 as of 2020.[11] In the United States, the wholesale cost of this amount is about US$430.[12] In 2017, it was the 109th most commonly prescribed medication in the United States, with more than six million prescriptions.[13][14]
Medical uses
Rivaroxaban is used to treat venous thromboembolism (VTE); deep vein thrombosis and pulmonary embolism.[15][16] In July 2012, the UK's NICE added its use in prevention of VTE.[17][18] It can be used to prevent VTE in people having hip or knee replacement surgery.[15]
It can be used to prevent stroke and VTE in people with non-valvular atrial fibrillation (AF), who have had a stroke before or who also have heart failure, high blood pressure or diabetes in addition to the AF.[15] In those with non-valvular AF, it appears to be as effective as warfarin in preventing ischemic strokes and embolic events.[19]
Rivaroxaban can be considered as an alternative to warfarin, particularly if a person is on several other medications that interact with warfarin, or if attending medical appointments and laboratory monitoring becomes difficult.[6]
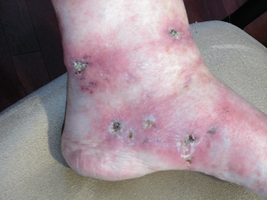
-
Livedoid vasculopathy presenting with ulcerations on the left lower extremity (pre-treatment).
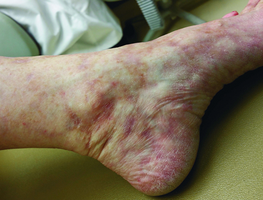
-
Well-healed ulcers after 8 months of rivaroxaban.
Dosage
The defined daily dose is 20 mg by mouth.[1]
The dose and duration of rivaroxaban varies according to the condition it is being used for.[15]
| Use | Dose | Comments |
|---|---|---|
| Treatment of VTE | 15mg/day BID for 21 days, then 20mg/day | [11][16] |
| Prevent recurrent VTE | 10mg/day after 6 months of full treatment | 20mg/day in those of higher risk such as VTE while taking 10mg/day rivaroxaban.[11][16] |
| Prevent VTE after knee replacement | 10mg/day for 2 weeks starting 6-10 hours after surgery | [11][16] |
| Prevent VTE after hip replacement | 10mg/day for 5 weeks starting 6-10 hours after surgery | [11][16] |
| Non-valvular AF | 20mg/day | If one of the following: heart failure, hypertension, previous stroke, TIA, age ≥ 75, diabetes.[11][16] |
| Following acute coronary syndrome | 2.5 mg twice daily for 12 months | With raised cardiac biomarkers (with aspirin alone or aspirin and clopidogrel).[11][16] |
| Coronary or peripheral artery disease | 2.5 mg twice daily | At high risk of ischaemic events (with aspirin).[11][16] |
Contraindications
It is best to avoid using rivaroxaban in people who cannot commit to taking their medication regularly without missing a dose.[6] Rivaroxaban is also not recommended when there is active bleeding or in those who have a high risk of bleeding such peptic ulcer disease or cancer.[15] Because of the difficulty associated with managing bleeding, rivaroxaban should be discontinued at least 24 hours before surgery, then restarted as soon as adequate hemostasis is established.[20]
Current dosing recommendations do not recommended administering rivaroxaban with drugs known to be strong combined CYP3A4/P-glycoprotein inhibitors because this results in significantly higher plasma concentrations of rivaroxaban.[21][22] Rivaroxaban is excreted in urine and faeces, with the result that for people with liver disease and kidney disease, a lower dose or an alternative may be more appropriate.[15]
Following a trial showing an increased chance of thrombotic events associated with rivaroxaban compared to warfarin, in people with Antiphospholipid syndrome with a history of previous thrombosis, Rivaroxaban is not recommended in people with antiphospholipd syndrome, especially if they have lupus anticoagulant, anticardiolipin antibodies, and anti-beta2 glycoprotein I antibodies.[11]
It is unclear if use in pregnancy and breastfeeding is safe.[5] As a result, it is not recommended for use in this group of people.[15]
Side effects
Side effects include bleeding, the most common being nose bleeds, bloody urine and gastrointestinal bleeding,[15] but there can be severe internal bleeding.[23][24][25] Rivaroxaban is associated with lower rates of serious and fatal bleeding events than warfarin but is associated with higher rates of bleeding in the gastrointestinal tract.[15] Other side effects include dizziness and anemia.[15]
As of 2015[update], post-marketing assessments showed liver toxicity, and further studies are needed to quantify this risk.[26][27]
Rivaroxaban has a boxed warning to make clear that people using the medication should not discontinue it before talking with their health care professional, because premature discontinuation can increase the risk of stroke.[21]
In 2015, rivaroxaban accounted for the highest number of reported cases of serious injury among regularly monitored medications to the FDA's Adverse Events Reporting System (AERS).[28]
Overdose
Taking more than the prescribed amount of rivaroxaban, or crushing, chewing or mixing it with other drugs or alcohol increases the risk of overdose, which can be fatal.[29]
Reversal agent
In October 2014, Portola Pharmaceuticals completed Phase I and II clinical trials for andexanet alfa as an antidote for Factor Xa inhibitors with few adverse effects, and started Phase III trials.[30][31] Andexanet alfa was expected to be approved in 2016.[32] Andexanet alfa was approved by the U.S. Food and Drug Administration in May, 2018, under the trade name AndexXa.[33][34] It is also available in the UK.[35]
Mechanism of action
Rivaroxaban works by directly blocking Factor Xa rather than inhibiting thrombin.[17][36] It might also block Factor Xa that is bound to clot.[36] It is a highly selective direct Factor Xa inhibitor with a rapid onset of action and peak blocking activity of between 1-4 hours after being given.[36] Inhibition of Factor Xa interrupts the intrinsic and extrinsic pathway of the blood coagulation cascade, inhibiting both thrombin formation and development of thrombi. Rivaroxaban does not inhibit thrombin (activated Factor II), and no effects on platelets have been demonstrated.[2] It allows predictable anticoagulation and dose adjustments and routine coagulation monitoring;[2] dietary restrictions are not needed.[32]
Unfractionated heparin (UFH), low molecular weight heparin (LMWH), and fondaparinux also inhibit the activity of factor Xa, indirectly, by binding to circulating antithrombin (AT III) and must be injected, whereas the orally active warfarin, phenprocoumon, and acenocoumarol are vitamin K antagonists (VKA), decreasing a number of coagulation factors, including Factor X.[37]
Rivaroxaban has predictable pharmacokinetics across a wide spectrum of patients (age, gender, weight, race) and has a flat dose response across an eightfold dose range (5–40 mg).[38] The oral bioavailability decreases with higher doses and increases when taken with food.[21][39]
Since it works quickly, initial heparin usage is not required.[15]
Chemistry
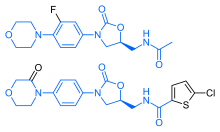
Rivaroxaban bears a striking structural similarity to the antibiotic linezolid: both drugs share the same oxazolidinone-derived core structure. Accordingly, rivaroxaban was studied for any possible antimicrobial effects and for the possibility of mitochondrial toxicity, which is a known complication of long-term linezolid use. Studies found that neither rivaroxaban nor its metabolites have any antibiotic effect against Gram-positive bacteria. As for mitochondrial toxicity, in vitro studies published before 2008 found the risk to be low.[40]
History
Rivaroxaban was initially developed by Bayer.[3] In the United States, it is marketed by Janssen Pharmaceutica (a part of Johnson & Johnson).[41] It is the first available active direct factor Xa inhibitor which is taken by mouth.[42]
Society and culture
Cost
As of 2020, a month supply of rivaroxaban costs the NHS about £50 in the United Kingdom,[11] where its cost reaches more than 50 times that of warfarin, although this difference may be offset by lower monitoring costs.[15] In the United States, the wholesale cost of this amount is about US$430.[12] In 2017, it was the 109th most commonly prescribed medication in the United States, with more than six million prescriptions.[13][14]
-
Rivaroxaban costs (US)
-
Rivaroxaban prescriptions (US)
Economics
Using rivaroxaban rather than warfarin costs 70 times more, according to Express Scripts Holding Co, the largest U.S. pharmacy benefits manager.[32] As of 2016, Bayer claimed that the drug was licensed in 130 countries and that more than 23 million people had been treated.[43]
Approval
In September 2008, Health Canada granted marketing authorization for rivaroxaban to prevent venous thromboembolism (VTE) in people who have undergone elective total hip replacement or total knee replacement surgery.[44]
In the same month, the European Commission also granted marketing authorization of rivaroxaban to prevent venous thromboembolism in adults undergoing elective hip and knee replacement.[45]
On July 1, 2011, the United States Food and Drug Administration (US FDA) approved rivaroxaban for prophylaxis of deep vein thrombosis (DVT), which may lead to pulmonary embolism (PE), in adults undergoing hip and knee replacement surgery.[46]
On November 4, 2011, the US FDA approved rivaroxaban for stroke prevention in people with non-valvular atrial fibrillation.[47]
Legal action
On March 25, 2019, over 25,000 lawsuits over rivaroxaban in the US were settled for $775 million to get paid out to those affected. Plaintiffs accused the drugmakers of not warning about the bleeding risks, claiming their injuries could have been prevented had doctors and patients been provided adequate information.[48]
Available forms
Rivaroxaban is available under the brand name Xeralto, in tablets of 2.5, 10, 15 and 20mg.[11]
Research
Researchers at the Duke Clinical Research Institute have been accused of withholding clinical data used to evaluate rivaroxaban.[49] Duke tested rivaroxaban in a clinical trial known as the ROCKET AF trial.[50] The clinical trial, published in 2011 in the New England Journal of Medicine[51] and headed by Robert Califf, then Commissioner of the FDA,[52][51] found rivaroxaban to be more effective than warfarin in reducing the likelihood of ischemic strokes in patients with atrial fibrillation.[51] The validity of the study was called into question in 2014 when pharmaceutical sponsors Bayer and Johnson & Johnson revealed that the INR blood monitoring devices used were not functioning properly,[49][50] A subsequent analysis by the Duke team published in February 2016 found that this had no significant effect on efficacy and safety in the trial.[53]
Under-representation of racial minorities in clinical trials has been noted. Compared to warfarin, efficacy and safety was found to be similar across racial subgroups.[51]
References
- ↑ 1.0 1.1 "WHOCC - ATC/DDD Index". www.whocc.no. Archived from the original on 18 August 2020. Retrieved 6 September 2020.
- ↑ 2.0 2.1 2.2 2.3 2.4 2.5 "Xarelto: Summary of Product Characteristics". Bayer Schering Pharma AG. 2008. Archived from the original on 2010-07-27. Retrieved 2009-02-11.
- ↑ 3.0 3.1 Turgeon, Mary Lou (2018). "28. Disorders of haemostats and thrombosis: blood coagulation factors, hyper coagulable state, and anticoagulant therapy". Clinical Hematology: Theory & Procedures, Enhanced Edition (6th ed.). Burlington: Jones & Bartlett Learning. pp. 575–576. ISBN 978-1-284-29449-1. Archived from the original on 2021-08-28. Retrieved 2021-03-20.
- ↑ 4.0 4.1 4.2 4.3 4.4 4.5 "Rivaroxaban Monograph for Professionals". Drugs.com. American Society of Health-System Pharmacists. Archived from the original on 27 March 2019. Retrieved 3 March 2019.
- ↑ 5.0 5.1 "Rivaroxaban Use During Pregnancy". Drugs.com. Archived from the original on 27 March 2019. Retrieved 3 March 2019.
- ↑ 6.0 6.1 6.2 Kiser, Kathryn (2017). Oral Anticoagulation Therapy: Cases and Clinical Correlation. Springer. p. 11. ISBN 9783319546438. Archived from the original on 2019-03-27. Retrieved 2019-03-27.
- ↑ "Generic Xarelto Availability". Drugs.com. Archived from the original on 16 June 2018. Retrieved 9 May 2017.
- ↑ World Health Organization (2023). The selection and use of essential medicines 2023: web annex A: World Health Organization model list of essential medicines: 23rd list (2023). Geneva: World Health Organization. hdl:10665/371090. WHO/MHP/HPS/EML/2023.02.
- ↑ "Orange Book: Approved Drug Products with Therapeutic Equivalence Evaluations". www.accessdata.fda.gov. Archived from the original on 2020-09-13. Retrieved 2019-04-24.
- ↑ "Bayer, J&J Win Ruling That Upholds Patent for Xarelto Drug". 2019-04-22. Archived from the original on 2019-08-01. Retrieved 2019-04-24.
- ↑ 11.00 11.01 11.02 11.03 11.04 11.05 11.06 11.07 11.08 11.09 11.10 BNF (80 ed.). London: BMJ Group and the Pharmaceutical Press. September 2020 – March 2021. pp. 137–139. ISBN 978-0-85711-369-6.
{{cite book}}: CS1 maint: date format (link) - ↑ 12.0 12.1 "NADAC as of 2019-02-27". Centers for Medicare and Medicaid Services. Archived from the original on 2019-03-06. Retrieved 3 March 2019.
- ↑ 13.0 13.1 "The Top 300 of 2020". ClinCalc. Archived from the original on 18 March 2020. Retrieved 11 April 2020.
- ↑ 14.0 14.1 "Rivaroxaban - Drug Usage Statistics". ClinCalc. Archived from the original on 8 July 2020. Retrieved 11 April 2020.
- ↑ 15.00 15.01 15.02 15.03 15.04 15.05 15.06 15.07 15.08 15.09 15.10 15.11 Hitchings, Andrew; Lonsdale, Dagan; Burrage, Daniel; Baker, Emma (2019). The Top 100 Drugs: Clinical Pharmacology and Practical Prescribing (2nd ed.). Elsevier. pp. 118–119. ISBN 978-0-7020-7442-4. Archived from the original on 2021-05-22. Retrieved 2021-11-09.
- ↑ 16.0 16.1 16.2 16.3 16.4 16.5 16.6 16.7 "Xarelto 20mg film-coated tablets - Summary of Product Characteristics (SmPC) - (emc)". www.medicines.org.uk. Archived from the original on 26 March 2021. Retrieved 26 March 2021.
- ↑ 17.0 17.1 Ritter, James M.; Flower, Rod; Henderson, Graeme; Loke, Yoon Kong; Rang, Humphrey P. (2020). "25. Haemostasis and thrombosis". Rang & Dale's Pharmacology. Elsevier. p. 325-327. ISBN 978-0-7020-7448-6. Archived from the original on 2021-08-28. Retrieved 2021-03-24.
- ↑ "Overview | Rivaroxaban for the treatment of deep vein thrombosis and prevention of recurrent deep vein thrombosis and pulmonary embolism | Guidance | NICE". www.nice.org.uk. Archived from the original on 13 May 2020. Retrieved 1 January 2020.
- ↑ Gómez-Outes A, Terleira-Fernández AI, Calvo-Rojas G, Suárez-Gea ML, Vargas-Castrillón E (2013). "Dabigatran, Rivaroxaban, or Apixaban versus Warfarin in Patients with Nonvalvular Atrial Fibrillation: A Systematic Review and Meta-Analysis of Subgroups". Thrombosis. 2013: 640723. doi:10.1155/2013/640723. PMC 3885278. PMID 24455237.
- ↑ Sunkara T, Ofori E, Zarubin V, Caughey ME, Gaduputi V, Reddy M (2016). "Perioperative Management of Direct Oral Anticoagulants (DOACs): A Systemic Review". Health Services Insights. 9 (Suppl 1): 25–36. doi:10.4137/HSI.S40701. PMC 5156547. PMID 28008269.
- ↑ 21.0 21.1 21.2 "Xarelto (rivaroxaban) label" (PDF). U.S. Food and Drug Association. Archived (PDF) from the original on 2018-09-20. Retrieved 2016-04-27.
- ↑ Mueck W, Kubitza D, Becka M (September 2013). "Co-administration of rivaroxaban with drugs that share its elimination pathways: pharmacokinetic effects in healthy subjects". British Journal of Clinical Pharmacology. 76 (3): 455–66. doi:10.1111/bcp.12075. PMC 3769672. PMID 23305158.
- ↑ "Medication Guide – Xarelto" (PDF). U.S. Food and Drug Administration. Archived (PDF) from the original on 2 May 2014. Retrieved 1 September 2014.
- ↑ "Xarelto Side Effects". WebMD. Archived from the original on 3 September 2014. Retrieved 1 September 2014.
- ↑ "Xarelto Side Effects Center". RxList. Archived from the original on 3 September 2014. Retrieved 1 September 2014.
- ↑ Raschi E, Poluzzi E, Koci A, Salvo F, Pariente A, Biselli M, Moretti U, Moore N, De Ponti F (August 2015). "Liver injury with novel oral anticoagulants: assessing post-marketing reports in the US Food and Drug Administration adverse event reporting system". British Journal of Clinical Pharmacology. 80 (2): 285–93. doi:10.1111/bcp.12611. PMC 4541976. PMID 25689417.
- ↑ Russmann S, Niedrig DF, Budmiger M, Schmidt C, Stieger B, Hürlimann S, Kullak-Ublick GA (August 2014). "Rivaroxaban postmarketing risk of liver injury" (PDF). Journal of Hepatology. 61 (2): 293–300. doi:10.1016/j.jhep.2014.03.026. PMID 24681117. Archived (PDF) from the original on 2018-12-11. Retrieved 2019-07-17.
- ↑ Schroeder, C. "ISMP Ranks Xarelto Most Dangerous Drug in the United States". DrugNews. DrugNews. Archived from the original on 11 August 2016. Retrieved 10 August 2016.
- ↑ "What is Rivaroxaban & How is it Abused? | Xarelto Treatment". Oxford Treatment Center. Archived from the original on 22 March 2021. Retrieved 22 March 2021.
- ↑ Schroeder, C. "Possible Antidote Could Help Blood Thinner Patients In Bleeding Emergencies". DrugNews. Archived from the original on 10 May 2015. Retrieved 20 August 2015.
- ↑ Mo Y, Yam FK (February 2015). "Recent advances in the development of specific antidotes for target-specific oral anticoagulants". Pharmacotherapy. 35 (2): 198–207. doi:10.1002/phar.1532. PMID 25644580.
- ↑ 32.0 32.1 32.2 Bill Berkrot (December 23, 2015). "New blood thinner 'antidote' to help doctors move past warfarin". Reuters. Archived from the original on April 3, 2019. Retrieved July 2, 2017.
- ↑ "Accelerated Approval for AndexXa" (PDF). FDA. Archived (PDF) from the original on 26 July 2018. Retrieved 1 August 2018.
- ↑ Inc., Portola Pharmaceuticals. "U.S. FDA Approves Portola Pharmaceuticals' Andexxa®, First and Only Antidote for the Reversal of Factor Xa Inhibitors". GlobeNewswire News Room. Archived from the original on 10 April 2021. Retrieved 1 August 2018.
- ↑ BNF (80 ed.). London: BMJ Group and the Pharmaceutical Press. September 2020 – March 2021. p. 128. ISBN 978-0-85711-369-6.
{{cite book}}: CS1 maint: date format (link) - ↑ 36.0 36.1 36.2 Dajas-Bailador, Federico (2019). Clinical Pharmacology for Prescribing. Oxford University Press. p. 138. ISBN 978-0-19-969493-8. Archived from the original on 2021-08-29. Retrieved 2021-03-24.
- ↑ Turpie AG (January 2008). "New oral anticoagulants in atrial fibrillation". European Heart Journal. 29 (2): 155–65. doi:10.1093/eurheartj/ehm575. PMID 18096568.
- ↑ Eriksson BI, Borris LC, Dahl OE, Haas S, Huisman MV, Kakkar AK, Muehlhofer E, Dierig C, Misselwitz F, Kälebo P (November 2006). "A once-daily, oral, direct Factor Xa inhibitor, rivaroxaban (BAY 59-7939), for thromboprophylaxis after total hip replacement". Circulation. 114 (22): 2374–81. doi:10.1161/CIRCULATIONAHA.106.642074. PMID 17116766.
- ↑ "Rivaroxaban: a blood-thinning medicine to treat and prevent blood clots". nhs.uk. 20 December 2018. Archived from the original on 22 March 2021. Retrieved 22 March 2021.
- ↑ European Medicines Agency (2008). "CHP Assessment Report for Xarelto (EMEA/543519/2008)" (PDF). Retrieved 2009-06-11.[permanent dead link]
- ↑ Staines, Richard (15 October 2019). "FDA approves Bayer/Janssen's Xarelto in new blood clot prevention use -". pharmaphorum.com. Archived from the original on 20 March 2021. Retrieved 20 March 2021.
- ↑ Abdulsattar Y, Bhambri R, Nogid A (May 2009). "Rivaroxaban (xarelto) for the prevention of thromboembolic disease: an inside look at the oral direct factor xa inhibitor". P & T. 34 (5): 238–44. PMC 2697099. PMID 19561868.
- ↑ "Bayer comments on article in The British Medical Journal (BMJ) regarding Xarelto" (PDF). Bayer AG Communications, Government Relations & Corporate Brand. 29 September 2016. Archived (PDF) from the original on 31 January 2017. Retrieved 19 January 2017.
- ↑ "Bayer's Xarelto Approved in Canada" (Press release). Bayer. 2008-09-16. Archived from the original on 2011-07-11. Retrieved 2010-01-31.
- ↑ "Bayer's Novel Anticoagulant Xarelto now also approved in the EU" (Press release). Bayer. 2008-02-10. Archived from the original on 2008-10-22. Retrieved 2010-01-31.
- ↑ "FDA Approves Xarelto® (rivaroxaban tablets) to Help Prevent Deep Vein Thrombosis in Patients Undergoing Knee or Hip Replacement Surgery" (Press release). Janssen Pharmaceutica. 2011-07-01. Archived from the original on 2011-11-05. Retrieved 2011-07-01.
- ↑ "FDA approves Xarelto to prevent stroke in people with common type of abnormal heart rhythm". US Food and Drug Association. 4 November 2011. Archived from the original on 5 November 2011. Retrieved 27 April 2016.
- ↑ "Bayer, Johnson & Johnson settle more than 25,000 lawsuits over blood thinner Xarelto for $775 million". washingtonpost.com. Archived from the original on 26 March 2019. Retrieved 7 April 2019.
- ↑ 49.0 49.1 Thomas, Katie (1 March 2016). "Document Claims Drug Makers Deceived a Top Medical Journal". The New York Times. Archived from the original on 5 March 2016. Retrieved 3 May 2016.
- ↑ 50.0 50.1 Patel, Vir. "Duke clinical trial under scrutiny in drug case". The Chronicle. Duke Student Publishing Company. Archived from the original on 2016-05-08. Retrieved 2016-05-03.
- ↑ 51.0 51.1 51.2 51.3 Patel MR, Mahaffey KW, Garg J, Pan G, Singer DE, Hacke W, Breithardt G, Halperin JL, Hankey GJ, Piccini JP, Becker RC, Nessel CC, Paolini JF, Berkowitz SD, Fox KA, Califf RM (September 2011). "Rivaroxaban versus warfarin in nonvalvular atrial fibrillation". The New England Journal of Medicine. 365 (10): 883–91. doi:10.1056/NEJMoa1009638. PMC 3860773. PMID 21830957.
- ↑ "Meet Robert M. Califf, M.D., Commissioner of Food and Drugs". U.S. Food and Drug Administration. U.S. Food and Drug Administration. Archived from the original on 15 May 2016. Retrieved 3 May 2016.
- ↑ Patel MR, Hellkamp AS, Fox KA (February 2016). "Point-of-Care Warfarin Monitoring in the ROCKET AF Trial" (PDF). The New England Journal of Medicine. 374 (8): 785–8. doi:10.1056/NEJMc1515842. PMID 26839968. Archived (PDF) from the original on 2018-07-22. Retrieved 2019-07-17.
External links
| External sites: | |
|---|---|
| Identifiers: |
|
- Pages using duplicate arguments in template calls
- CS1 maint: date format
- All articles with dead external links
- Articles with dead external links from April 2018
- Articles with invalid date parameter in template
- Articles with permanently dead external links
- Chemical articles with unknown parameter in Infobox drug
- Chemical articles without CAS registry number
- Articles without EBI source
- Chemical pages without ChemSpiderID
- Chemical pages without DrugBank identifier
- Articles without KEGG source
- Articles without UNII source
- Drugs missing an ATC code
- Drug has EMA link
- Drugboxes which contain changes to verified fields
- Articles containing potentially dated statements from 2015
- All articles containing potentially dated statements
- Articles with changed CASNo identifier
- Articles with changed KEGG identifier
- RTT
- Direct Xa inhibitors
- Morpholines
- Thiophenes
- Chloroarenes
- 3-(4-methoxyphenyl)-2-oxazolidinones
- Johnson & Johnson brands
- Janssen Pharmaceutica
- Bayer brands
- World Health Organization essential medicines (alternatives)