Paraneoplastic cerebellar degeneration
| Paraneoplastic cerebellar degeneration | |
|---|---|
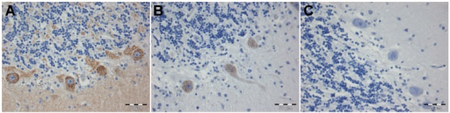 | |
| a-c) Staining of human Purkinje cells with CDR2 and CDR2L antibodies | |
| Specialty | Oncology |
Paraneoplastic cerebellar degeneration (PCD) is a paraneoplastic syndrome associated with a broad variety of tumors including lung cancer, ovarian cancer, breast cancer, Hodgkin’s lymphoma and others. PCD is a rare condition that occurs in less than 1% of cancer patients.
As is the case with other paraneoplastic syndromes,[1] PCD is believed to be due to an autoimmune reaction targeted against components of the central nervous system, mostly to Purkinje cells.[2][3][4][5]
It is thought to be triggered when tumor cells (in PCD, most commonly ovarian or breast cancer[1][6]) ectopically express proteins normally expressed in the cerebellum. This is believed to trigger an anti-tumor immune response that may be clinically significant, but also an anti-neural immune response.[7] A broad spectrum of neuronal and glial proteins has been identified as target antigens in PCD.[2][3][4]
Signs and symptoms
Neurological symptoms may include, among others, dysarthria, truncal, limb and gait ataxia and nystagmus.[1] Symptoms often develop subacutely and progress rapidly over a period of weeks or months to a plateau period that can last for months to years and which often reflects complete loss of Purkinje cells.
Pathophysiology
The anti-Purkinje cell antibodies originally described in PCD led to the hypothesis that the antibody might be pathogenic, much as earlier studies had demonstrated pathogenicity of anti-acetylcholine receptor antibodies in myasthenia gravis. However, when the antibody was used to clone the cDNA encoding the cdr2 antigen, it was found to be an intracellular protein. This led to the suggestion[8] that there might be a cell-mediated component (T cell) in disease pathogenesis. cdr2 antigen-specific CD8+ T cells were subsequently described[9] in more anti-Yo-positive PCD patients,.[10] These T cells are likely components in both the anti-tumor immune response and in the neuronal degeneration.
Diagnosis

Of particular note, PCD symptoms precede the diagnosis of the underlying cancer in the majority of cases,[12]
and often present insidiously and progress rapidly for weeks to months to a severely disabled state followed by a variable plateau period that can last for months to years.[1]
Therefore, newly developing cerebellar ataxia should always prompt proper diagnostic measures to exclude PCD.
Treatment
Tumor removal is still the therapeutic mainstay with very early treatment being essential to prevent irreversible neuronal loss. Immunosuppressive or immunomodulatory treatments are often ineffective. There may be a role for high-dose gammaglobulin therapy in the treatment PCD, but due to the rare occurrence of this disease, controlled trials of this therapy may be difficult.[1]
References
- ↑ 1.0 1.1 1.2 1.3 1.4 Paraneoplastic Syndromes, 2011, Darnell & Posner
- ↑ 2.0 2.1 S. Jarius, B. Wildemann: ‘Medusa head ataxia’: the expanding spectrum of Purkinje cell antibodies in autoimmune cerebellar ataxia. Part 1: Anti-mGluR1, anti-Homer-3, anti-Sj/ITPR1 and anti-CARP VIII 2015; 12, 166 (free) Archived 2020-04-26 at the Wayback Machine
- ↑ 3.0 3.1 S. Jarius, B. Wildemann: ‘Medusa head ataxia’: the expanding spectrum of Purkinje cell antibodies in autoimmune cerebellar ataxia. Part 2: Anti-PKC-gamma, anti-GluR-delta2, anti-Ca/ARHGAP26 and anti-VGCC 2015; 12, 167 (free) Archived 2020-04-26 at the Wayback Machine
- ↑ 4.0 4.1 S. Jarius, B. Wildemann: ‘Medusa head ataxia’: the expanding spectrum of Purkinje cell antibodies in autoimmune cerebellar ataxia. Part 3: Anti-Yo/CDR2, anti-Nb/AP3B2, PCA-2, anti-Tr/DNER, other antibodies, diagnostic pitfalls, summary and outlook 2015; 12, 168 (free) Archived 2020-04-26 at the Wayback Machine
- ↑ Jaeckle KA, Graus F, Houghton A, Cardon-Cardo C, Nielsen SL, Posner JB (1985). "Autoimmune response of patients with paraneoplastic cerebellar degeneration to a Purkinje cell cytoplasmic protein antigen". Ann. Neurol. 18 (5): 592–600. doi:10.1002/ana.410180513. PMID 2416270. S2CID 21318072.
- ↑ Peterson K, Rosenblum MK, Kotanides H, Posner JB (1992). "Paraneoplastic cerebellar degeneration. I. A clinical analysis of 55 anti-Yo antibody-positive patients". Neurology. 42 (10): 1931–7. doi:10.1212/wnl.42.10.1931. PMID 1407575.
- ↑ Roberts WK, Darnell RB (2004). "Neuroimmunology of the paraneoplastic neurological degenerations". Curr. Opin. Immunol. 16 (5): 616–22. doi:10.1016/j.coi.2004.07.009. PMID 15342008.
- ↑ Darnell, R.B. (1996), "Onconeural antigens and the paraneoplastic neurologic disorders: at the intersection of cancer, immunity, and the brain", Proc Natl Acad Sci U S A, 93 (10): 4529–4536, Bibcode:1996PNAS...93.4529D, doi:10.1073/pnas.93.10.4529, PMC 39311, PMID 8643438
- ↑ Albert ML, Austin LM, Darnell RB (2000). "Detection and treatment of activated T cells in the cerebrospinal fluid of patients with paraneoplastic cerebellar degeneration". Ann. Neurol. 47 (1): 9–17. doi:10.1002/1531-8249(200001)47:1<9::aid-ana5>3.0.co;2-i. PMID 10632096. S2CID 12989492.
- ↑ Darnell RB, Albert ML (2000). "cdr2-specific CTLs are detected in the blood of all patients with paraneoplastic cerebellar degeneration analyzed". Ann. Neurol. 48 (2): 270–1. doi:10.1002/1531-8249(200008)48:2<270::aid-ana25>3.3.co;2-p. PMID 10939585.
- ↑ Liontos, Michalis; Fiste, Oraianthi; Drakopoulou, Danai; Thomakos, Nikolaos; Goula, Kalliroi; Zagouri, Flora; Anagnostouli, Maria; Dimopoulos, Meletios-Athanasios (1 August 2021). "Paraneoplastic cerebellar degeneration in platinum-responsive endometrial cancer: A case report and review of literature". Gynecologic Oncology Reports. 37: 100826. doi:10.1016/j.gore.2021.100826. ISSN 2352-5789.
- ↑ Frings, Markus; Antoch, Gerald; Knorn, Philipp; Freudenberg, Lutz; Bier, Ulrich; Timmann, Dagmar; Maschke, Matthias (2005). "Strategies in detection of the primary tumour in anti-Yo associated paraneoplastic cerebellar degeneration". Journal of Neurology. 252 (2): 197–201. doi:10.1007/s00415-005-0635-0. ISSN 0340-5354. PMID 15729526. S2CID 34363554.
External links
| Classification | |
|---|---|
| External resources |