Primary hyperparathyroidism
| Primary hyperparathyroidism | |
|---|---|
 | |
| Thyroid and parathyroid. | |
| Specialty | Endocrinology |
Primary hyperparathyroidism is usually caused by a tumor within the parathyroid gland. The symptoms of the condition relate to the elevated calcium levels, which can cause digestive symptoms, kidney stones, psychiatric abnormalities, and bone disease.
The diagnosis is initially made on blood tests; an elevated level of calcium together with a raised level of parathyroid hormone are typically found. To identify the source of the excessive hormone secretion, medical imaging may be performed. Parathyroidectomy, the surgical removal of one or more parathyroid glands, may be required to control symptoms.
Signs and symptoms
The signs and symptoms of primary hyperparathyroidism are those of hypercalcemia. They are classically summarized by "stones, bones, abdominal groans, thrones and psychiatric overtones".[citation needed]
- "Stones" refers to kidney stones, nephrocalcinosis, and diabetes insipidus (polyuria and polydipsia). These can ultimately lead to kidney failure.
- "Bones" refers to bone-related complications. The classic bone disease in hyperparathyroidism is osteitis fibrosa cystica, which results in pain and sometimes pathological fractures. Other bone diseases associated with hyperparathyroidism are osteoporosis, osteomalacia, and arthritis.
- "Abdominal groans" refers to gastrointestinal symptoms of constipation, indigestion, nausea and vomiting. Hypercalcemia can lead to peptic ulcers and acute pancreatitis. The peptic ulcers can be an effect of increased gastric acid secretion by hypercalcemia.[1]
- "Thrones" refers to polyuria and constipation
- "Psychiatric overtones" refers to effects on the central nervous system. Symptoms include lethargy, fatigue, depression, memory loss, psychosis, ataxia, delirium, and coma.
Left ventricular hypertrophy may also be seen.[2]
Other signs include proximal muscle weakness, itching, and band keratopathy of the eyes.[citation needed]
When subjected to formal research, symptoms of depression, pain, and gastric dysfunction seem to correlate with mild cases of hypercalcemia.[3]
Causes
The most common cause of primary hyperparathyroidism is a sporadic, single parathyroid adenoma[4] resulting from a clonal mutation (~97%). Less common are parathyroid hyperplasia[5] (~2.5%), parathyroid carcinoma (malignant tumor), and adenomas in more than one gland (together ~0.5%).Primary hyperparathyroidism is also a feature of several familial endocrine disorders: Multiple endocrine neoplasia type 1 and type 2A (MEN type 1 and MEN type 2A), and familial hyperparathyroidism.[citation needed]
Genetic associations include:
| OMIM | Name | Gene |
|---|---|---|
| 145000 | HRPT1 | MEN1, HRPT2 |
| 145001 | HRPT2 | HRPT2 |
| 610071 | HRPT3 | unknown at 2p13.3-14[6] |
In all cases, the disease is idiopathic, but is thought to involve inactivation of tumor suppressor genes (Menin gene in MEN1), or involve gain of function mutations (RET proto-oncogene MEN 2a).[citation needed]
Recently, it was demonstrated that liquidators of the Chernobyl power plant are faced with a substantial risk of primary hyperparathyroidism, possibly caused by radioactive strontium isotopes.[7]
Diagnosis
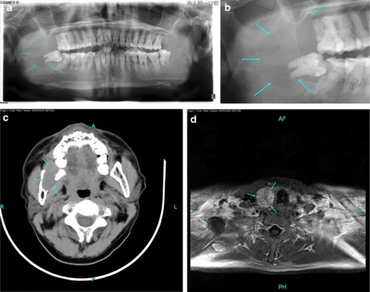
The diagnosis of primary hyperparathyroidism is made by blood tests.[citation needed]
Serum calcium levels are elevated, and the parathyroid hormone level is abnormally high compared with an expected low level in response to the high calcium. A relatively elevated parathyroid hormone has been estimated to have a sensitivity of 60–80% and a specificity of approximately 90% for primary hyperparathyroidism.[8]
A more powerful variant of comparing the balance between calcium and parathyroid hormone is to perform a 3-hour calcium infusion. After infusion, a parathyroid hormone level above a cutoff of 14 ng/l has a sensitivity of 100% and a specificity of 93% in detecting primary hyperparathyroidism, with a confidence interval of 80% to 100%.[9]
Urinary cAMP is occasionally measured; it is generally elevated due to activation of Gs proteins when PTH binds to its receptor.[10]
Biochemical confirmation of primary hyperparathyroidism is following by investigations to localize the culprit lesion. Primary hyperparathyroidism is most commonly due to solitary parathyroid adenoma. Less commonly it may be due to double parathyroid adenomas or parathyroid hyperplasia. Tc99 sestamibi scan of head, neck and upper thorax is the most commonly used test for localizing parathyroid adenomas having a sensitivity and specificity of 70–80%. Sensitivity falls down to 30% in case of double/multiple parathyroid adenomas or in case of parathyroid hyperplasia. Ultrasonography is also a useful test in localizing suspicious parathyroid lesions.[citation needed]
Treatment
Treatment is usually surgical removal of the gland(s) containing adenomas, but medication may also be required.[citation needed]
Surgery
The surgical removal of one or more of the parathyroid glands is known as a parathyroidectomy; this operation was first performed in 1925.[11] The symptoms of the disease, listed above, are indications for surgery. Surgery reduces all cause mortality as well as resolving symptoms. However, cardiovascular mortality is not significantly reduced.[12]
The 2002 NIH Workshop on Asymptomatic Primary Hyperparathyroidism developed criteria for surgical intervention . The criteria were revised at the Third International Workshop on the Management of Asymptomatic Primary Hyperparathyroidism . These criteria were chosen on the basis of clinical experience and observational and clinical trial data as to which patients are more likely to have end-organ effects of primary hyperparathyroidism (nephrolithiasis, skeletal involvement), disease progression if surgery is deferred, and the most benefit from surgery. The panel emphasized the need for parathyroidectomy to be performed by surgeons who are highly experienced and skilled in the operation. The Third International Workshop guidelines concluded that surgery is indicated in asymptomatic patients who meet any one of the following conditions:[citation needed]
- Serum calcium concentration of 1.0 mg/dL (0.25 mmol/L) or more above the upper limit of normal
- Creatinine clearance that is reduced to <60 mL/min
- Bone density at the hip, lumbar spine, or distal radius that is more than 2.5 standard deviations below peak bone mass (T score <-2.5) and/or previous fragility fracture
- Age less than 50 years
Operative intervention can be delayed in patients over 50 years of age who are asymptomatic or minimally symptomatic and who have serum calcium concentrations <1.0 mg/dL (0.2 mmol/L) above the upper limit of normal, and in patients who are medically unfit for surgery[citation needed]
More recently, three randomized controlled trials have studied the role of surgery in patients with asymptomatic hyperparathyroidism. The largest study reported that surgery resulted in an increase in bone mass, but no improvement in quality of life after one to two years among patients in the following groups:[13]
- Untreated, asymptomatic primary hyperparathyroidism
- Serum calcium between 2.60–2.85 mmol/liter (10.4–11.4 mg/dl)
- Age between 50 and 80 yr
- No medications interfering with Ca metabolism
- No hyperparathyroid bone disease
- No previous operation in the neck
- Creatinine level < 130 μmol/liter (<1.47 mg/dl)
Two other trials reported improvements in bone density and some improvement in quality of life with surgery.[14][15]
Medications
Medications that are sometimes required include estrogen replacement therapy in postmenopausal women and bisphosphonates. Bisphosphonates may improve bone turnover.[16]Newer medications termed "calcimimetics" used in secondary hyperparathyroidism are now being used in primary hyperparathyroidism. Calcimimetics reduce the amount of parathyroid hormone released by the parathyroid glands. They are recommended in patients in whom surgery is inappropriate.[17]
Epidemiology
Primary hyperparathyroidism affects approximately 1 per 1,000 people (0.1%),[18] while there are 25–30 new cases per 100,000 people per year in the United States.[19] The prevalence of primary hyperparathyroidism has been estimated to be 3 in 1000 in the general population and as high as 21 in 1000 in postmenopausal women.[20] It is almost exactly three times as common in women as men.[citation needed]
Primary hyperparathyroidism is associated with increased all-cause mortality.[12]
Children
In contrast with primary hyperparathyroidism in adults, primary hyperparathyroidism in children is considered a rare endocrinopathy. Pediatric primary hyperparathyroidism can be distinguished by its more severe manifestations, in contrast to the less intense manifestations in adult primary hyperparathyroidism. Multiple endocrine neoplasia is more likely to be associated with childhood and adolescent primary hyperparathyroidism. The fundamental skeletal radiologic manifestation include diffuse osteopenia, pathologic fractures and the coexistence of resorption and sclerosis at numerous sites. Skeletal lesions can be specifically bilateral, symmetric and multifocal, exhibiting different types of bone resorption. Pathologic fractures of the femoral neck and spine can potentially initiate serious complications. Because pediatric primary hyperparathyroidism is frequently associated with pathologic fractures it can be misdiagnosed as osteogenesis imperfecta. Pediatric patients with primary hyperparathyroidism are best remedied by parathyroidectomy. Early diagnosis of pediatric primary hyperparathyroidism is all-important to minimize disease complications and start off timely and relevant treatment.[21][22]
Research directions
Future developments such as calcimimetic agents (e.g. cinacalcet) which activate the parathyroid calcium-sensing receptor may offer a good alternative to surgery.[citation needed]
See also
References
- ↑ Barreras, R. F.; Donaldson, R. M. (1967). "Role of Calcium in Gastric Hypersecretion, Parathyroid Adenoma and Peptic Ulcer". New England Journal of Medicine. 276 (20): 1122–1124. doi:10.1056/NEJM196705182762005. PMID 6024167.
- ↑ Stefenelli T, Abela C, Frank H, et al. (1997). "Cardiac abnormalities in patients with primary hyperparathyroidism: implications for follow-up". J. Clin. Endocrinol. Metab. 82 (1): 106–12. doi:10.1210/jc.82.1.106. PMID 8989242. Archived from the original on 2011-05-16. Retrieved 2021-12-31.
- ↑ Bargren AE, Repplinger D, Chen H, Sippel RS (2011). "Can biochemical abnormalities predict symptomatology in patients with primary hyperparathyroidism?". J Am Coll Surg. 213 (3): 410–4. doi:10.1016/j.jamcollsurg.2011.06.401. PMID 21723154.
- ↑ "Endocrine Pathology". Archived from the original on 2009-03-08. Retrieved 2009-05-08.
- ↑ "Endocrine Pathology". Archived from the original on 2009-03-07. Retrieved 2009-05-08.
- ↑ Warner JV, Nyholt DR, Busfield F, et al. (March 2006). "Familial isolated hyperparathyroidism is linked to a 1.7 Mb region on chromosome 2p13.3–14". J. Med. Genet. 43 (3): e12. doi:10.1136/jmg.2005.035766. PMC 2563254. PMID 16525030.
- ↑ Boehm BO, Rosinger S, Belyi D, Dietrich JW (2011). "The parathyroid as a target for radiation damage". N. Engl. J. Med. 365 (7): 676–8. doi:10.1056/NEJMc1104982. PMID 21848480.
- ↑ [1] Lepage, R.; d'Amour, P.; Boucher, A.; Hamel, L.; Demontigny, C.; Labelle, F. (1988). "Clinical performance of a parathyrin immunoassay with dynamically determined reference values". Clinical Chemistry. 34 (12): 2439–2443. doi:10.1093/clinchem/34.12.2439. PMID 3058363.
- ↑ Titon, I.; Cailleux-Bounacer, A.; Basuyau, J. P.; Lefebvre, H.; Savoure, A.; Kuhn, J. M. (2007). "Evaluation of a standardized short-time calcium suppression test in healthy subjects: Interest for the diagnosis of primary hyperparathyroidism". European Journal of Endocrinology. 157 (3): 351–357. doi:10.1530/EJE-07-0132. PMID 17766719.
- ↑ Jameson, J. Larry, éditeur intellectuel. (2018-08-13). Harrison's principles of internal medicine. ISBN 9781259644030. OCLC 1056749127.
{{cite book}}: CS1 maint: multiple names: authors list (link) - ↑ Weber T, Eberle J, Messelhäuser U, et al. (2013). "Parathyroidectomy, elevated depression scores, and suicidal ideation in patients with primary hyperparathyroidism: results of a prospective multicenter study". JAMA Surg. 148 (2): 109–115. doi:10.1001/2013.jamasurg.316. PMID 23560281.
- ↑ 12.0 12.1 Vestergaard, P.; Mosekilde, L. (2003). "Cohort study on effects of parathyroid surgery on multiple outcomes in primary hyperparathyroidism". BMJ. 327 (7414): 530–534. doi:10.1136/bmj.327.7414.530. PMC 192894. PMID 12958111.
- ↑ Bollerslev J, Jansson S, Mollerup CL, et al. (2007). "Medical observation, compared with parathyroidectomy, for asymptomatic primary hyperparathyroidism: a prospective, randomized trial". J. Clin. Endocrinol. Metab. 92 (5): 1687–92. doi:10.1210/jc.2006-1836. PMID 17284629.
- ↑ Ambrogini E, Cetani F, Cianferotti L, et al. (2007). "Surgery or surveillance for mild asymptomatic primary hyperparathyroidism: a prospective, randomized clinical trial". J. Clin. Endocrinol. Metab. 92 (8): 3114–21. doi:10.1210/jc.2007-0219. PMID 17535997.
- ↑ Rao DS, Phillips ER, Divine GW, Talpos GB (2004). "Randomized controlled clinical trial of surgery versus no surgery in patients with mild asymptomatic primary hyperparathyroidism". J. Clin. Endocrinol. Metab. 89 (11): 5415–22. doi:10.1210/jc.2004-0028. PMID 15531491.
- ↑ Khan AA, Bilezikian JP, Kung AW, et al. (2004). "Alendronate in primary hyperparathyroidism: a double-blind, randomized, placebo-controlled trial". J. Clin. Endocrinol. Metab. 89 (7): 3319–25. doi:10.1210/jc.2003-030908. PMID 15240609.
- ↑ "Archive copy" (PDF). Archived (PDF) from the original on 2014-10-05. Retrieved 2021-12-31.
{{cite web}}: CS1 maint: archived copy as title (link) - ↑ Deshmukh, R. G.; Alsagoff, S. A. L.; Krishnan, S.; Dhillon, K. S.; Khir, A. S. M. (1998). "Primary hyperparathyroidism presenting with pathological fracture". Journal of the Royal College of Surgeons of Edinburgh. 43 (6): 424–427. PMID 9990797. Archived from the original on 2011-05-13. Retrieved 2011-06-18.
- ↑ Bilezikian, John P.; Silverberg, Shonni J. (2002). "Primary hyperparathyroidism: Epidemiology and clinical consequences". Clinical Reviews in Bone and Mineral Metabolism. 1 (1): 25–34. doi:10.1385/BMM:1:1:25. S2CID 74793206.
- ↑ Bolland, M. J.; Grey, A. B.; Gamble, G. D.; Reid, I. R. (2004). "Association between Primary Hyperparathyroidism and Increased Body Weight: A Meta-Analysis". Journal of Clinical Endocrinology & Metabolism. 90 (3): 1525–1530. doi:10.1210/jc.2004-1891. PMID 15613408.
- ↑ EL-Sobky TA, Ahmad KA, Samir S, EL Mikkawy DME (2016). "Primary hyperparathyroidism in a child: The musculoskeletal manifestations of a late presenting rare endocrinopathy". Egypt J Radiol Nucl Med. 47 (4): 1613–16. doi:10.1016/j.ejrnm.2016.09.002.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ↑ Mallet E (2008). "Primary hyperparathyroidism in neonates and childhood. The French experience (1984–2004)". Horm Res. 69 (3): 180–8. doi:10.1159/000112592. PMID 18219222. S2CID 25046706.
External links
| Classification | |
|---|---|
| External resources |
- Pages with script errors
- CS1 maint: multiple names: authors list
- CS1 maint: archived copy as title
- All articles with unsourced statements
- Articles with unsourced statements from October 2021
- Articles with invalid date parameter in template
- Articles with unsourced statements from August 2020
- Parathyroid disorders