Portal:Minerals
Portal maintenance status: (May 2019)
|
The Minerals Portal

In geology and mineralogy, a mineral or mineral species is, broadly speaking, a solid substance with a fairly well-defined chemical composition and a specific crystal structure that occurs naturally in pure form.
The geological definition of mineral normally excludes compounds that occur only in living organisms. However, some minerals are often biogenic (such as calcite) or organic compounds in the sense of chemistry (such as mellite). Moreover, living organisms often synthesize inorganic minerals (such as hydroxylapatite) that also occur in rocks.
The concept of mineral is distinct from rock, which is any bulk solid geologic material that is relatively homogeneous at a large enough scale. A rock may consist of one type of mineral or may be an aggregate of two or more different types of minerals, spacially segregated into distinct phases.
Some natural solid substances without a definite crystalline structure, such as opal or obsidian, are more properly called mineraloids. If a chemical compound occurs naturally with different crystal structures, each structure is considered a different mineral species. Thus, for example, quartz and stishovite are two different minerals consisting of the same compound, silicon dioxide. (Full article...)
Mineralogy is a subject of geology specializing in the scientific study of the chemistry, crystal structure, and physical (including optical) properties of minerals and mineralized artifacts. Specific studies within mineralogy include the processes of mineral origin and formation, classification of minerals, their geographical distribution, as well as their utilization. (Full article...)
Selected articles
-
Image 1

Chalcopyrite (/ˌkælkəˈpaɪˌraɪt, -koʊ-/ KAL-kə-PY-ryte, -koh-) is a copper iron sulfide mineral and the most abundant copper ore mineral. It has the chemical formula CuFeS2 and crystallizes in the tetragonal system. It has a brassy to golden yellow color and a hardness of 3.5 to 4 on the Mohs scale. Its streak is diagnostic as green-tinged black.
On exposure to air, chalcopyrite tarnishes to a variety of oxides, hydroxides, and sulfates. Associated copper minerals include the sulfides bornite (Cu5FeS4), chalcocite (Cu2S), covellite (CuS), digenite (Cu9S5); carbonates such as malachite and azurite, and rarely oxides such as cuprite (Cu2O). It is rarely found in association with native copper. Chalcopyrite is a conductor of electricity.
Copper can be extracted from chalcopyrite ore using various methods. The two predominant methods are pyrometallurgy and hydrometallurgy, the former being the most commercially viable. (Full article...) -
Image 2A lustrous crystal of zircon perched on a tan matrix of calcite from the Gilgit District of Pakistan
Zircon (/ˈzɜːrkɒn, -kən/) is a mineral belonging to the group of nesosilicates and is a source of the metal zirconium. Its chemical name is zirconium(IV) silicate, and its corresponding chemical formula is ZrSiO4. An empirical formula showing some of the range of substitution in zircon is (Zr1–y, REEy)(SiO4)1–x(OH)4x–y. Zircon precipitates from silicate melts and has relatively high concentrations of high field strength incompatible elements. For example, hafnium is almost always present in quantities ranging from 1 to 4%. The crystal structure of zircon is tetragonal crystal system. The natural color of zircon varies between colorless, yellow-golden, red, brown, blue, and green.
The name derives from the Persian zargun, meaning "gold-hued". This word is changed into "jargoon", a term applied to light-colored zircons. The English word "zircon" is derived from Zirkon, which is the German adaptation of this word. Yellow, orange, and red zircon is also known as "hyacinth", from the flower hyacinthus, whose name is of Ancient Greek origin. (Full article...) -
Image 3Galena with minor pyrite
Galena, also called lead glance, is the natural mineral form of lead(II) sulfide (PbS). It is the most important ore of lead and an important source of silver.
Galena is one of the most abundant and widely distributed sulfide minerals. It crystallizes in the cubic crystal system often showing octahedral forms. It is often associated with the minerals sphalerite, calcite and fluorite. (Full article...) -
Image 4

Zeolite exhibited in the Estonian Museum of Natural History
Zeolite is a family of several microporous, crystalline aluminosilicate materials commonly used as commercial adsorbents and catalysts. They mainly consist of silicon, aluminium, oxygen, and have the general formula Mn+
1/n(AlO
2)−
(SiO
2)
x・yH
2O where Mn+
1/n is either a metal ion or H+. These positive ions can be exchanged for others in a contacting electrolyte solution. H+
exchanged zeolites are particularly useful as solid acid catalysts.
The term was originally coined in 1756 by Swedish mineralogist Axel Fredrik Cronstedt, who observed that rapidly heating a material, believed to have been stilbite, produced large amounts of steam from water that had been adsorbed by the material. Based on this, he called the material zeolite, from the Greek ζέω (zéō), meaning "to boil" and λίθος (líthos), meaning "stone".
Zeolites occur naturally, but are also produced industrially on a large scale. , 253 unique zeolite frameworks have been identified, and over 40 naturally occurring zeolite frameworks are known. Every new zeolite structure that is obtained is examined by the International Zeolite Association Structure Commission (IZA-SC) and receives a three-letter designation. (Full article...) -
Image 5
Diamond is a solid form of the element carbon with its atoms arranged in a crystal structure called diamond cubic. Another solid form of carbon known as graphite is the chemically stable form of carbon at room temperature and pressure, but diamond is metastable and converts to it at a negligible rate under those conditions. Diamond has the highest hardness and thermal conductivity of any natural material, properties that are used in major industrial applications such as cutting and polishing tools. They are also the reason that diamond anvil cells can subject materials to pressures found deep in the Earth.
Because the arrangement of atoms in diamond is extremely rigid, few types of impurity can contaminate it (two exceptions are boron and nitrogen). Small numbers of defects or impurities (about one per million of lattice atoms) can color a diamond blue (boron), yellow (nitrogen), brown (defects), green (radiation exposure), purple, pink, orange, or red. Diamond also has a very high refractive index and a relatively high optical dispersion.
Most natural diamonds have ages between 1 billion and 3.5 billion years. Most were formed at depths between 150 and 250 kilometres (93 and 155 mi) in the Earth's mantle, although a few have come from as deep as 800 kilometres (500 mi). Under high pressure and temperature, carbon-containing fluids dissolved various minerals and replaced them with diamonds. Much more recently (hundreds to tens of million years ago), they were carried to the surface in volcanic eruptions and deposited in igneous rocks known as kimberlites and lamproites.
Synthetic diamonds can be grown from high-purity carbon under high pressures and temperatures or from hydrocarbon gases by chemical vapor deposition (CVD). Imitation diamonds can also be made out of materials such as cubic zirconia and silicon carbide. Natural, synthetic, and imitation diamonds are most commonly distinguished using optical techniques or thermal conductivity measurements. (Full article...) -
Image 6

Garnets ( /ˈɡɑːrnɪt/) are a group of silicate minerals that have been used since the Bronze Age as gemstones and abrasives.
All species of garnets possess similar physical properties and crystal forms, but differ in chemical composition. The different species are pyrope, almandine, spessartine, grossular (varieties of which are hessonite or cinnamon-stone and tsavorite), uvarovite and andradite. The garnets make up two solid solution series: pyrope-almandine-spessartine (pyralspite), with the composition range [Mg,Fe,Mn]3Al2(SiO4)3; and uvarovite-grossular-andradite (ugrandite), with the composition range Ca3[Cr,Al,Fe]2(SiO4)3. (Full article...) -
Image 7Deep green isolated fluorite crystal resembling a truncated octahedron, set upon a micaceous matrix, from Erongo Mountain, Erongo Region, Namibia (overall size: 50 mm × 27 mm, crystal size: 19 mm wide, 30 g)
Fluorite (also called fluorspar) is the mineral form of calcium fluoride, CaF2. It belongs to the halide minerals. It crystallizes in isometric cubic habit, although octahedral and more complex isometric forms are not uncommon.
The Mohs scale of mineral hardness, based on scratch hardness comparison, defines value 4 as fluorite.
Pure fluorite is colourless and transparent, both in visible and ultraviolet light, but impurities usually make it a colorful mineral and the stone has ornamental and lapidary uses. Industrially, fluorite is used as a flux for smelting, and in the production of certain glasses and enamels. The purest grades of fluorite are a source of fluoride for hydrofluoric acid manufacture, which is the intermediate source of most fluorine-containing fine chemicals. Optically clear transparent fluorite has anomalous partial dispersion, that is, its refractive index varies with the wavelength of light in a manner that differs from that of commonly used glases, so fluorite is useful in making apochromatic lenses, making it valuable, particularly in photographic optics. Fluorite optics are also usable in the far-ultraviolet and mid-infrared ranges, where conventional glasses are too opaque for use. Fluorite also has low dispersion, and a high refractive index for its density, which can make it useful for some specialized purposes in optics. (Full article...) -
Image 8
Graphite (/ˈɡræfaɪt/) is a crystalline form of the element carbon. It consists of stacked layers of graphene. Graphite occurs naturally and is the most stable form of carbon under standard conditions. Synthetic and natural graphite are consumed on a large scale (1.3 million metric tons per year in 2022) for uses in pencils, lubricants, and electrodes. Under high pressures and temperatures it converts to diamond. It is a good (but not excellent) conductor of both heat and electricity. (Full article...) -
Image 9

Crystal structure of table salt (sodium in purple, chlorine in green)
In crystallography, crystal structure is a description of the ordered arrangement of atoms, ions, or molecules in a crystalline material. Ordered structures occur from the intrinsic nature of the constituent particles to form symmetric patterns that repeat along the principal directions of three-dimensional space in matter.
The smallest group of particles in the material that constitutes this repeating pattern is the unit cell of the structure. The unit cell completely reflects the symmetry and structure of the entire crystal, which is built up by repetitive translation of the unit cell along its principal axes. The translation vectors define the nodes of the Bravais lattice.
The lengths of the principal axes, or edges, of the unit cell and the angles between them are the lattice constants, also called lattice parameters or cell parameters. The symmetry properties of the crystal are described by the concept of space groups. All possible symmetric arrangements of particles in three-dimensional space may be described by the 230 space groups.
The crystal structure and symmetry play a critical role in determining many physical properties, such as cleavage, electronic band structure, and optical transparency. (Full article...) -
Image 10

Corundum is a crystalline form of aluminium oxide (Al2O3) typically containing traces of iron, titanium, vanadium, and chromium. It is a rock-forming mineral. It is a naturally transparent material, but can have different colors depending on the presence of transition metal impurities in its crystalline structure. Corundum has two primary gem varieties: ruby and sapphire. Rubies are red due to the presence of chromium, and sapphires exhibit a range of colors depending on what transition metal is present. A rare type of sapphire, padparadscha sapphire, is pink-orange.
The name "corundum" is derived from the Tamil-Dravidian word kurundam (ruby-sapphire) (appearing in Sanskrit as kuruvinda).
Because of corundum's hardness (pure corundum is defined to have 9.0 on the Mohs scale), it can scratch almost all other minerals. It is commonly used as an abrasive on sandpaper and on large tools used in machining metals, plastics, and wood. Emery, a variety of corundum with no value as a gemstone, is commonly used as an abrasive. It is a black granular form of corundum, in which the mineral is intimately mixed with magnetite, hematite, or hercynite.
In addition to its hardness, corundum has a density of 4.02 g/cm3 (251 lb/cu ft), which is unusually high for a transparent mineral composed of the low-atomic mass elements aluminium and oxygen. (Full article...) -
Image 11

Gypsum is a soft sulfate mineral composed of calcium sulfate dihydrate, with the chemical formula CaSO4·2H2O. It is widely mined and is used as a fertilizer and as the main constituent in many forms of plaster, drywall and blackboard or sidewalk chalk. Gypsum also crystallizes as translucent crystals of selenite. It forms as an evaporite mineral and as a hydration product of anhydrite. The Mohs scale of mineral hardness defines gypsum as hardness value 2 based on scratch hardness comparison.
Fine-grained white or lightly tinted forms of gypsum known as alabaster have been used for sculpture by many cultures including Ancient Egypt, Mesopotamia, Ancient Rome, the Byzantine Empire, and the Nottingham alabasters of Medieval England. (Full article...) -
Image 12

Rutile is an oxide mineral composed of titanium dioxide (TiO2), the most common natural form of TiO2. Rarer polymorphs of TiO2 are known, including anatase, akaogiite, and brookite.
Rutile has one of the highest refractive indices at visible wavelengths of any known crystal and also exhibits a particularly large birefringence and high dispersion. Owing to these properties, it is useful for the manufacture of certain optical elements, especially polarization optics, for longer visible and infrared wavelengths up to about 4.5 micrometres. Natural rutile may contain up to 10% iron and significant amounts of niobium and tantalum.
Rutile derives its name from the Latin rutilus ('red'), in reference to the deep red color observed in some specimens when viewed by transmitted light. Rutile was first described in 1803 by Abraham Gottlob Werner using specimens obtained in Horcajuelo de la Sierra, Madrid (Spain), which is consequently the type locality. (Full article...) -
Image 13
Beryl (/ˈbɛrəl/ BERR-əl) is a mineral composed of beryllium aluminium silicate with the chemical formula Be3Al2Si6O18. Well-known varieties of beryl include emerald and aquamarine. Naturally occurring hexagonal crystals of beryl can be up to several meters in size, but terminated crystals are relatively rare. Pure beryl is colorless, but it is frequently tinted by impurities; possible colors are green, blue, yellow, pink, and red (the rarest). It is an ore source of beryllium. (Full article...) -
Image 14Amethyst cluster from Artigas, Uruguay
Amethyst is a violet variety of quartz. The name comes from the Koine Greek αμέθυστος amethystos from α- a-, "not" and μεθύσκω (Ancient Greek) methysko / μεθώ metho (Modern Greek), "intoxicate", a reference to the belief that the stone protected its owner from drunkenness. Ancient Greeks wore amethyst and carved drinking vessels from it in the belief that it would prevent intoxication.
Amethyst, a semiprecious stone, is often used in jewelry. (Full article...) -
Image 15Brazilian trigonal hematite crystal
Hematite (/ˈhiːməˌtaɪt, ˈhɛmə-/), also spelled as haematite, is a common iron oxide compound with the formula, Fe2O3 and is widely found in rocks and soils. Hematite crystals belong to the rhombohedral lattice system which is designated the alpha polymorph of Fe
2O
3. It has the same crystal structure as corundum (Al
2O
3) and ilmenite (FeTiO
3). With this it forms a complete solid solution at temperatures above 950 °C (1,740 °F).
Hematite naturally occurs in black to steel or silver-gray, brown to reddish-brown, or red colors. It is mined as an important ore mineral of iron. It is electrically conductive. Hematite varieties include kidney ore, martite (pseudomorphs after magnetite), iron rose and specularite (specular hematite). While these forms vary, they all have a rust-red streak. Hematite is not only harder than pure iron, but also much more brittle. Maghemite is a polymorph of hematite (γ-Fe
2O
3) with the same chemical formula, but with a spinel structure like magnetite.
Large deposits of hematite are found in banded iron formations. Gray hematite is typically found in places that have still, standing water or mineral hot springs, such as those in Yellowstone National Park in North America. The mineral can precipitate in the water and collect in layers at the bottom of the lake, spring, or other standing water. Hematite can also occur in the absence of water, usually as the result of volcanic activity.
Clay-sized hematite crystals can also occur as a secondary mineral formed by weathering processes in soil, and along with other iron oxides or oxyhydroxides such as goethite, which is responsible for the red color of many tropical, ancient, or otherwise highly weathered soils. (Full article...) -
Image 16

Green fluorite with prominent cleavage
Cleavage, in mineralogy and materials science, is the tendency of crystalline materials to split along definite crystallographic structural planes. These planes of relative weakness are a result of the regular locations of atoms and ions in the crystal, which create smooth repeating surfaces that are visible both in the microscope and to the naked eye. If bonds in certain directions are weaker than others, the crystal will tend to split along the weakly bonded planes. These flat breaks are termed "cleavage". The classic example of cleavage is mica, which cleaves in a single direction along the basal pinacoid, making the layers seem like pages in a book. In fact, mineralogists often refer to "books of mica".
Diamond and graphite provide examples of cleavage. Each is composed solely of a single element, carbon. In diamond, each carbon atom is bonded to four others in a tetrahedral pattern with short covalent bonds. The planes of weakness (cleavage planes) in a diamond are in four directions, following the faces of the octahedron. In graphite, carbon atoms are contained in layers in a hexagonal pattern where the covalent bonds are shorter (and thus even stronger) than those of diamond. However, each layer is connected to the other with a longer and much weaker van der Waals bond. This gives graphite a single direction of cleavage, parallel to the basal pinacoid. So weak is this bond that it is broken with little force, giving graphite a slippery feel as layers shear apart. As a result, graphite makes an excellent dry lubricant.
While all single crystals will show some tendency to split along atomic planes in their crystal structure, if the differences between one direction or another are not large enough, the mineral will not display cleavage. Corundum, for example, displays no cleavage. (Full article...) -
Image 17
Apatite is a group of phosphate minerals, usually hydroxyapatite, fluorapatite and chlorapatite, with high concentrations of OH−, F− and Cl− ion, respectively, in the crystal. The formula of the admixture of the three most common endmembers is written as Ca10(PO4)6(OH,F,Cl)2, and the crystal unit cell formulae of the individual minerals are written as Ca10(PO4)6(OH)2, Ca10(PO4)6F2 and Ca10(PO4)6Cl2.
The mineral was named apatite by the German geologist Abraham Gottlob Werner in 1786, although the specific mineral he had described was reclassified as fluorapatite in 1860 by the German mineralogist Karl Friedrich August Rammelsberg. Apatite is often mistaken for other minerals. This tendency is reflected in the mineral's name, which is derived from the Greek word ἀπατάω (apatáō), which means to deceive. (Full article...) -
Image 18Quartz crystal cluster from Brazil
Quartz is a hard, crystalline mineral composed of silica (silicon dioxide). The atoms are linked in a continuous framework of SiO4 silicon–oxygen tetrahedra, with each oxygen being shared between two tetrahedra, giving an overall chemical formula of SiO2. Quartz is, therefore, classified structurally as a framework silicate mineral and compositionally as an oxide mineral. Quartz is the second most abundant mineral in Earth's continental crust, behind feldspar.
Quartz exists in two forms, the normal α-quartz and the high-temperature β-quartz, both of which are chiral. The transformation from α-quartz to β-quartz takes place abruptly at 573 °C (846 K; 1,063 °F). Since the transformation is accompanied by a significant change in volume, it can easily induce microfracturing of ceramics or rocks passing through this temperature threshold.
There are many different varieties of quartz, several of which are classified as gemstones. Since antiquity, varieties of quartz have been the most commonly used minerals in the making of jewelry and hardstone carvings, especially in Europe and Asia.
Quartz is the mineral defining the value of 7 on the Mohs scale of hardness, a qualitative scratch method for determining the hardness of a material to abrasion. (Full article...) -
Image 19

Borax (also referred to as sodium borate, tincal (/ˈtɪŋkəl/) and tincar (/ˈtɪŋkər/)) is a salt (ionic compound), a hydrated or anhydrous borate of sodium, with the chemical formula Na2H20B4O17 (also written as Na2B4O7·10H2O).
It is a colorless crystalline solid that dissolves in water to make a basic solution.
It is commonly available in powder or granular form and has many industrial and household uses, including as a pesticide, as a metal soldering flux, as a component of glass, enamel, and pottery glazes, for tanning of skins and hides, for artificial aging of wood, as a preservative against wood fungus, and as a pharmaceutic alkalizer. In chemical laboratories, it is used as a buffering agent.
The terms tincal and tincar refer to native borax, historically mined from dry lake beds in various parts of Asia. (Full article...) -
Image 20Dolomite (white) on talc
Dolomite (/ˈdɒl.əˌmaɪt, ˈdoʊ.lə-/) is an anhydrous carbonate mineral composed of calcium magnesium carbonate, ideally CaMg(CO3)2. The term is also used for a sedimentary carbonate rock composed mostly of the mineral dolomite (see Dolomite (rock)). An alternative name sometimes used for the dolomitic rock type is dolostone. (Full article...) -
Image 21
A rock containing three crystals of pyrite (FeS2). The crystal structure of pyrite is primitive cubic, and this is reflected in the cubic symmetry of its natural crystal facets.
In crystallography, the cubic (or isometric) crystal system is a crystal system where the unit cell is in the shape of a cube. This is one of the most common and simplest shapes found in crystals and minerals.
There are three main varieties of these crystals:- Primitive cubic (abbreviated cP and alternatively called simple cubic)
- Body-centered cubic (abbreviated cI or bcc)
- Face-centered cubic (abbreviated cF or fcc)
Note: the term fcc is often used in synonym for the cubic close-packed or ccp structure occurring in metals. However, fcc stands for a face-centered-cubic Bravais lattice, which is not necessarily close-packed when a motif is set onto the lattice points. E.g. the diamond and the zincblende lattices are fcc but not close-packed.
Each is subdivided into other variants listed below. Although the unit cells in these crystals are conventionally taken to be cubes, the primitive unit cells often are not. (Full article...) -
Image 22
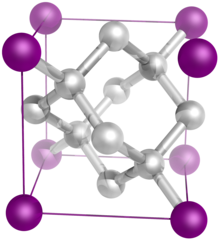
The diamond crystal structure belongs to the face-centered cubic lattice, with a repeated two-atom pattern.
In crystallography, a crystal system is a set of point groups (a group of geometric symmetries with at least one fixed point). A lattice system is a set of Bravais lattices. Space groups are classified into crystal systems according to their point groups, and into lattice systems according to their Bravais lattices. Crystal systems that have space groups assigned to a common lattice system are combined into a crystal family.
The seven crystal systems are triclinic, monoclinic, orthorhombic, tetragonal, trigonal, hexagonal, and cubic. Informally, two crystals are in the same crystal system if they have similar symmetries (albeit there are many exceptions). (Full article...) -
Image 23

Turquoise is an opaque, blue-to-green mineral that is a hydrous phosphate of copper and aluminium, with the chemical formula CuAl6(PO4)4(OH)8·4H2O. It is rare and valuable in finer grades and has been prized as a gemstone for millennia due to its hue.
Like most other opaque gems, turquoise has been devalued by the introduction of treatments, imitations, and synthetics into the market. The robin egg blue or sky blue color of the Persian turquoise mined near the modern city of Nishapur, Iran, has been used as a guiding reference for evaluating turquoise quality. (Full article...) -
Image 24
Mineralogy applies principles of chemistry, geology, physics and materials science to the study of minerals
Mineralogy is a subject of geology specializing in the scientific study of the chemistry, crystal structure, and physical (including optical) properties of minerals and mineralized artifacts. Specific studies within mineralogy include the processes of mineral origin and formation, classification of minerals, their geographical distribution, as well as their utilization. (Full article...) -
Image 25

Tourmaline (/ˈtʊərməlɪn, -ˌliːn/ TOOR-mə-lin, -leen) is a crystalline silicate mineral group in which boron is compounded with elements such as aluminium, iron, magnesium, sodium, lithium, or potassium. This gemstone comes in a wide variety of colors.
The name is derived from the Sinhalese tōramalli (ටෝරමල්ලි), which refers to the carnelian gemstones. (Full article...)
Selected mineralogist
-
Image 1Luca Bindi (born 1971) is an Italian geologist. He holds the Chair of Mineralogy and Crystallography and is the Head of the Department of Earth Sciences of the University of Florence. He is also a research associate at the Istituto di Geoscienze e Georisorse of the National Research Council (Italy) (CNR). He has received national and international scientific awards that include the President of the Republic Prize 2015 in the category of Physical, Mathematical and Natural Sciences. Since 2019 is a Member of the National Academy of Lincei.
He is the Italian scientist who has contributed to the description of the highest number of new minerals and is among the top ten researchers in the world for the number of new mineralogical species described. In his career he has described about 2% of the 6,000 minerals known in nature. Most of the new materials were discovered in the precious patrimony of the collections of the Museum System of the Florentine University, with its approximately fifty thousand specimens. The researcher is entitled to a further record: among the 130 minerals he described there are 15 extraterrestrials (almost 3% of the 500 discovered), identified in meteorite fragments. (Full article...) -
Image 2Günther Friedrich (15 April 1929 – 24 November 2014) was a German mineralogist and university professor at the RWTH University at Aachen. He was an expert in the field of the creation of marine Manganese nodule concretions. (Full article...)
-
Image 3Pencil drawing of Miers by William Rothenstein, 1917
Sir Henry Alexander Miers, FRS (25 May 1858 – 10 December 1942) was a British mineralogist and crystallographer.
Born in Rio de Janeiro, Brazil, he was educated at Eton College and Trinity College, Oxford. He was elected a Fellow of the Royal Society in 1896. (Full article...) -
Image 4Vesselina Vassileva Breskovska (Bulgarian: Веселина Василева Бресковска) (December 6, 1928, Granit, Stara Zagora Province, Bulgaria – August 12, 1997, Sofia, Bulgaria) was a 20th-century Bulgarian geologist, mineralogist and crystallographer. (Full article...)
-
Image 5Karl Hugo Strunz (24 February 1910 – 19 April 2006) was a German mineralogist. He is best known for creating the Nickel-Strunz classification, the ninth edition of which was published together with Ernest Henry Nickel. (Full article...)
-
Image 6

Johannes Mathesius (1504–1565)
Johannes Mathesius (June 24, 1504 – October 7, 1565), also called Johann Mathesius or John Mathesius, was a German minister and a Lutheran reformer. He is best known for his compilation of Martin Luther's Table Talk, or notes taken of Luther's conversation and published afterwards. He rivaled Anton Lauterbach in his diligence in notetaking, and surpassed him in the discrimination with which he arranged it. (Full article...) -
Image 7

William Babington
William Babington FRS FGS (21 May 1756 – 29 April 1833) was an Anglo-Irish physician and mineralogist. (Full article...) -
Image 8For the French Napoleonic War General see Marshall Étienne Macdonald
Marshall McDonald (October 18, 1835 – September 1, 1895) was an American engineer, geologist, mineralogist, pisciculturist, and fisheries scientist. McDonald served as the commissioner of the United States Commission of Fish and Fisheries from 1888 until his death in 1895. He is best known for his inventions of a number of fish hatching apparatuses and a fish ladder that enabled salmon and other migrating fish species to ascend the rapids of watercourses resulting in an increased spawning ground. McDonald's administration of the U.S. Commission of Fish and Fisheries was notably free of scandal and furthered the "protection and culture" of fish species throughout the United States. (Full article...) -
Image 9

Johann Carl Gehler (17 May 1732 – 6 May 1796) was a German physician, mineralogist, and anatomist.
Born 17 May 1732 in Görlitz, Gehler studied medicine from 1751 to 1758 at the University of Leipzig, where he was a pupil of physician and botanist Christian Gottlieb Ludwig. While a student at Leipzig, he furthered his interest in natural sciences, publishing the mineralogical treatise, De characteribus fossilium externis (1757), as a result. Following graduation, he continued his education by studying mineralogy in Freiberg and obstetrics in Strasbourg as a student of Johann Jakob Fried (1689–1769). (Full article...) -
Image 10Leopold Heinrich Fischer (19 December 1817, Fribourg-en-Brisgau – 2 February 1886, Fribourg-en-Brisgau) was a German zoologist and mineralogist. (Full article...)
-
Image 11

Leonid Alekseyevich Kulik (Russian: Леонид Алексеевич Кулик; 19 August 1883 – 14 or 24 April 1942) was a Soviet mineralogist who is noted for his research into meteorites. (Full article...) -
Image 12Werner Schreyer (14 November 1930 in Nuremberg; 12 February 2006 in Bochum) was a German mineralogist and experimental metamorphic petrologist. Schreyer completed his undergraduate work in geology and petrology at the University of Erlangen-Nuremberg, obtained his doctorate from the University of Munich in 1957, and in 1966 received his Habilitation from the University of Kiel. He was a professor at Ruhr University Bochum from 1966 to 1996. In 2002 Schreyer became the first German to be awarded the Mineralogical Society of America's highest honor, the Roebling Medal. Schreyer was a leading expert on phase relations in the MgO–Al2O3–SiO2–H2O (MASH) system, specializing in cordierite and minerals with equivalent chemical compositions, and high pressure and ultra high-pressure metamorphic mineral assemblages.
The mineral Schreyerite (V2Ti3O9) was named after Schreyer. (Full article...) -
Image 13

Rodney Charles Ewing (born September 20, 1946) is an American mineralogist and materials scientist whose research is focused on the properties of nuclear materials.
He is the Frank Stanton Professor in Nuclear Security at the Center for International Security and Cooperation, a Senior Fellow of the Freeman Spogli Institute for International Studies, a Senior Fellow of the Precourt Institute for Energy, an Affiliate of the Stanford Woods Institute for the Environment, and a professor in the School of Earth, Energy and Environmental Sciences at Stanford University. (Full article...) -
Image 14Paul Heinrich von Groth (1910); photograph by Theodor Hilsdorf
Paul Heinrich Ritter von Groth (23 June 1843 – 2 December 1927) was a German mineralogist. His most important contribution to science was his systematic classification of minerals based on their chemical compositions and crystal structures. (Full article...) -
Image 15Louis Jean-Pierre Cabri (born February 23, 1934, in Cairo) is an eminent Canadian scientist in the field of platinum group elements (PGE) mineralogy with expertise in precious metal mineralogy and base metals at the Canada Centre for Mineral and Energy Technology (CANMET). First as Research Scientist and later as Principal Scientist (1996–1999). In the 1970s he discovered two new Cu–Fe sulfide minerals, "mooihoekite" and "haycockite". In 1983 Russian mineralogists named a new mineral after him: cabriite (Pd2SnCu). (Full article...)
-
Image 16Maynard Bixby (June 28, 1853 – February 18, 1935) was an American mineralogist and mineral collector. (Full article...)
-
Image 17
James Smithson FRS (c. 1765 – 27 June 1829) was a British chemist and mineralogist. He published numerous scientific papers for the Royal Society during the early 1800s as well as defining calamine, which would eventually be renamed after him as "smithsonite". He was the founding donor of the Smithsonian Institution, which also bears his name.
Born in Paris, France, as the illegitimate child of Elizabeth Hungerford Keate Macie and Hugh Percy (born Hugh Smithson), the 1st Duke of Northumberland, he was given the French name Jacques-Louis Macie. His birth date was not recorded and the exact location of his birth is unknown; it is possibly in the Pentemont Abbey. Shortly after his birth he naturalized to Britain where his name was anglicized to James Louis Macie. He adopted his father's original surname of Smithson in 1800, following his mother's death. He attended university at Pembroke College, Oxford in 1782, eventually graduating with a Master of Arts in 1786. As a student he participated in a geological expedition to Scotland and studied chemistry and mineralogy. Highly regarded for his blowpipe analysis and his ability to work in miniature, Smithson spent much of his life traveling extensively throughout Europe; he published some 27 papers in his life. (Full article...) -
Image 18

Jean-Claude Delamétherie
Jean-Claude Delamétherie (also de La Métherie, de Lamétherie, 4 September 1743 – 1 July 1817) was a French mineralogist, geologist and paleontologist. (Full article...) -
Image 19Franz-Joseph Müller, Freiherr von Reichenstein or Franz-Joseph Müller von Reichenstein (1 July 1740 or 4 October 1742 – 12 October 1825 or 1826) was an Austrian mineralogist and mining engineer. Müller held several positions in the Habsburg monarchy administration of mines and coinage in the Banat, Transylvania, and Tyrol. During his time in Transylvania he discovered tellurium in 1782. In his later career he became a member of the imperial council in Vienna and was knighted and elevated to the rank Freiherr in 1820. (Full article...)
-
Image 20
(Karl) Heinrich/Harry (Ferdinand) Rosenbusch (24 June 1836 – 20 January 1914) was a German petrographer.
Harry Rosenbusch was born in Einbeck. He taught at Heidelberg University (1877–1908), where he founded the Mineralogisches-geologisches Institut. He died, aged 77, in Heidelberg. (Full article...) -
Image 21
Alexandra Navrotsky (born 20 June 1943 in New York City) is a physical chemist in the field of nanogeoscience. She is an elected member of the United States National Academy of Sciences (NAS) and the American Philosophical Society (APS). She was a board member of the Earth Sciences and Resources division of the NAS from 1995 until 2000.
In 2005, she was awarded the Urey Medal, by the European Association of Geochemistry.
In 2006, she was awarded the Harry H. Hess Medal, by the American Geophysical Union.
She is currently the director of NEAT ORU (Nanomaterials in Environment, Agriculture, and Technology Organized Research Unit), a primary program in nanogeoscience. She is distinguished professor at University of California, Davis. (Full article...) -
Image 22

Archdeacon Joseph Campbell, 1923
Joseph Campbell (1856–1933) was an Anglican priest and mineralogist in Australia. (Full article...) -
Image 23Werner Schreyer (14 November 1930 in Nuremberg; 12 February 2006 in Bochum) was a German mineralogist and experimental metamorphic petrologist. Schreyer completed his undergraduate work in geology and petrology at the University of Erlangen-Nuremberg, obtained his doctorate from the University of Munich in 1957, and in 1966 received his Habilitation from the University of Kiel. He was a professor at Ruhr University Bochum from 1966 to 1996. In 2002 Schreyer became the first German to be awarded the Mineralogical Society of America's highest honor, the Roebling Medal. Schreyer was a leading expert on phase relations in the MgO–Al2O3–SiO2–H2O (MASH) system, specializing in cordierite and minerals with equivalent chemical compositions, and high pressure and ultra high-pressure metamorphic mineral assemblages.
The mineral Schreyerite (V2Ti3O9) was named after Schreyer. (Full article...) -
Image 24

René Just Haüy (French pronunciation: [aɥi]) FRS MWS FRSE (28 February 1743 – 1 June 1822) was a French priest and mineralogist, commonly styled the Abbé Haüy after he was made an honorary canon of Notre Dame. Due to his innovative work on crystal structure and his four-volume Traité de Minéralogie (1801), he is often referred to as the "Father of Modern Crystallography". During the French Revolution he also helped to establish the metric system. (Full article...) -
Image 25Charles Louis Christ (March 12, 1916 – June 29, 1980) was an American scientist, geochemist and mineralogist. (Full article...)
Related portals
Get involved
For editor resources and to collaborate with other editors on improving Wikipedia's Minerals-related articles, see WikiProject Rocks and minerals.
General images
-
Image 1Hübnerite, the manganese-rich end-member of the wolframite series, with minor quartz in the background (from Mineral)
-
Image 2Schist is a metamorphic rock characterized by an abundance of platy minerals. In this example, the rock has prominent sillimanite porphyroblasts as large as 3 cm (1.2 in). (from Mineral)
-
Image 3Asbestiform tremolite, part of the amphibole group in the inosilicate subclass (from Mineral)
-
Image 4Diamond is the hardest natural material, and has a Mohs hardness of 10. (from Mineral)
-
Image 5Gypsum desert rose (from Mineral)
-
Image 6Sphalerite crystal partially encased in calcite from the Devonian Milwaukee Formation of Wisconsin (from Mineral)
-
Image 8When minerals react, the products will sometimes assume the shape of the reagent; the product mineral is termed a pseudomorph of (or after) the reagent. Illustrated here is a pseudomorph of kaolinite after orthoclase. Here, the pseudomorph preserved the Carlsbad twinning common in orthoclase. (from Mineral)
-
Image 12Native gold. Rare specimen of stout crystals growing off of a central stalk, size 3.7 x 1.1 x 0.4 cm, from Venezuela. (from Mineral)
-
Image 13Epidote often has a distinctive pistachio-green colour. (from Mineral)
-
Image 14Red cinnabar (HgS), a mercury ore, on dolomite. (from Mineral)
-
Image 16Muscovite, a mineral species in the mica group, within the phyllosilicate subclass (from Mineral)
-
Image 18Perfect basal cleavage as seen in biotite (black), and good cleavage seen in the matrix (pink orthoclase). (from Mineral)
-
Image 20Black andradite, an end-member of the orthosilicate garnet group. (from Mineral)
-
Image 21Pink cubic halite (NaCl; halide class) crystals on a nahcolite matrix (NaHCO3; a carbonate, and mineral form of sodium bicarbonate, used as baking soda). (from Mineral)
-
Image 22Mohs hardness kit, containing one specimen of each mineral on the ten-point hardness scale (from Mohs scale)
-
Image 23Mohs Scale versus Absolute Hardness (from Mineral)
-
Image 26An example of elbaite, a species of tourmaline, with distinctive colour banding. (from Mineral)
Did you know ...?
- ... that Treak Cliff Cavern (pictured) is one of only two remaining active sources of the ornamental mineral Blue John?
- ...that chalcocite, a profitable and desirable kind of copper ore, was particularly plentiful in the now-depleted copper mines of Cornwall, England and Bristol, Connecticut?
- ... that Gloria J. Romero is leading a campaign in the California Legislature to remove serpentine as the official state rock because it is a source of cancer-causing asbestos?
- ... that the mineral abelsonite probably formed from chlorophyll and is the only known crystalline geoporphyrin?
Subcategories
Topics
| Overview | ||
|---|---|---|
| Common minerals | ||
Ore minerals, mineral mixtures and ore deposits | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Ores |
| ||||||||
| Deposit types | |||||||||
| Borates | |||||
|---|---|---|---|---|---|
| Carbonates | |||||
| Oxides |
| ||||
| Phosphates | |||||
| Silicates | |||||
| Sulfides | |||||
| Other |
| ||||
| Crystalline | |||||||
|---|---|---|---|---|---|---|---|
| Cryptocrystalline | |||||||
| Amorphous | |||||||
| Miscellaneous | |||||||
| Notable varieties |
| ||||||
| Oxide minerals |
| ||||
|---|---|---|---|---|---|
| Silicate minerals | |||||
| Other | |||||
Gemmological classifications by E. Ya. Kievlenko (1980), updated | |||||||||
| Jewelry stones |
| ||||||||
| Jewelry-Industrial stones |
| ||||||||
| Industrial stones |
| ||||||||
Mineral identification | |
|---|---|
| "Special cases" ("native elements and organic minerals") |
|
|---|---|
| "Sulfides and oxides" |
|
| "Evaporites and similars" |
|
| "Mineral structures with tetrahedral units" (sulfate anion, phosphate anion, silicon, etc.) |
|
Associated Wikimedia
The following Wikimedia Foundation sister projects provide more on this subject:
-
Commons
Free media repository -
Wikibooks
Free textbooks and manuals -
Wikidata
Free knowledge base -
Wikinews
Free-content news -
Wikiquote
Collection of quotations -
Wikisource
Free-content library -
Wikiversity
Free learning tools -
Wiktionary
Dictionary and thesaurus
References
- Pages with French IPA
- Manually maintained portal pages from May 2019
- All manually maintained portal pages
- Portals with triaged subpages from May 2019
- All portals with triaged subpages
- All portals
- Portals with named maintainer
- Automated article-slideshow portals with 31–40 articles in article list
- Automated article-slideshow portals with 201–500 articles in article list
- Geology portals
- Minerals
- Portals needing placement of incoming links