Pathogen transmission
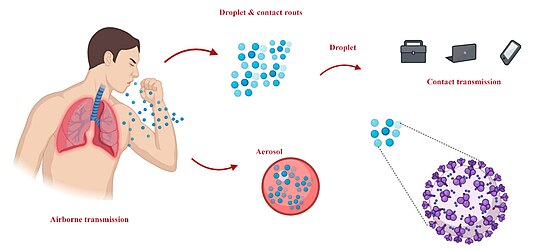
In medicine, public health, and biology, transmission is the passing of a pathogen causing communicable disease from an infected host individual or group to a particular individual or group, regardless of whether the other individual was previously infected.[1] The term strictly refers to the transmission of microorganisms directly from one individual to another by one or more of the following means:
- airborne transmission – very small dry and wet particles that stay in the air for long periods of time allowing airborne contamination even after the departure of the host. Particle size < 5 μm.
- droplet transmission – small and usually wet particles that stay in the air for a short period of time. Contamination usually occurs in the presence of the host. Particle size > 5 μm.
- direct physical contact – touching an infected individual, including sexual contact
- indirect physical contact – usually by touching a contaminated surface, including soil (fomite)
- fecal–oral transmission – usually from unwashed hands, contaminated food or water sources due to lack of sanitation and hygiene, an important transmission route in pediatrics, veterinary medicine and developing countries.
Transmission can also be indirect, via another organism, either a vector (e.g. a mosquito or fly) or an intermediate host (e.g. tapeworm in pigs can be transmitted to humans who ingest improperly cooked pork). Indirect transmission could involve zoonoses or, more typically, larger pathogens like macroparasites with more complex life cycles. Transmissions can be autochthonous (i.e. between two individuals in the same place) or may involve travel of the microorganism or the affected hosts.
An infectious disease agent can be transmitted in two ways: as horizontal disease agent transmission from one individual to another in the same generation (peers in the same age group)[2] by either direct contact (licking, touching, biting), or indirect contact through air – cough or sneeze (vectors or fomites that allow the transmission of the agent causing the disease without physical contact)[3] or by vertical disease transmission, passing the agent causing the disease from parent to offspring, such as in prenatal or perinatal transmission.[4]
The term infectivity describes the ability of an organism to enter, survive and multiply in the host, while the infectiousness of a disease agent indicates the comparative ease with which the disease agent is transmitted to other hosts.[5] Transmission of pathogens can occur by direct contact, through contaminated food, body fluids or objects, by airborne inhalation or through vector organisms.[6]
Transmissibility is the probability of an infection, given a contact between an infected host and a noninfected host.[7]
Community transmission means that the source of infection for the spread of an illness is unknown or a link in terms of contacts between patients and other people is missing. It refers to the difficulty in grasping the epidemiological link in the community beyond confirmed cases.[8][9][10]
Local transmission means that the source of the infection has been identified within the reporting location (such as within a country, region or city).[11]
Routes of transmission
The route of transmission is important to epidemiologists because patterns of contact vary between different populations and different groups of populations depending on socio-economic, cultural and other features. For example, low personal and food hygiene due to the lack of a clean water supply may result in increased transmission of diseases by the fecal-oral route, such as cholera. Differences in incidence of such diseases between different groups can also throw light on the routes of transmission of the disease. For example, if it is noted that polio is more common in cities in underdeveloped countries, without a clean water supply, than in cities with a good plumbing system, we might advance the theory that polio is spread by the fecal-oral route. Two routes are considered to be airborne: Airborne infections and droplet infections.[citation needed]
Airborne infection
"Airborne transmission refers to infectious agents that are spread via droplet nuclei (residue from evaporated droplets) containing infective microorganisms. These organisms can survive outside the body and remain suspended in the air for long periods of time. They infect others via the upper and lower respiratory tracts."[12] The size of the particles for airborne infections need to be < 5 μm.[13] It includes both dry and wet aerosols and thus requires usually higher levels of isolation since it can stay suspended in the air for longer periods of time. i.e., separate ventilation systems or negative pressure environments are needed to avoid general contamination. e.g., tuberculosis, chickenpox, measles.[citation needed]
Droplet infection

A common form of transmission is by way of respiratory droplets, generated by coughing, sneezing, or talking. Respiratory droplet transmission is the usual route for respiratory infections. Transmission can occur when respiratory droplets reach susceptible mucosal surfaces, such as in the eyes, nose or mouth. This can also happen indirectly via contact with contaminated surfaces when hands then touch the face. Before drying, respiratory droplets are large and cannot remain suspended in the air for long, and are usually dispersed over short distances.[12] The size of the particles for droplet infections are > 5 μm.[13]
Organisms spread by droplet transmission include respiratory viruses such as influenza virus, parainfluenza virus, adenoviruses, rhinovirus, respiratory syncytial virus, human metapneumovirus, Bordetella pertussis, pneumococci, streptococcus pyogenes, diphtheria, rubella,[15] and coronaviruses.[16] Spread of respiratory droplets from the wearer can be reduced through wearing of a surgical mask.[14]
Direct contact
Direct contact occurs through skin-to-skin contact, kissing, and sexual intercourse. Direct contact also refers to contact with soil or vegetation harboring infectious organisms.[17] Additionally, while fecal–oral transmission is primarily considered an indirect contact route, direct contact can also result in transmission through feces.[18][19]
Diseases that can be transmitted by direct contact are called contagious (contagious is not the same as infectious; although all contagious diseases are infectious, not all infectious diseases are contagious). These diseases can also be transmitted by sharing a towel (where the towel is rubbed vigorously on both bodies) or items of clothing in close contact with the body (socks, for example) if they are not washed thoroughly between uses. For this reason, contagious diseases often break out in schools, where towels are shared and personal items of clothing accidentally swapped in the changing rooms.[citation needed]
Some diseases that are transmissible by direct contact include athlete's foot, impetigo, syphilis, warts, and conjunctivitis.[20]
Sexual
This refers to any disease that can be caught during sexual activity with another person, including vaginal or anal sex or (less commonly) through oral sex (see below). Transmission is either directly between surfaces in contact during intercourse (the usual route for bacterial infections and those infections causing sores) or from secretions (semen or the fluid secreted by the excited female) which carry infectious agents that get into the partner's blood stream through tiny tears in the penis, vagina or rectum (this is a more usual route for viruses). In this second case, anal sex is considerably more hazardous since the penis opens more tears in the rectum than the vagina, as the vagina is more elastic and more accommodating.[citation needed]
Some diseases transmissible by the sexual route include HIV/AIDS, chlamydia, genital warts, gonorrhea, hepatitis B, syphilis, herpes, and trichomoniasis.[citation needed]
Oral sexual
Sexually transmitted diseases such as HIV and hepatitis B are thought to not normally be transmitted through mouth-to-mouth contact, although it is possible to transmit some STDs between the genitals and the mouth, during oral sex. In the case of HIV this possibility has been established. It is also responsible for the increased incidence of herpes simplex virus 1 (which is usually responsible for oral infections) in genital infections and the increased incidence of the type 2 virus (more common genitally) in oral infections.[citation needed]
Oral
Diseases that are transmitted primarily by oral means may be caught through direct oral contact such as kissing, or by indirect contact such as by sharing a drinking glass or a cigarette. Diseases that are known to be transmissible by kissing or by other direct or indirect oral contact include all of the diseases transmissible by droplet contact and (at least) all forms of herpes viruses, namely Cytomegalovirus infections herpes simplex virus (especially HSV-1) and infectious mononucleosis.[citation needed]
Mother-to-child transmission

This is from mother to child (more rarely father to child), often in utero, during childbirth (also referred to as perinatal infection) or during postnatal physical contact between parents and offspring. In mammals, including humans, it occurs also via breast milk (transmammary transmission). Infectious diseases that can be transmitted in this way include: HIV, hepatitis B and syphilis. Many mutualistic organisms are transmitted vertically.[21]
Iatrogenic
Transmission due to medical procedures, such as touching a wound, an injection or transplantation of infected material. Some diseases that can be transmitted iatrogenically include: Creutzfeldt–Jakob disease by injection of contaminated human growth hormone, MRSA and many more.[citation needed]
Indirect contact
Indirect contact transmission, also known as vehicleborne transmission, involves transmission through contamination of inanimate objects. Vehicles that may indirectly transmit an infectious agent include food, water, biologic products such as blood, and fomites such as handkerchiefs, bedding, or surgical scalpels. A vehicle may passively carry a pathogen, as in the case of food or water may carrying hepatitis A virus. Alternatively, the vehicle may provide an environment in which the agent grows, multiplies, or produces toxin, such as improperly canned foods provide an environment that supports production of botulinum toxin by Clostridium botulinum.[17]
Transmission by other organisms
A vector is an organism that does not cause disease itself but that transmits infection by conveying pathogens from one host to another.[22]
Vectors may be mechanical or biological. A mechanical vector picks up an infectious agent on the outside of its body and transmits it in a passive manner. An example of a mechanical vector is a housefly, which lands on cow dung, contaminating its appendages with bacteria from the feces, and then lands on food prior to consumption. The pathogen never enters the body of the fly. In contrast, biological vectors harbor pathogens within their bodies and deliver pathogens to new hosts in an active manner, usually a bite. Biological vectors are often responsible for serious blood-borne diseases, such as malaria, viral encephalitis, Chagas disease, Lyme disease and African sleeping sickness. Biological vectors are usually, though not exclusively, arthropods, such as mosquitoes, ticks, fleas and lice. Vectors are often required in the life cycle of a pathogen. A common strategy used to control vector-borne infectious diseases is to interrupt the life cycle of a pathogen by killing the vector.[citation needed]
Fecal–oral

In the fecal-oral route, pathogens in fecal particles pass from one person to the mouth of another person. Although it is usually discussed as a route of transmission, it is actually a specification of the entry and exit portals of the pathogen, and can operate across several of the other routes of transmission.[17] Fecal–oral transmission is primarily considered as an indirect contact route through contaminated food or water. However, it can also operate through direct contact with feces or contaminated body parts, such as through anal sex.[18][19] It can also operate through droplet or airborne transmission through the toilet plume from contaminated toilets.[23][24]
Main causes of fecal–oral disease transmission include lack of adequate sanitation and poor hygiene practices - which can take various forms. Fecal oral transmission can be via foodstuffs or water that has become contaminated. This can happen when people do not adequately wash their hands after using the toilet and before preparing food or tending to patients.[citation needed]
The fecal-oral route of transmission can be a public health risk for people in developing countries who live in urban slums without access to adequate sanitation. Here, excreta or untreated sewage can pollute drinking water sources (groundwater or surface water). The people who drink the polluted water can become infected. Another problem in some developing countries, is open defecation which leads to disease transmission via the fecal-oral route.[citation needed]
Even in developed countries there are periodic system failures resulting in a sanitary sewer overflow. This is the typical mode of transmission for infectious agents such as cholera, hepatitis A, polio, Rotavirus, Salmonella, and parasites (e.g. Ascaris lumbricoides).[citation needed]
Tracking
Tracking the transmission of infectious diseases is called disease surveillance. Surveillance of infectious diseases in the public realm traditionally has been the responsibility of public health agencies, on an international, national, or local level. Public health staff relies on health care workers and microbiology laboratories to report cases of reportable diseases to them. The analysis of aggregate data can show the spread of a disease and is at the core of the specialty of epidemiology. To understand the spread of the vast majority of non-notifiable diseases, data either need to be collected in a particular study, or existing data collections can be mined, such as insurance company data or antimicrobial drug sales for example.[citation needed]
For diseases transmitted within an institution, such as a hospital, prison, nursing home, boarding school, orphanage, refugee camp, etc., infection control specialists are employed, who will review medical records to analyze transmission as part of a hospital epidemiology program, for example.[citation needed]
Because these traditional methods are slow, time-consuming, and labor-intensive, proxies of transmission have been sought. One proxy in the case of influenza is tracking of influenza-like illness at certain sentinel sites of health care practitioners within a state, for example.[25] Tools have been developed to help track influenza epidemics by finding patterns in certain web search query activity. It was found that the frequency of influenza-related web searches as a whole rises as the number of people sick with influenza rises. Examining space-time relationships of web queries has been shown to approximate the spread of influenza[26] and dengue.[27]
Computer simulations of infectious disease spread have been used.[28] Human aggregation can drive transmission, seasonal variation and outbreaks of infectious diseases, such as the annual start of school, bootcamp, the annual Hajj etc. Most recently, data from cell phones have been shown to be able to capture population movements well enough to predict the transmission of certain infectious diseases, like rubella.[29]
Relationship with virulence and survival
Pathogens must have a way to be transmitted from one host to another to ensure their species' survival. Infectious agents are generally specialized for a particular method of transmission. Taking an example from the respiratory route, from an evolutionary perspective viruses or bacteria that cause their host to develop coughing and sneezing symptoms have a great survival advantage, as they are much more likely to be ejected from one host and carried to another. This is also the reason that many microorganisms cause diarrhea.[citation needed]
The relationship between virulence and transmission is complex and has important consequences for the long term evolution of a pathogen. Since it takes many generations for a microbe and a new host species to co-evolve, an emerging pathogen may hit its earliest victims especially hard. It is usually in the first wave of a new disease that death rates are highest. If a disease is rapidly fatal, the host may die before the microbe can be passed along to another host. However, this cost may be overwhelmed by the short-term benefit of higher infectiousness if transmission is linked to virulence, as it is for instance in the case of cholera (the explosive diarrhea aids the bacterium in finding new hosts) or many respiratory infections (sneezing and coughing create infectious aerosols).[citation needed]
Anything that reduces the rate of transmission of an infection carries positive externalities, which are benefits to society that are not reflected in a price to a consumer. This is recognized implicitly when vaccines are offered for free or at a cost to the patient less than the purchase price.[citation needed]
Beneficial microorganisms
The mode of transmission is also an important aspect of the biology of beneficial microbial symbionts, such as coral-associated dinoflagellates or human microbiota. Organisms can form symbioses with microbes transmitted from their parents, from the environment or unrelated individuals, or both.[citation needed]
Vertical transmission
Vertical transmission refers to acquisition of symbionts from parents (usually mothers). Vertical transmission can be intracellular (e.g. transovarial), or extracellular (for example through post-embryonic contact between parents and offspring). Both intracellular and extracellular vertical transmission can be considered a form of non-genetic inheritance or parental effect. It has been argued that most organisms experience some form of vertical transmission of symbionts.[30] Canonical examples of vertically transmitted symbionts include the nutritional symbiont Buchnera in aphids (transovarially transmitted intracellular symbiont) and some components of the human microbiota (transmitted during passage of infants through the birth canal and also through breastfeeding).[citation needed]
Horizontal transmission
Some beneficial symbionts are acquired horizontally, from the environment or unrelated individuals. This requires that host and symbiont have some method of recognizing each other or each other's products or services. Often, horizontally acquired symbionts are relevant to secondary rather than primary metabolism, for example for use in defense against pathogens,[31] but some primary nutritional symbionts are also horizontally (environmentally) acquired.[32] Additional examples of horizontally transmitted beneficial symbionts include bioluminescent bacteria associated with bobtail squid and nitrogen-fixing bacteria in plants.[citation needed]
Mixed-mode transmission
Many microbial symbionts, including human microbiota, can be transmitted both vertically and horizontally. Mixed-mode transmission can allow symbionts to have the “best of both worlds” – they can vertically infect host offspring when host density is low, and horizontally infect diverse additional hosts when a number of additional hosts are available. Mixed-mode transmission make the outcome (degree of harm or benefit) of the relationship more difficult to predict, because the evolutionary success of the symbiont is sometimes but not always tied to the success of the host.[21]
See also
- Bioaerosol
- Bugchasing
- Cross-species transmission
- Infectious disease: Transmission
- Transmission coefficient (epidemiology)
- Transmission risks and rates
References
- ↑ Bush, Albert O.; Fernandez, Jacqueline C.; Esch, Gerald W.; Seed, J. Richard (2001). Parasitism: The Diversity and Ecology of Animal Parasites. Cambridge, UK: Cambridge University Press. pp. 391–399. ISBN 9780521664479.
- ↑ Horizontal Disease Transmission Archived 27 September 2007 at the Wayback Machine, online-medical-dictionary.org. date ?
- ↑ Routes of transmission of infectious diseases agents Archived 15 March 2012 at the Wayback Machine from Modes of Introduction of Exotic Animal Disease Agents by Katharine M. Kurkjian & Susan E. Little of The University of Georgia College of Veterinary Medicine, date?
- ↑ Vertical transmission Archived 28 March 2007 at the Wayback Machine (definition -- medterms.com) date?
- ↑ "Glossary of Notifiable Conditions". Washington State Department of Health. Archived from the original on 7 January 2010. Retrieved 3 February 2010.
- ↑ Ryan KJ; Ray CG (editors) (2004). Sherris Medical Microbiology (4th ed.). McGraw Hill. ISBN 978-0-8385-8529-0.
{{cite book}}: CS1 maint: multiple names: authors list (link) - ↑ J.H. Jones, Notes on R0 Archived 2014-09-12 at the Wayback Machine. Stanford University (2007).
- ↑ "Novel Coronavirus (COVID-19) Resources". Archived from the original on 2021-03-10. Retrieved 2023-02-11.
- ↑ "Gainers and losers in the time of nCoV". The Manila Times. 10 February 2020. Archived from the original on 18 April 2020. Retrieved 11 February 2023.
- ↑ "Global economy looks woozy". Dallas Morning News. 28 February 2020. Archived from the original on 25 May 2022. Retrieved 11 February 2023 – via pressreader.com.(subscription required)
- ↑ "Coronavirus disease 2019 (COVID-19) Situation Report – 47" (PDF). World Health Organization. Archived (PDF) from the original on 8 March 2020. Retrieved 8 March 2020.
- ↑ 12.0 12.1 NHMRC (2010). "Clinical Educators Guide for the Prevention and Control of Infection in Healthcare" (PDF). Commonwealth of Australia. Archived (PDF) from the original on 5 May 2022. Retrieved 10 November 2020.
- ↑ 13.0 13.1 "Prevention of hospital-acquired infections" (PDF). World Health Organization (WHO). Archived from the original (PDF) on 26 March 2020.
- ↑ 14.0 14.1 "Respiratory Protection Against Airborne Infectious Agents for Health Care Workers: Do surgical masks protect workers?" (OSH Answers Fact Sheets). Canadian Centre for Occupational Health and Safety. 28 February 2017. Retrieved 28 February 2017.
- ↑ "What is Diseases contagious from droplets?". Archived from the original on 16 July 2015.
- ↑ "Pass the message: Five steps to kicking out coronavirus". www.who.int. Archived from the original on 24 March 2020. Retrieved 24 March 2020.
- ↑ 17.0 17.1 17.2 "Principles of Epidemiology: Chain of Infection". U.S. Centers for Disease Control and Infection. 2019-02-18. Archived from the original on 2020-07-23. Retrieved 2020-07-21.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain.
- ↑ 18.0 18.1 LaMorte, Wayne W. (2016-01-06). "Common Vehicle Spread". Boston University School of Public Health. Archived from the original on 2020-05-14. Retrieved 2020-07-21.
- ↑ 19.0 19.1 Whittier, Christopher A. (2017-04-16), "Fecal-Oral Transmission", in Bezanson, Michele; MacKinnon, Katherine C; Riley, Erin; Campbell, Christina J (eds.), The International Encyclopedia of Primatology, Hoboken, NJ, USA: John Wiley & Sons, Inc., p. 1, doi:10.1002/9781119179313.wbprim0193, ISBN 978-1-119-17931-3
- ↑ Morse, Stephen A.; Mietzner, Timothy A.; Miller, Steve; Riedel, Stefan (2019). Jawetz, Melnick & Adelberg's Medical Microbiology (28th ed.). New York. ISBN 978-1-260-01202-6.
- ↑ 21.0 21.1 Ebert, Dieter (2013). "The Epidemiology and Evolution of Symbionts with Mixed-Mode Transmission". Annual Review of Ecology, Evolution, and Systematics. 44: 623–643. doi:10.1146/annurev-ecolsys-032513-100555.
- ↑ Pathogens and vectors Archived 24 January 2015 at the Wayback Machine. MetaPathogen.com.
- ↑ Johnson, David L.; Mead, Kenneth R.; Lynch, Robert A.; Hirst, Deborah V.L. (March 2013). "Lifting the lid on toilet plume aerosol: A literature review with suggestions for future research". American Journal of Infection Control. 41 (3): 254–258. doi:10.1016/j.ajic.2012.04.330. PMC 4692156. PMID 23040490.
- ↑ Jones, RM; Brosseau, L. M. (May 2015). "Aerosol transmission of infectious disease". Journal of Occupational and Environmental Medicine. 57 (5): 501–8. doi:10.1097/JOM.0000000000000448. PMID 25816216. S2CID 11166016.
- ↑ Polgreen P.M., Chen E., Segre A.M., Harris M., Pentella M., Rushton G. (2009). "Optimizing Influenza Sentinel Surveillance at the State Level American". Journal of Epidemiology. 170 (10): 1300–1306. doi:10.1093/aje/kwp270. PMC 2800268. PMID 19822570.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ↑ Ginsberg J., Mohebbi M.H., Patel R.S., Brammer L., Smolinski M.S., Brilliant L. (2008). "Detecting influenza epidemics using search engine query data" (PDF). Nature. 457 (7232): 1012–1014. doi:10.1038/nature07634. PMID 19020500. S2CID 125775. Archived from the original (PDF) on 2018-10-24.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ↑ Chan E.H., Sahai V., Conrad C., Brownstein J.S. (2011). "Using Web Search Query Data to Monitor Dengue Epidemics: A New Model for Neglected Tropical Disease Surveillance". PLOS Negl Trop Dis. 5 (5): e1206. doi:10.1371/journal.pntd.0001206. PMC 3104029. PMID 21647308.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ↑ Siettos CI, Russo L (15 May 2013). "Mathematical modeling of infectious disease dynamics". Virulence. 4 (4): 295–306. doi:10.4161/viru.24041. PMC 3710332. PMID 23552814.
- ↑ Wesolowski A, Metcalf CJ, Eagle N, Kombich J, Grenfell BT, Bjørnstad ON, Lessler J, Tatem AJ, Buckee CO. (1 September 2015). "Quantifying seasonal population fluxes driving rubella transmission dynamics using mobile phone data". PNAS. 112 (35): 11114–11119. Bibcode:2015PNAS..11211114W. doi:10.1073/pnas.1423542112. PMC 4568255. PMID 26283349.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ↑ Funkhouser, Lisa; Bordenstein, Seth (2013). "Mom Knows Best: The Universality of Maternal Microbial Transmission". PLOS Biology. 11 (8): e1001631. doi:10.1371/journal.pbio.1001631. PMC 3747981. PMID 23976878.
- ↑ Kaltenpoth, Martin; Engl, Tobias (2013). "Defensive microbial symbionts in Hymenoptera". Functional Ecology. 28 (2): 315–327. doi:10.1111/1365-2435.12089. hdl:11858/00-001M-0000-000E-B76B-E.
- ↑ Nussbaumer, Andrea; Fisher, Charles; Bright, Monika (2006). "Horizontal endosymbiont transmission in hydrothermal vent tubeworms". Nature. 441 (7091): 345–348. Bibcode:2006Natur.441..345N. doi:10.1038/nature04793. PMID 16710420. S2CID 18356960.
- Webarchive template wayback links
- CS1 maint: multiple names: authors list
- Pages containing links to subscription-only content
- Source attribution
- Articles with hatnote templates targeting a nonexistent page
- All articles with unsourced statements
- Articles with unsourced statements from June 2021
- Articles with invalid date parameter in template
- Articles with unsourced statements from September 2015
- Articles with unsourced statements
- Articles with unsourced statements from February 2023
- Disease transmission
- Human diseases and disorders