Osteoid osteoma
| Osteoid osteoma | |
|---|---|
| Other names: Benign osteogenic tumors[1] | |
 | |
| Osteoid osteoma of the trochanter minor: X-ray and MRI with marked sclerosis around the nidus | |
| Specialty | Orthopedics |
| Symptoms | Bone pain at night, swelling[1] |
| Frequency | 2Males:Females[1] |
An osteoid osteoma is a non-cancerous bone tumor of the osteogenic type, which may present with a bony swelling and pain at night, typically in one of the long bones of the leg.[1][2] It affects mainly children and young adults and does not spread.[3]
It arises from osteoblasts and some components of osteoclasts. It was originally thought to be a smaller version of an osteoblastoma. Osteoid osteomas tend to be less than 1.5 cm in size. The tumor can be in any bone in the body but are most common in long bones, such as the femur and tibia. They account for 10-12% of all benign bone tumors. "Osteoid osteomas may occur at any age, and are most common in people between the ages of 4 and 25 years old. Males are affected approximately three times more commonly than females."[3]
Signs and symptoms
Clinical features vary according to the location of the tumor.[3] Generally there is a dull pain that is worse at night.[3] Physical activity does not usually affect the pain and the pain may have been present for years before seeking help. There may be a swelling.[3]
The most common symptom is pain that can be relieved with over the counter pain medication in the beginning. After the benign tumor develops further the pain can not be alleviated with medication and minor to severe swelling starts to occur. Although, in some cases the pain level remains the same for years, and regular NSAIDs intake keeps the pain at bay. The tumor is often found through x-ray imaging. "Conventional radiographs reveal a well-demarcated lytic lesion (nidus) surrounded by a distinct zone of sclerosis" that allow doctors to identify the tumor.[4]
Characterized by being less than 1.5 cm in diameter, osteoid osteomas most frequently occur in young men (Male:Female ratio 3:1) and may occur in any bone of the body, most frequently around the knee but often also seen in the vertebrae, in the long bones and less commonly in the mandible or other craniofacial bones.[citation needed]
Severe pain typically occurs at night, but can also be constant at any time of day.[5] The chief complaint may only be of dull pain which is non radiating and persistent throughout 24 hours but increases significantly at night. Pain tends to be relieved with NSAIDs such as ibuprofen.[6]
Mechanism

On histological examination, osteoid osteoma consists of a small (< 1.52 cm), yellowish-to-red nidus of osteoid and woven bone with interconnected trabeculae, and a background and rim of highly vascularized, fibrous connective tissue. Varying degrees of sclerotic bone reaction may surround the lesion. Benign osteoblastoma is virtually indistinguishable from osteoid osteoma. The usual appearance included a fibrovascular stroma with numerous osteoblasts, osteoid tissue, well-formed woven bone, and giant cells. The osteoblasts were generally small and regular in shape.[7]
Diagnosis
Radiographs in osteoid osteoma typically show a round lucency, containing a dense sclerotic central nidus (the characteristic lesion in this kind of tumor) surrounded by sclerotic bone. The nidus is seldom larger than 1.5 cm.[citation needed]
The lesion can in most cases be detected on CT scan, bone scans and angiograms. Plain radiographs are not always diagnostic. MRI adds little to the CT findings which are useful for localisation. Radionuclide scanning shows intense uptake which is useful for localisation at surgery using a hand held detector, and for confirmation that the entire lesion has been removed.[8][9]
-
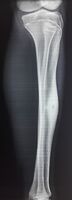
1 a. Osteoid osteoma (front view) showing cortical thickening mid-tibia
-
1 b. Osteoid osteoma (side view): clearly seen by high resolution CT with a small well-defined lucent nidus in the mid tibial shaft.
-
2. CT scan showing an osteoid osteoma of the fibula with a clearly visible nidus
-
Nidus demonstrated by high resolution CT
Treatment
Pain may be relieved by nonsteroidal anti-inflammatory drugs.
Treatment varies based on the health of the patient. If he/she is otherwise healthy and is not significantly bothered by the pain, the tumor is treated symptomatically with anti-inflammatories. If this therapy fails or the location of the tumor could lead to growth disturbances, scoliosis, or osteoarthritis, surgical or percutaneous ablation may be considered.[10] If surgery is preferred, the individual may be referred to a podiatrist or an orthopedic surgeon to perform the procedure. Post-surgery therapy and strengthening may be needed, depending on the tumor location and health of the individual. While shown to be effective, surgical resection has many potential complications, including difficult intraoperative identification of the tumor, local recurrence from incomplete resection, and resection of weight bearing bone that can necessitate prolonged hospital stays and complicate recovery.[11]
Recently, CT guided radiofrequency ablation has emerged as a less invasive alternative to surgical resection. In this technique, which can be performed under conscious sedation, an RF probe is introduced into the tumor nidus through a cannulated needle under CT guidance and heat is applied locally to destroy tumor cells. Since the procedure was first introduced for the treatment of osteoid osteomas in the early 1990s,[12] it has been shown in numerous studies to be less invasive and expensive, to result in less bone destruction and to have equivalent safety and efficacy to surgical techniques, with 66 to 96% of patients reporting freedom from symptoms.[13][14][15] While initial success rates with RFA are high, symptom recurrence after RFA treatment has been reported, with some studies demonstrating a recurrence rate similar to that of surgical treatment.[16] As of July 17, 2014, treatment with incisionless surgery utilizing an MRI to guide high-intensity ultrasound waves to destroy a benign bone tumor in the leg has been demonstrated.[17]
On November 27, 2020, the Food and Drug Administration approved the Sonalleve MR-HIFU system (Profound Medical Inc.) for the treatment of osteoid osteoma in the extremities.
MR-guided High Intensity Focused Ultrasound (MR-HIFU) treatment is an image guided technique combining high intensity focused ultrasound ablation with real time monitoring of temperature change during the sonication.
The clinical results support the probable benefit of Sonalleve MR-HIFU system for the ablation of painful osteoid osteoma. Efficacy was evaluated in a study of nine patients treated with MR-HIFU, without technical difficulties or serious adverse events. There was a statistically significant decrease in their pain scores within 4 weeks of treatment. No pain medication usage was achieved in 8 of 9 patients after 4 weeks. [18]
References
- ↑ 1.0 1.1 1.2 1.3 WHO Classification of Tumours Editorial Board, ed. (2020). "Osteoid osteoma". Soft Tissue and Bone Tumours: WHO Classification of Tumours. Vol. 3 (5th ed.). Lyon (France): International Agency for Research on Cancer. pp. 394–396. ISBN 978-92-832-4503-2. Archived from the original on 2021-06-13. Retrieved 2021-05-10.
- ↑ "Bone tumours. What are Bone Tumours?". patient.info. Archived from the original on 24 April 2021. Retrieved 24 April 2021.
- ↑ 3.0 3.1 3.2 3.3 3.4 "Osteoid Osteoma". www.orthoinfo.org. American Academy of Orthopaedic Surgeons. Archived from the original on 3 May 2021. Retrieved 3 May 2021.
- ↑ Singh, Arun Pal. "Osteoid Osteoma-Diagnosis And Treatment". Archived from the original on 2011-04-26. Retrieved 2011-04-25.
- ↑ Mungo, David V.; Zhang, Xinping; O'Keefe, Regis J.; Rosier, Randy N.; Puzas, J. Edward; Schwarz, Edward M. (2002). "COX-1 and COX-2 expression in osteoid osteomas". Journal of Orthopaedic Research. 20 (1): 159–62. doi:10.1016/S0736-0266(01)00065-1. PMID 11853083.
- ↑ Atesok, Kivanc I.; Alman, Benjamin A.; Schemitsch, Emil H.; Peyser, Amos; Mankin, Henry (2011). "Osteoid osteoma and osteoblastoma". The Journal of the American Academy of Orthopaedic Surgeons. 19 (11): 678–89. doi:10.5435/00124635-201111000-00004. PMID 22052644. Archived from the original on 2021-08-28. Retrieved 2015-07-06.
- ↑ "Osteoid Osteomas and Osteoblastomas of the Spine". Archived from the original on 2021-06-06. Retrieved 2011-02-21.
- ↑ Osteoid Osteoma Imaging at eMedicine
- ↑ Lateur, L.; Baert, A. L. (1977). "Localisation and diagnosis of osteoid osteoma of the carpal area by angiography". Skeletal Radiology. 2 (2): 75–9. doi:10.1007/BF00360985.
- ↑ "Osteoid Osteoma | BoneTumor.org". www.bonetumor.org. Archived from the original on 2021-06-06. Retrieved 2021-03-24.
- ↑ Sim, F. H.; Dahlin, C. D.; Beabout, J. W. (1975-03-01). "Osteoid-osteoma: diagnostic problems". J Bone Joint Surg Am. 57 (2): 154–159. doi:10.2106/00004623-197557020-00004. ISSN 0021-9355. PMID 1112841. Archived from the original on 2016-10-06. Retrieved 2016-08-07.
- ↑ Rosenthal, D I; Alexander, A; Rosenberg, A E; Springfield, D (1992-04-01). "Ablation of osteoid osteomas with a percutaneously placed electrode: a new procedure". Radiology. 183 (1): 29–33. doi:10.1148/radiology.183.1.1549690. ISSN 0033-8419. PMID 1549690.
- ↑ Weber, Marc-André; Sprengel, Simon David; Omlor, Georg W.; Lehner, Burkhard; Wiedenhöfer, Bernd; Kauczor, Hans-Ulrich; Rehnitz, Christoph (2015-04-25). "Clinical long-term outcome, technical success, and cost analysis of radiofrequency ablation for the treatment of osteoblastomas and spinal osteoid osteomas in comparison to open surgical resection". Skeletal Radiology. 44 (7): 981–993. doi:10.1007/s00256-015-2139-z. ISSN 0364-2348. PMID 25910709.
- ↑ Rosenthal, Daniel I.; Hornicek, Francis J.; Torriani, Martin; Gebhardt, Mark C.; Mankin, Henry J. (2003-10-01). "Osteoid Osteoma: Percutaneous Treatment with Radiofrequency Energy". Radiology. 229 (1): 171–175. doi:10.1148/radiol.2291021053. ISSN 0033-8419. PMID 12944597.[permanent dead link]
- ↑ Rimondi, Eugenio; Mavrogenis, Andreas F.; Rossi, Giuseppe; Ciminari, Rosanna; Malaguti, Cristina; Tranfaglia, Cristina; Vanel, Daniel; Ruggieri, Pietro (2011-08-14). "Radiofrequency ablation for non-spinal osteoid osteomas in 557 patients". European Radiology. 22 (1): 181–188. doi:10.1007/s00330-011-2240-1. ISSN 0938-7994. PMID 21842430.
- ↑ Rosenthal, Daniel I.; Hornicek, Francis J.; Wolfe, Michael W.; Jennings, L. Candace; Gebhardt, Mark C.; Mankin, Henry J. (1998-06-01). "Percutaneous Radiofrequency Coagulation of Osteoid Osteoma Compared with Operative Treatment*". J Bone Joint Surg Am. 80 (6): 815–21. CiteSeerX 10.1.1.1018.5024. doi:10.2106/00004623-199806000-00005. ISSN 0021-9355. PMID 9655099. Archived from the original on 2016-10-06. Retrieved 2016-08-07.
- ↑ Focused Ultrasound Foundation. "Bone tumor destroyed using incisionless surgery: First in North American child." ScienceDaily. ScienceDaily, 6 August 2014. <www.sciencedaily.com/releases/2014/08/140806142126.htm>.
- ↑ Research, Center for Drug Evaluation and (2020-11-27). "FDA approves device for treatment of osteoid osteoma in the extremities". FDA. Archived from the original on 2020-11-27. Retrieved 2020-11-28.
External links
| Classification | |
|---|---|
| External resources |
- Pages with script errors
- All articles with dead external links
- Articles with dead external links from July 2018
- Articles with invalid date parameter in template
- Articles with permanently dead external links
- All articles with unsourced statements
- Articles with unsourced statements from July 2015
- Anatomical pathology
- Osseous and chondromatous neoplasia