Perimycin
| |
| Names | |
|---|---|
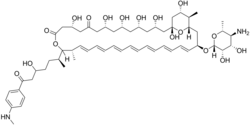
| IUPAC name
(1R,3S,5S,7R,9R,13R,17R,18S,19E,21E,23Z,25Z,27E,29E,31E,33R,35S,36R,37S)-33-[(2R,3S,4S,5S,6R)-5-amino-3,4-dihydroxy-6-methyloxan-2-yl]oxy-1,3,5,7,9,13,37-heptahydroxy-17-[(2S)-5-hydroxy-7-[4-(methylamino)phenyl]-7-oxoheptan-2-yl]-18,36-dimethyl-16,39-dioxabicyclo[33.3.1]nonatriaconta-19,21,23,25,27,29,31-heptaene-11,15-dione
| |
| Other names
4ʼ-Amino-3ʼ-deamino-18-decarboxy-40-demethyl-4ʼ-deoxy-3,7-dideoxo-3,3ʼ,7-trihydroxy-N47,18,dimethyl-5-oxocandicidin-D-cyclic-15,19-hemiacetal
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
| C59H88N2O17 | |
| Molar mass | 1097.33 g/mol |
| Appearance | Amorphous, golden-yellow solid |
| Melting point | Indefinite |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Perimycin, also known as aminomycin and fungimycin, is polyene antibiotic produced by Streptomyces coelicolor var. aminophilus.[1][2][3][4] The compound exhibits antifungal properties.[citation needed]
Composition
Perimycin is produced naturally as a mixture of three types: A, B and C, with type A being the major component. All types consist of a polyketide core with a perosamine sugar moiety. The variations occur at the end of the core opposite the perosamine moiety. Perimycin A has an aromatic group in this position, whereas the identities of the analogous groups in the other perimycin types are currently undetermined.[citation needed]
Usage
Polyene antibiotics in general are often toxic to humans and have poor bioavailability. Thus, with the notable exception of amphotericin B, they are often not used clinically. Perimycin has been shown to be an effective antifungal compound, but is not widely used in clinical settings.[5]
References
- ^ Hamilton-Miller JM (June 1973). "Chemistry and biology of the polyene macrolide antibiotics". Bacteriological Reviews. 37 (2): 166–96. doi:10.1128/br.37.2.166-196.1973. PMC 413810. PMID 4578757.
- ^ Liu CM, McDaniel LE, Schaffner CP (March 1972). "Fungimycin, biogenesis of its aromatic moiety". The Journal of Antibiotics. 25 (3): 187–8. doi:10.7164/antibiotics.25.187. PMID 5034814.
- ^ Lee CH, Schaffner CP (May 1969). "Perimycin. The structure of some degradation products". Tetrahedron. 25 (10): 2229–32. doi:10.1016/S0040-4020(01)82770-8. PMID 5788396.
- ^ Pawlak J, Sowiński P, Borowski E, Gariboldi P (September 1995). "Stereostructure of perimycin A". The Journal of Antibiotics. 48 (9): 1034–8. doi:10.7164/antibiotics.48.1034. PMID 7592049.
- ^ Elmer, Y. Tu; Charlotte E. Joslina; Lisa M. Nijmc; Robert S. Federd; Sandeep Jaina; Megan E. Shoffe (July 2009). "Polymicrobial Keratitis: Acanthamoeba and Infectious Crystalline Keratopathy". American Journal of Ophthalmology. 148 (1): 13–19.e2. doi:10.1016/j.ajo.2009.01.020. PMC 2830559. PMID 19327742.
- Articles without InChI source
- Articles without EBI source
- Articles without KEGG source
- Articles containing unverified chemical infoboxes
- Chembox image size set
- Articles with short description
- Short description matches Wikidata
- All articles with unsourced statements
- Articles with unsourced statements from September 2015
- Articles with unsourced statements from December 2022
- Macrolide antibiotics
- Antifungals
- Polyenes
- All stub articles
- Pharmacology stubs