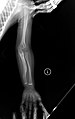
Osteogenesis imperfecta
| Osteogenesis imperfecta | |
|---|---|
| Other names: Brittle bone disease,[1] Lobstein syndrome,[2] fragilitas ossium,[1] Vrolik disease,[1] osteopsathyrosis, Porak disease, Durante disease[3] | |
 | |
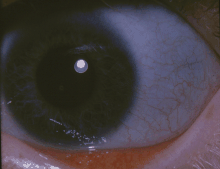
| The classic blue sclerae of a person with osteogenesis imperfecta | |
| Specialty | Pediatrics, medical genetics, osteology |
| Symptoms | Bones that break easily, blue tinge to the whites of the eye, short height, loose joints, hearing loss[1][4] |
| Duration | Long term[4] |
| Causes | Genetic (autosomal dominant, new mutation)[1] |
| Diagnostic method | Based on symptoms, DNA testing[4] |
| Treatment | Healthy lifestyle (exercise, no smoking), metal rods through the long bones[5] |
| Prognosis | Depends on the type[4] |
| Frequency | 1 in 15,000 people[1] |
Osteogenesis imperfecta (OI), also known as brittle bone disease, is a group of genetic disorders that mainly affect the bones.[1][6] It results in bones that break easily.[1] The severity may be mild to severe.[1] Other symptoms may include a blue tinge to the whites of the eye, short height, loose joints, hearing loss, breathing problems and problems with the teeth.[1][4] Complications may include cervical artery dissection and aortic dissection.[7][8]
The underlying mechanism is usually a problem with connective tissue due to a lack of type I collagen.[1] This occurs in more than 90% of cases due to mutations in the COL1A1 or COL1A2 genes.[1] These genetic problems are often inherited from a person's parents in an autosomal dominant manner or occur via a new mutation.[1] There are at least eight main types, with type I being the least severe and type II the most severe.[1] Diagnosis is often based on symptoms and may be confirmed by collagen or DNA testing.[4]
There is no cure.[4] Maintaining a healthy lifestyle by exercising and avoiding smoking can help prevent fractures.[5] Treatment may include care of broken bones, pain medication, physical therapy, braces or wheelchairs and surgery.[5] A type of surgery that puts metal rods through long bones may be done to strengthen them.[5] Tentative evidence supports the use of medications of the bisphosphonate type.[9][10]
OI affects about one in 15,000 people.[1] Outcomes depend on the type of disease.[4] Most people, however, have good outcomes.[4] The condition has been described since ancient history.[11] The term "osteogenesis imperfecta" came into use in 1895 and means imperfect bone formation.[1][11]
Signs and symptoms
Connective tissues
OI causes very thin blood vessels and may also result in people being bruised easily. The weakening of the muscles will result in bone deformities and growth issues.[citation needed]
Hearing
About 50% of the adults with OI experience significant hearing loss, often being affected by earlier hearing loss compared to the general population. Hearing loss in OI may or may not be associated with visible deformities of the ossicles and inner ear.[12] Hearing loss frequently begins between the second and fourth decade of life, and may be conductive, sensorineural, or mixed in nature.[13]
Neurological
OI is associated with a number of neurological abnormalities, usually involving the central nervous system, due to deformation of the skeletal structures enveloping it. Neurological complications may adversely affect life expectancy, and neurosurgical intervention may be needed to correct severe complications.[14]
Gastrointestinal
OI may be associated with recurrent abdominal pain and chronic constipation, according to two studies on subjects affected by OI.[15][16][17]
Subtypes
There are at least nine different types of OI. Type I is the most common. Symptoms vary from person to person.
| Type | Description | Gene | OMIM | Mode of inheritance | Incidence |
| I | mild | Null COL1A1 allele | 166200 | autosomal dominant, 60% de novo[18] | 1 in 30.000[19] |
| II | severe and usually lethal in the perinatal period | COL1A1, COL1A2, | 166210 (IIA), 610854 (IIB) | autosomal dominant, ~100% de novo[18] | 1 in 40.000[20] to 1 in 100.000[19] |
| III | considered progressive and deforming | COL1A1, COL1A2 | 259420 | autosomal dominant, ~100% de novo[18] | 1 in 60.000[19] |
| IV | deforming, but with normal sclerae most of the time | COL1A1, COL1A2 | 166220 | autosomal dominant, 60% de novo[18] | |
| V | shares the same clinical features of IV, but has unique histologic findings ("mesh-like") | IFITM5 | 610967 | autosomal dominant[18][21] | |
| VI | shares the same clinical features of IV, but has unique histologic findings ("fish scale") | SERPINF1 | 610968 | autosomal recessive[18] | |
| VII | associated with cartilage associated protein | CRTAP | 610682 | autosomal recessive[18] | |
| VIII | severe to lethal, associated with the protein leprecan | LEPRE1, P3H1 | 610915 | autosomal recessive | |
| IX | PPIB | autosomal recessive |
Type I
Collagen is of normal quality but is produced in insufficient quantities.
- Bones fracture easily
- Slight spinal curvature
- Loose joints
- Poor muscle tone
- Discoloration of the sclera (whites of the eyes), usually giving them a blue-gray color. The blue-gray color of the sclera is due to the underlying choroidal veins which show through. This is due to the sclera being thinner than normal because the defective Type I collagen is not forming correctly.
- Early loss of hearing in some children[22]
- Slight protrusion of the eyes
IA and IB are defined to be distinguished by the absence/presence of dentinogenesis imperfecta (characterized by opalescent teeth; absent in IA, present in IB). Life expectancy is slightly reduced compared to the general population due to the possibility of fatal bone fractures and complications related to OI Type I such as basilar invagination.[citation needed]
Type II
Collagen is not of a sufficient quality or quantity.
- Most cases result in death within the first year of life due to respiratory failure or intracerebral hemorrhage
- Severe respiratory problems due to underdeveloped lungs
- Severe bone deformity and small stature
Type II can be further subclassified into groups A, B, and C, which are distinguished by radiographic evaluation of the long bones and ribs. Type IIA demonstrates broad and short long bones with broad and beaded ribs. Type IIB demonstrates broad and short long bones with thin ribs that have little or no beading. Type IIC demonstrates thin and longer long bones with thin and beaded ribs.
Type III
Collagen improperly formed, enough collagen is made but it is defective.
- Bones fracture easily, sometimes even before birth
- Bone deformity, often severe
- Respiratory problems possible
- Short stature, spinal curvature and sometimes barrel-shaped rib cage
- Triangular face[23]
- Loose joints (double-jointed)
- Poor muscle tone in arms and legs
- Discolouration of the sclera (the 'whites' of the eyes are blue)
- Early loss of hearing possible
Type III is distinguished among the other classifications as being the "progressive deforming" type, wherein a neonate presents with mild symptoms at birth and develops the aforementioned symptoms throughout life. Lifespans may be normal, albeit with severe physical handicapping.
Type IV
Collagen quantity is sufficient, but is not of a high enough quality
- Bones fracture easily, especially before puberty
- Short stature, spinal curvature, and barrel-shaped rib cage
- Bone deformity is mild to moderate
- Early loss of hearing
Similar to Type I, Type IV can be further subclassified into types IVA and IVB characterized by absence (IVA) or presence (IVB) of dentinogenesis imperfecta.
Type V

Having the same clinical features as Type IV, it is distinguished histologically by "mesh-like" bone appearance. Further characterized by the "V triad" consisting of (a) radio-opaque band adjacent to growth plates, (b) hypertrophic calluses at fracture sites, and (c) calcification of the radio-ulnar interosseous membrane.[24]
OI Type V leads to calcification of the membrane between the two forearm bones, making it difficult to turn the wrist. Another symptom is abnormally large amounts of repair tissue (hyperplasic callus) at the site of fractures. Other features of this condition include radial head dislocation, long bone bowing, and mixed hearing loss.
At least some cases of this type are caused by mutations in the IFITM5 gene.[21]
-
OI Type V in an adult
-
OI Type V in a child
Type VI
With the same clinical features as Type IV, it is distinguished histologically by "fish-scale" bone appearance. Type VI has recently been found to be caused by a loss of function mutation in the SERPINF1 gene. SERPINF1, a member of the serpin family, is also known as pigment epithelium-derived factor (PEDF), the most powerful endogenous antiangiogenic factor in mammals.
Type VII
In 2006, a recessive form called "Type VII" was discovered (phenotype severe to lethal). Thus far it seems to be limited to a First Nations people in Quebec.[25] Mutations in the gene CRTAP causes this type.[26]
Type VIII
OI caused by mutation in the gene LEPRE1 is classified as type VIII.[26]
Type XI
OI caused by mutations in FKBP10 on chromosome 17q21.[27] The mutations cause a decrease in secretion of trimeric procollagen molecules. These mutations can also cause autosomal recessive Bruck syndrome which is similar to OI.
Type XII
OI caused by a frameshift mutation in SP7. This mutation causes bone deformities, fractures, and delayed tooth eruption.[28]
Type XIII
OI caused by a mutation in the bone morphogenic protein 1 (BMP1) gene.[29][30] This mutation causes recurrent fractures, high bone mass, and hyperextensive joints.[31]
Type XIV
OI caused by mutations in the TMEM38B gene. This mutation causes recurrent fractures and osteopenia.
Type XV
OI caused by homozygous or compound heterozygous mutations in the WNT1 gene on chromosome 12q13. It is autosomal recessive.[32][31]
Type XVI
OI caused by mutations in the CREB3L1 gene. This mutation causes prenatal onset of recurrent fractures of the ribs and long bones, demineralization, decreased ossification of the skull, and blue sclerae. Family members who are heterozygous for OI XVI may have recurrent fractures, osteopenia and blue sclerae.[32]
Type XVII
OI caused by homozygous mutation in the SPARC gene on chromosome 5q33.
Type XVIII
OI caused by homozygous mutation in the FAM46A gene on chromosome 6q14. Characterized by congenital bowing of the long bones, wormian bones, blue sclerae, vertebral collapse, and multiple fractures in the first years of life.[33]
Others
A family with recessive osteogenesis imperfecta has been reported to have a mutation in the TMEM38B gene on chromosome 9.[34] This gene encodes TRIC-B, a component of TRIC, a monovalent cation-specific channel involved in calcium release from intracellular stores.
It is extremely likely that there are other genes associated with this disease that have yet to be reported.
Genetics
Osteogenesis imperfecta is a genetic disorder that causes increased bone fractures and collagen defects. The main causes for developing the disorder are a result of mutations in the COL1A1 and COL1A2 genes which are responsible for the production of collagen type 1.[35] Approximately 90% of people with OI are heterozygous for mutations in both the COL1A1 and COL1A2 genes.[36] There are several factors that are results of the dominant form of the OI disorder. These factors include; intracellular stress, abnormal tissue mineralization, abnormal cell to cell interactions, abnormal cell-matrix interactions, a compromised cell matrix structure, and disturbances between non-collagenous proteins and collagen.[37] Previous research lead to the belief that OI was an autosomal dominant disorder with few other variations in genomes.[38] However, In the past several years, there has been the identification of autosomal recessive forms of the disorder.[39] Recessive forms of OI relate heavily to defects in the collagen chaperones responsible for production of pro-collagen and the assembly of the related proteins.[40] Examples of collagen chaperones that are defective in OI patients include chaperone HSP47 (Cole-Carpenter syndrome) and FKBP65.[41] Mutations in these chaperones result in an improper folding pattern in the collagen 1 proteins which causes the recessive form of the disorder.[41] There are three significant types of OI that are a result of mutations in the collagen prolyl 3-hydroxylation complex (components CRTAP, P3H1, and CyPB).[41] These components are responsible for the modification of collagen a1(l)Pro986.[41] Mutations in other genes such as SP7, SERPINF1, TMEM38B and BMP1 can also lead to irregularly formed proteins and enzymes that result in the recessive form of Osteogenesis Imperfecta.[41] There are now links to defects in other proteins caused by genetic mutations ranging in function from structural proteins to enzymatic proteins.[38] A link between proteins such as pigment epithelium-derived factor (PEDF) and bone-restricted interferon-induced transmembrane protein (BRIL) are causes for type V and VI Osteogenesis Imperfecta.[42] Defects in these proteins lead to defective bone mineralization which aids in the formation of the brittle bone symptom of Osteogenesis Imperfecta.[42] Additionally, mutations in the COL1A1 and COL1A2 genes can result in signal disruptions of the extracellular matrix signaling that is present within the collagen proteins, causing worsened symptoms of the disorder.[43] A single point mutation in the untranslated 5' region of the IFITM5 gene was recently discovered and linked directly to OI type V.[44] Another single point mutation in the region that codes for collagen proteins on the IFITM5 gene was also found to be present in patients with substantially more severe versions of OI than just type V.[44] Osteogenesis imperfecta has also been seen as an X-linked related genetic disorder in some rare cases but continues to be a primarily heterozygous dominant disorder.[45]
Pathophysiology
People with OI are born with defective connective tissue, or without the ability to make it, usually because of a deficiency of Type-I collagen.[46] This deficiency arises from an amino acid substitution of glycine to bulkier amino acids in the collagen triple helix structure. The larger amino acid side-chains create steric hindrance that creates a bulge in the collagen complex, which in turn influences both the molecular nanomechanics and the interaction between molecules, which are both compromised.[47] As a result, the body may respond by hydrolyzing the improper collagen structure. If the body does not destroy the improper collagen, the relationship between the collagen fibrils and hydroxyapatite crystals to form bone is altered, causing brittleness.[48] Another suggested disease mechanism is that the stress state within collagen fibrils is altered at the locations of mutations, where locally larger shear forces lead to rapid failure of fibrils even at moderate loads as the homogeneous stress state found in healthy collagen fibrils is lost.[47] These recent works suggest that OI must be understood as a multi-scale phenomenon, which involves mechanisms at the genetic, nano-, micro- and macro-level of tissues. Most people with OI receive it from a parent but in 35% of cases it is an individual (de novo or "sporadic") mutation.[citation needed]
Diagnosis
Diagnosis is typically based on medical imaging, including plain X-rays, and symptoms. Signs on medical imaging include abnormalities in all extremeties and the spine.[49] An OI diagnosis can be confirmed through DNA or collagen testing, but in many cases, the occurrence of bone fractures with little trauma and the presence of other clinical features such as blue sclera are sufficient for a diagnosis. A skin biopsy can be performed to determine the structure and quantity of type I collagen. DNA testing can confirm the diagnosis, however, it cannot exclude it because not all mutations causing OI are known and/or tested for. OI type II is often diagnosed by ultrasound during pregnancy, where already multiple fractures and other characteristic features may be present. Relative to control, OI cortical bone shows increased porosity, canal diameter, and connectivity in micro-computed tomography.[50] Severe types of OI can usually be detected before birth by using an in-vitro genetic testing technique.[51]
Genetic testing

In order to determine whether osteogenesis imperfecta is present, genetic sequencing of the COL1A1, COL1A2, and IFITM5 genes may be done.[53][54]
Duplication and deletion testing is also suggested to parents who suspect their child has OI.[53]
The presence of frameshift mutations caused by duplications and deletions is generally the cause of increased severity within the disease.[53]
Differential diagnosis
An important differential diagnosis of OI is child abuse, as both may present with multiple fractures in various stages of healing. Differentiating them can be difficult, especially when no other characteristic features of OI are present. Other differential diagnoses include rickets, osteomalacia, and other rare skeletal syndromes.
Treatment
There is no cure.[4] Maintaining a healthy lifestyle by exercising and avoiding smoking can help prevent fractures. Treatment may include care of broken bones, pain medication, physical therapy, braces or wheelchairs, and surgery. A type of surgery that puts metal rods through long bones may be done to strengthen them.[5]
Bone infections are treated as and when they occur with the appropriate antibiotics and antiseptics.
Bisphosphonates
In 1998, a clinical trial demonstrated the effectiveness of intravenous pamidronate, a bisphosphonate which had previously been used in adults to treat osteoporosis. In severe OI, pamidronate reduced bone pain, prevented new vertebral fractures, reshaped previously fractured vertebral bodies, and reduced the number of long-bone fractures.[55]
Although oral bisphosphonates are more convenient and cheaper, they are not absorbed as well, and intravenous bisphosphonates are generally more effective, although this is under study. Some studies have found oral and intravenous bisphosphonates, such as oral alendronate and intravenous pamidronate, equivalent.[56] In a trial of children with mild OI, oral risedronate increased bone mineral densities, and reduced nonvertebral fractures. However, it did not decrease new vertebral fractures.[57][58] A Cochrane review in 2016 concluded that though bisphosphonates seem to improve bone mineral density, it is uncertain whether this leads to a reduction in fractures or an improvement in the quality of life of individuals with osteogenesis imperfecta.[10]
Bisphosphonates are less effective for OI in adults.[59]
Surgery
Metal rods can be surgically inserted in the long bones to improve strength, a procedure developed by Harold A. Sofield at Shriners Hospitals for Children in Chicago. During the late 1940s, Sofield, Chief of Staff at Shriners Hospitals in Chicago, worked there with large numbers of children with OI and experimented with various methods to strengthen the bones in these children.[60] In 1959, with Edward A. Miller, Sofield wrote a seminal article describing a solution that seemed radical at the time: the placement of stainless steel rods into the intramedullary canals of the long bones to stabilize and strengthen them. His treatment proved extremely useful in the rehabilitation and prevention of fractures; it was adopted throughout the world and still forms the basis for orthopedic treatment of OI.
Spinal fusion can be performed to correct scoliosis, although the inherent bone fragility makes this operation more complex in OI patients. Surgery for basilar impressions can be carried out if pressure being exerted on the spinal cord and brain stem is causing neurological problems.
Physiotherapy
Physiotherapy is used to strengthen muscles and improve motility in a gentle manner, while minimizing the risk of fracture. This often involves hydrotherapy, light resistance exercises, and the use of support cushions to improve posture. Individuals are encouraged to change positions regularly throughout the day to balance the muscles being used and the bones under pressure.
Exercise is generally recommended.[61]
Physical aids
With adaptive equipment such as crutches, powered wheelchairs, splints, grabbing arms, or modifications to the home, many individuals with OI can maintain a significant degree of autonomy.
Teeth
More than 1 in 2 people with OI also have dentinogenesis imperfecta (DI) - a congenital disorder of formation of dentine.[62] Dental treatment may pose as a challenge as a result of the various deformities, skeletal and dental, due to OI. Children with OI should go for a dental check-up as soon as their teeth erupt, this may minimize tooth structure loss as a result of abnormal dentine, and they should be monitored regularly to preserve their teeth and oral health.[62]
Many people with OI are treated with bisphosphonates, and there are several complications with dental procedures when a person is taking BP, namely bisphosphonate-related osteonecrosis of the jaw (BRONJ).
History
The condition, or types of it, has had various other names over the years and in different nations. Among some of the most common alternatives are Ekman-Lobstein syndrome, Vrolik syndrome, and the colloquial glass-bone disease. The name osteogenesis imperfecta dates to at least 1895[63] and has been the usual medical term in the 20th century to present. The current four type system began with Sillence in 1979.[64] An older system deemed less severe types "osteogenesis imperfecta tarda" while more severe forms were deemed "osteogenesis imperfecta congenita."[65] As this terminology did not differentiate well between the types, and all forms of osteogenesis are congenital, this naming convention has since fallen out of favour.
The condition has been found in an ancient Egyptian mummy from 1000 BC. The Norse king Ivar the Boneless may have had this condition, as well. The earliest studies of it began in 1788 with the Swede Olof Jakob Ekman. He described the condition in his doctoral thesis and mentioned cases of it going back to 1678. In 1831, Edmund Axmann described it in himself and two brothers. Jean Lobstein dealt with it in adults in 1833. Willem Vrolik did work on the condition in the 1850s. The idea that the adult and newborn forms were the same came in 1897 with Martin Benno Schmidt.[66]
Epidemiology
In the United States, the incidence of osteogenesis imperfecta is estimated to be one per 20,000 live births.[67] An estimated 20,000 to 50,000 people are affected by OI in the United States. The most common types are I - IV, while the rest are very rare.[68]
Frequency is approximately the same across groups, but for unknown reasons, the Shona and Ndebele of Zimbabwe seem to have a higher proportion of Type III to Type I than other groups.[69] A similar pattern was found in segments of the Nigerian and South African populations.[citation needed] In these varied cases, the total number of OIs of all four types was roughly the same as any other ethnicity.
Society and culture
The Brittle Bone Society is a UK charity that supports people with the condition.
Australia
The OI Society of Australia was foundation was founded in 1977. The aim is to offer information about the disease, support research, and to create awareness to the public about those with osteogenesis Imperfecta. The foundation holds a conference every two years to discuss educational events and support Wishbone Day.[70]
Canada
The Canadian Osteogenesis Imperfecta Society was established in 1983, it is an international non-profit organization that helps with assisted living with those affected by OI. They provide emotional support, foster and support Canadian medical research in the causes of OI for all types involved. This organization also keeps and up-to-date library of medical research and findings of this disease for the public.[71]
Other animals
In dogs, OI is an autosomal-recessive condition, meaning that dogs with two copies of the allele will be affected.[72] Beagles, Standard Wirehaired Dachshunds, Golden Retrievers, Poodles, Bedlington Terriers, Norwegian Elkhounds, and the Standard and Miniature Smooth haired Dachshund have all been known to be possible carriers of OI, as well as mice and some breeds of fish.[72] In Golden Retrievers, it is caused by a mutation in the COL1A1, and in Beagles, the COL1A2. A separate mutation in the SERPINH1 gene has been found to cause the condition in Dachshunds.[73] Many breed organizations and veterinarians offer OI tests to tell if a dog is a carrier of OI. Dogs who are heterozygous for OI should only be bred to non-carriers. Homozygous carriers should never be bred, unless it is to a non-carrier.[74]
Animal models of OI are critical to the diagnosis, treatment, and possible cure for their human counterparts. The experimental treatments and therapies used on animals play an important role in the successful treatment of OI in humans.[75] Although dogs, mice, fish, and humans are not genetically identical, some animal models have been officially recognized to represent the varying types of OI in humans. The research on animal treatment runs parallel to the success of human treatment of OI.[75]
References
- ↑ 1.00 1.01 1.02 1.03 1.04 1.05 1.06 1.07 1.08 1.09 1.10 1.11 1.12 1.13 1.14 1.15 "osteogenesis imperfecta". Genetics Home Reference. 11 October 2016. Archived from the original on 18 October 2016. Retrieved 15 October 2016.
- ↑ William, Berger (2006). Andrews' Diseases of the Skin: Clinical Dermatology (10th ed.). Saunders. p. 517. ISBN 978-0721629216.
- ↑ "Brittle Bone Disorder". 1996. Archived from the original on 8 August 2018. Retrieved 6 November 2018.
- ↑ 4.0 4.1 4.2 4.3 4.4 4.5 4.6 4.7 4.8 4.9 "Osteogenesis Imperfecta Overview". NIAMS. June 2015. Archived from the original on 18 October 2016. Retrieved 15 October 2016.
- ↑ 5.0 5.1 5.2 5.3 5.4 "What Is Osteogenesis Imperfecta? Fast Facts: An Easy-to-Read Series of Publications for the Public". NIAMS. November 2014. Archived from the original on 18 October 2016. Retrieved 15 October 2016.
- ↑ "Osteogenesis imperfecta". rarediseases.info.nih.gov. Archived from the original on 2021-03-19. Retrieved 2018-04-17.
- ↑ Grond-Ginsbach, C; Debette, S (March 2009). "The association of connective tissue disorders with cervical artery dissections". Current Molecular Medicine. 9 (2): 210–4. doi:10.2174/156652409787581547. PMID 19275629. Archived from the original on 2021-08-29. Retrieved 2019-12-01.
- ↑ McNeeley, MF; Dontchos, BN; Laflamme, MA; Hubka, M; Sadro, CT (December 2012). "Aortic dissection in osteogenesis imperfecta: case report and review of the literature". Emergency Radiology. 19 (6): 553–6. doi:10.1007/s10140-012-1044-1. PMID 22527359.
- ↑ Harrington, J; Sochett, E; Howard, A (December 2014). "Update on the evaluation and treatment of osteogenesis imperfecta". Pediatric Clinics of North America. 61 (6): 1243–57. doi:10.1016/j.pcl.2014.08.010. PMID 25439022.
- ↑ 10.0 10.1 Dwan, K; Phillipi, CA; Steiner, RD; Basel, D (19 October 2016). "Bisphosphonate therapy for osteogenesis imperfecta". The Cochrane Database of Systematic Reviews. 10: CD005088. doi:10.1002/14651858.CD005088.pub4. PMC 6611487. PMID 27760454.
- ↑ 11.0 11.1 Kelly, Evelyn B. (2012). Encyclopedia of Human Genetics and Disease. ABC-CLIO. p. 613. ISBN 9780313387135. Archived from the original on 2017-11-05.
- ↑ Vernick, David (2005-11-02). "OI Issues: Hearing Loss". Archived from the original on 2018-11-08. Retrieved 4 November 2018.
- ↑ Senn, A (2012). "Genetic Heterogeneity in Osteogenesis Imperfecta". Otology & Neurotology. 33 (9): 1562–1566. doi:10.1097/MAO.0b013e31826bf19b. PMC 3498599. PMID 22996160.
- ↑ Hoggard, N (2012-12-01). "Craniospinal Abnormalities and Neurologic Complications of Osteogenesis Imperfecta: Imaging Overview". Radiographics. 32 (7): 2101–12. doi:10.1148/rg.327125716. PMID 23150860.
- ↑ Violas, Philippe; Fassier, François; Hamdy, Reggie; Duhaime, Morris; Glorieux, Francis H. (September 2002). "Acetabular Protrusion in Osteogenesis Imperfecta". Journal of Pediatric Orthopaedics. 22 (5): 622. doi:10.1097/01241398-200209000-00010. ISSN 0271-6798.
- ↑ Lee, JH (September 1995). "Gastrointestinal Problems". The Journal of Bone and Joint Surgery. American Volume. 77 (9): 1352–6. doi:10.2106/00004623-199509000-00010. PMID 7673285.
- ↑ "Constipation and OI - Osteogenesis Imperfecta Foundation | OIF.org". www.oif.org. Archived from the original on 2015-07-27. Retrieved 2019-06-04.
- ↑ 18.0 18.1 18.2 18.3 18.4 18.5 18.6 Steiner RD, Pepin MG, Byers PH, Pagon RA, Bird TD, Dolan CR, Stephens K, Adam MP (January 28, 2005). "COL1A1/2 Osteogenesis Imperfecta". University of Washington, Seattle. PMID 20301472. Archived from the original on 18 January 2017. Retrieved 26 March 2012.
{{cite journal}}: Cite journal requires|journal=(help) - ↑ 19.0 19.1 19.2 van Dijk, F.S.; Cobben, J.M.; Kariminejad, A.; Maugeri, A.; Nikkels, P.G.J.; van Rijn, R.R.; Pals, G. (2011). "Osteogenesis Imperfecta: A Review with Clinical Examples". Molecular Syndromology. 2 (1): 1–20. doi:10.1159/000332228. ISSN 1661-8777. PMC 3343766. PMID 22570641.
- ↑ Surabhi Subramanian; Vibhu Krishnan Viswanathan. "Osteogenesis Imperfecta". StatPearls Publishing. Archived from the original on 2020-10-21. Retrieved 2019-07-30.
{{cite web}}: CS1 maint: multiple names: authors list (link) Last Update: February 3, 2019. - ↑ 21.0 21.1 Shapiro JR, Lietman C, Grover M, Lu JT, Nagamani SC, Dawson BC, Baldridge DM, Bainbridge MN, Cohn DH, Blazo M, Roberts TT, Brennen FS, Wu Y, Gibbs RA, Melvin P, Campeau PM, Lee BH (2013). "Phenotypic variability of osteogenesis imperfecta type V caused by an IFITM5 mutation". J. Bone Miner. Res. 28 (7): 1523–30. doi:10.1002/jbmr.1891. PMC 3688672. PMID 23408678.
- ↑ Fuller E, Lin V, Bell M, Bharatha A, Yeung R, Aviv RI, Symons SP (2011). "Case of the month #171: osteogenesis imperfecta of the temporal bone". Can Assoc Radiol J. 62 (4): 296–8. doi:10.1016/j.carj.2010.04.002. PMID 22018338.
- ↑ Page 771 Archived 2013-06-08 at the Wayback Machine in: Chen, Harold (2006). Atlas of genetic diagnosis and counseling. Totowa, NJ: Humana Press. ISBN 978-1-58829-681-8.
- ↑ Glorieux FH, Rauch F, Plotkin H, Ward L, Travers R, Roughley P, Lalic L, Glorieux DF, Fassier F, Bishop NJ (2000). "Type V osteogenesis imperfecta: A new form of brittle bone disease". J. Bone Miner. Res. 15 (9): 1650–1658. doi:10.1359/jbmr.2000.15.9.1650. PMID 10976985. Archived from the original on 2021-08-28. Retrieved 2019-12-01.
- ↑ "Recessive Form of OI Discovered by Foundation-funded Researcher" (PDF). Archived from the original (PDF) on 2007-08-12. Retrieved 2008-04-09.
- ↑ 26.0 26.1 Genetics Home Reference Archived 2008-12-19 at the Wayback Machine: Genetic Conditions > Osteogenesis imperfecta (Reviewed November 2007)
- ↑ "OMIM Entry - # 610968 - OSTEOGENESIS IMPERFECTA, TYPE XI; OI11". www.omim.org. Archived from the original on 2019-05-15. Retrieved 2018-11-11.
- ↑ Sam, JE; Dharmalingam, M (2017). "Osteogenesis Imperfecta". Indian Journal of Endocrinology and Metabolism. 21 (6): 903–908. doi:10.4103/ijem.IJEM_220_17. PMC 5729682. PMID 29285457.
- ↑ "OMIM Entry # 616229 - OSTEOGENESIS IMPERFECTA, TYPE XVI; OI16". www.omim.org. Archived from the original on 2019-07-30. Retrieved 2018-11-11.
- ↑ Shapiro, Jay R. (2014), "Clinical and Genetic Classification of Osteogenesis Imperfecta and Epidemiology", Osteogenesis Imperfecta, Elsevier, pp. 15–22, doi:10.1016/b978-0-12-397165-4.00002-2, ISBN 9780123971654
- ↑ 31.0 31.1 Sam, JustinEasow; Dharmalingam, Mala (2017-11-01). "Osteogenesis Imperfecta". Indian Journal of Endocrinology and Metabolism. 21 (6): 903–908. doi:10.4103/ijem.IJEM_220_17. PMC 5729682. PMID 29285457.
- ↑ 32.0 32.1 "Springer Reference", OMIM (TM), Springer-Verlag, 2011, doi:10.1007/springerreference_36173[dead link]
- ↑ "OMIM Entry - # 617952 - OSTEOGENESIS IMPERFECTA, TYPE XVIII; OI18". www.omim.org. Archived from the original on 2021-08-29. Retrieved 2020-08-07.
- ↑ Volodarsky M, Markus B, Cohen I, Staretz-Chacham O, Flusser H, Landau D, Shelef I, Langer Y, Birk OS (2013). "A deletion mutation in TMEM38B associated with autosomal recessive osteogenesis imperfecta". Hum Mutat. 34 (4): 582–6. doi:10.1002/humu.22274. PMID 23316006.
- ↑ Palomo T, Vilaça T, Lazareto-castro M (1 December 2017). "Osteogenesis imperfecta: diagnosis and treatment". Current Opinion in Endocrinology, Diabetes and Obesity. 24 (6): 381–388. doi:10.1097/MED.0000000000000367. PMID 28863000.
- ↑ Valadares FR, Carneiro TB, Santos PM, Oliveira AC, Zabel B (18 July 2014). "What is new in genetics and osteogenesis imperfecta classification?". J Pediatr (Rio J). 90 (6): 536–541. doi:10.1016/j.jped.2014.05.003. PMID 25046257.
- ↑ Forlino A, Cabral WA, Barnes AM, Marini JC (14 June 2011). "New perspectives on osteogenesis imperfecta". Nat Rev Endocrinol. 7 (9): 540–557. doi:10.1038/nrendo.2011.81. PMC 3443407. PMID 21670757.
- ↑ 38.0 38.1 Forlino A, Marini JC (16 April 2016). "Osteogenesis imperfecta". Lancet. 387 (10028): 1657–1671. doi:10.1016/S0140-6736(15)00728-X. PMID 26542481.
- ↑ Drögemüller C, Becker D, Brunner A, Haase B, Kircher P, Seeliger F, Fehr M, Baumann U, Lindblad-Toh K, Leeb T (2009). Barsh GS (ed.). "A Missense Mutation in the SERPINH1 Gene in Dachshunds with Osteogenesis Imperfecta". PLOS Genetics. 5 (7): e1000579. doi:10.1371/journal.pgen.1000579. PMC 2708911. PMID 19629171.
- ↑ Rohrbach M, Giunta C (12 July 2012). "Recessive osteogenesis imperfecta: clinical, radiological, and molecular findings". Am J Med Genet C Semin Med Genet. 160C(3) (3): 175–189. doi:10.1002/ajmg.c.31334. PMID 22791419.
- ↑ 41.0 41.1 41.2 41.3 41.4 Marini JC, Blissett AR (14 June 2014). "New genes in bone development: what's new in osteogenesis imperfecta". J Clin Endocrinol Metab. 98 (8): 3095–3103. doi:10.1210/jc.2013-1505. PMC 3733862. PMID 23771926.
- ↑ 42.0 42.1 Marini JC, Reich A, Smith SM (August 2014). "Osteogenesis imperfecta due to mutation in non-collagenous genes: lessons in the biology of bone formation". Current Opinion in Pediatrics. 26 (4): 500–507. doi:10.1097/MOP.0000000000000117. PMC 4183132. PMID 25007323.
- ↑ Lim J, Grafe L, Alexander S, Lee B (15 February 2017). "Genetic causes and mechanisms of Osteogenesis Imperfecta". Bone. 102: 40–49. doi:10.1016/j.bone.2017.02.004. PMC 5607741. PMID 28232077.
- ↑ 44.0 44.1 Hanagata N (2 June 2015). "IFITM5 mutations and osteogenesis imperfecta". J Bone Miner Metab. 34 (2): 123–131. doi:10.1007/s00774-015-0667-1. PMID 26031935.
- ↑ Marini JC, Forlino A, Bächinger HP, Bishop NJ, Byers PH, et al. (18 August 2018). "Osteogenesis imperfecta". Nat Rev Dis Primers. 3: 17052. doi:10.1038/nrdp.2017.52. PMID 28820180.
- ↑ Rauch F, Glorieux FH (2004). "Osteogenesis imperfecta". Lancet. 363 (9418): 1377–85. doi:10.1016/S0140-6736(04)16051-0. PMID 15110498.
- ↑ 47.0 47.1 Gautieri A, Uzel S, Vesentini S, Redaelli A, Buehler MJ (2009). "Molecular and mesoscale disease mechanisms of Osteogenesis Imperfecta". Biophysical Journal. 97 (3): 857–865. doi:10.1016/j.bpj.2009.04.059. PMC 2718154. PMID 19651044.
- ↑ "Osteogenesis Imperfecta Foundation: Fast Facts". Archived from the original on 2007-06-28. Retrieved 2007-07-05.
- ↑ EL-Sobky, TA; Shawky, RM; Sakr, HM; Elsayed, SM; Elsayed, NS; Ragheb, SG; Gamal, R (15 November 2017). "A systematized approach to radiographic assessment of commonly seen genetic bone diseases in children: A pictorial review". J Musculoskelet Surg Res. 1 (2): 25. doi:10.4103/jmsr.jmsr_28_17.
- ↑ Jameson, John R.; Albert, Carolyne I.; Busse, Bjoern; Smith, Peter A.; Harris, Gerald F. (March 29, 2013). "3D micron-scale imaging of the cortical bone canal network in human osteogenesis imperfecta (OI)". In Weaver, John B; Molthen, Robert C (eds.). Medical Imaging 2013: Biomedical Applications in Molecular, Structural, and Functional Imaging. Vol. 8672. International Society for Optics and Photonics. pp. 86721L. doi:10.1117/12.2007209.
- ↑ Westgren M, Götherström c (3 June 2015). "Stem cell transplantation before birth - a realistic option for treatment of osteogenesis imperfecta?". Prenat Diagn. 35 (9): 827–832. doi:10.1002/pd.4611. PMID 25962526.
- ↑ Zhytnik, Lidiia; Simm, Kadri; Salumets, Andres; Peters, Maire; Märtson, Aare; Maasalu, Katre (27 May 2020). "Reproductive options for families at risk of Osteogenesis Imperfecta: a review". Orphanet Journal of Rare Diseases. 15 (1): 128. doi:10.1186/s13023-020-01404-w. ISSN 1750-1172. Retrieved 15 October 2023.
- ↑ 53.0 53.1 53.2 Pepin MG, Byers PH (14 November 2015). "What every clinical geneticist should know about testing for osteogenesis imperfecta in suspected child abuse cases". Am J Med Genet C Semin Med Genet. 169 (4): 307–313. doi:10.1002/ajmg.c.31459. PMID 26566591.
- ↑ "Is Osteogenesis Imperfecta Inherited?". 4 April 2014. Archived from the original on 29 August 2021. Retrieved 7 November 2018.
- ↑ Glorieux FH, Bishop NJ, Plotkin H, Chabot G, Lanoue G, Travers R (1998). "Cyclic administration of pamidronate in children with severe osteogenesis imperfecta". N. Engl. J. Med. 339 (14): 947–52. doi:10.1056/NEJM199810013391402. PMID 9753709. Archived from the original on 2021-08-29. Retrieved 2019-12-01.Free full text
- ↑ DiMeglio LA, Peacock M (2006). "Two-year clinical trial of oral alendronate versus intravenous pamidronate in children with osteogenesis imperfecta". J. Bone Miner. Res. 21 (1): 132–40. doi:10.1359/JBMR.051006. PMID 16355282.
- ↑ Bishop Nick (2013). "Risedronate in children with osteogenesis imperfecta: a randomised, double-blind, placebo-controlled trial". Lancet. 382 (9902): 1424–1432. doi:10.1016/S0140-6736(13)61091-0. PMID 23927913. Archived from the original on 2013-10-31. Retrieved 2016-07-12.
- ↑ Ward Leanne M (2013). "Oral bisphosphonates for paediatric osteogenesis imperfecta?". Lancet. 382 (9902): 1388–1389. doi:10.1016/S0140-6736(13)61531-7. PMID 23927912. Archived from the original on 2013-11-02. Retrieved 2016-07-12.
- ↑ Chevrel G, Schott AM, Fontanges E, Charrin JE, Lina-Granade G, Duboeuf F, Garnero P, Arlot M, Raynal C, Meunier PJ (2006). "Effects of oral alendronate on BMD in adult patients with osteogenesis imperfecta: a 3-year randomized placebo-controlled trial". J. Bone Miner. Res. 21 (2): 300–6. doi:10.1359/JBMR.051015. PMID 16418786.
- ↑ "A Leader in the Treatment of Osteogensis Imperfecta (OI)". Archived from the original on 2007-09-28. Retrieved 2007-07-05.
{{cite web}}: CS1 maint: bot: original URL status unknown (link) - ↑ "Osteogenesis Imperfecta Foundation | OIF.org". www.oif.org. Archived from the original on 2018-11-11. Retrieved 2018-11-10.
- ↑ 62.0 62.1 Mina Biria; Fatemeh Mashhadi Abbas; Sedighe Mozaffar; Rahil Ahmadi (2012). "Dentinogenesis imperfecta associated with osteogenesis imperfecta". Dent Res J (Isfahan). 9 (4): 489–494. PMC 3491340. PMID 23162594.
- ↑ K. Buday, Beiträge zur Lehre von der Osteogenesis imperfecta (1895)
- ↑ Sillence DO, Senn A, Danks DM (1979). "Genetic heterogeneity in osteogenesis imperfecta". J. Med. Genet. 16 (2): 101–16. doi:10.1136/jmg.16.2.101. PMC 1012733. PMID 458828.
- ↑ "Osteogenesis Imperfecta Foundation: Glossary". Archived from the original on 2007-08-07. Retrieved 2007-07-05.
- ↑ synd/1743 at Who Named It?
- ↑ Genetics of Osteogenesis Imperfecta Archived 2010-12-30 at the Wayback Machine Author: Horacio Plotkin. Updated: Feb 29, 2016
- ↑ "Osteogenesis Imperfecta Panel". University of Nebraska Medical center. Archived from the original on 2019-07-30. Retrieved 2019-07-30.
- ↑ Viljoen D, Beighton P (1987). "Osteogenesis imperfecta type III: an ancient mutation in Africa?". Am. J. Med. Genet. 27 (4): 907–12. doi:10.1002/ajmg.1320270417. PMID 3425600.
- ↑ "The OI Society of Australia". 2006. Archived from the original on 3 September 2018. Retrieved 6 November 2018.
- ↑ Sandor, Max (2008). "Genetic and Rare Diseases Information Center (GARD)". Archived from the original on 8 November 2018. Retrieved 6 November 2018.
- ↑ 72.0 72.1 "Osteogenesis Imperfecta in Dogs - Symptoms, Causes, Diagnosis, Treatment, Recovery, Management, Cost". WagWalking. Archived from the original on 2018-11-08. Retrieved 2018-11-07.
- ↑ Eckardt J, Kluth S, Dierks C, Philipp U, Distl O (2013). "Population screening for the mutation associated with osteogenesis imperfecta in dachshunds". Vet. Rec. 172 (14): 364. doi:10.1136/vr.101122. PMID 23315765. Archived from the original on 2021-08-30. Retrieved 2019-12-01.
- ↑ "Osteogenesis Imperfecta - CAG - Center for Animal Genetics". CAG - Center for Animal Genetics. Archived from the original on 2018-11-08. Retrieved 2018-11-07.
- ↑ 75.0 75.1 Carriero, Alessandra; Enderli, Tanya; Burtch, Stephanie; Templet, Jara (September 2016). "Animal models of osteogenesis imperfecta: applications in clinical research". Orthopedic Research and Reviews. 8: 41–55. doi:10.2147/ORR.S85198. ISSN 1179-1462. PMC 6209373. PMID 30774469. Archived from the original on 2018-11-08. Retrieved 2018-11-07.
External links
| Classification | |
|---|---|
| External resources |
- GeneReview/NCBI/NIH/UW entry on Osteogenesis Imperfecta Archived 2017-01-18 at the Wayback Machine
- synd/1743 at Who Named It?
- Osteogenesis Imperfecta Overview Archived 2010-07-27 at the Wayback Machine NIH Osteoporosis and Related Bone Diseases ~ National Resource Center
- Pages with script errors
- CS1 errors: missing periodical
- CS1 maint: multiple names: authors list
- Webarchive template wayback links
- All articles with dead external links
- Articles with dead external links from December 2019
- Articles with invalid date parameter in template
- CS1: long volume value
- CS1 maint: bot: original URL status unknown
- All articles with unsourced statements
- Articles with unsourced statements from June 2019
- Articles with unsourced statements from June 2008
- Articles with unsourced statements from June 2011
- Articles with unsourced statements from April 2016
- Articles with hatnote templates targeting a nonexistent page
- Abnormalities of dermal fibrous and elastic tissue
- Skeletal disorders
- Collagen disease
- Rare diseases
- RTT