Mitral valve prolapse
| Mitral valve prolapse | |
|---|---|
| Other names: Floppy mitral valve syndrome, systolic click murmur syndrome, billowing mitral leaflet[1] | |
 | |
| Drawing of the leaflets of the mitral valve prolapsing back into the left atrium. | |
| Specialty | Cardiology |
| Symptoms | None, chest pain, shortness of breath, palpitations[1] |
| Complications | Mitral regurgitation, atrial fibrillation, infective endocarditis, stroke, cardiac arrest[1] |
| Usual onset | Often present at birth[2] |
| Duration | Lifelong |
| Types | Fibroelastic deficiency, Barlow's disease[3] |
| Causes | Unknown[2] |
| Risk factors | Ehlers-Danlos syndrome, Marfan syndrome, osteogenesis imperfecta[1] |
| Diagnostic method | Echocardiogram, auscultation[1] |
| Treatment | Watchful waiting, mitral valve repair or replacement[3] |
| Frequency | 2-3%[4] |
Mitral valve prolapse (MVP) is a valvular heart disease in which mitral valve leaflets enter into the left atrium when the ventricle contracts.[1] Symptoms vary from none, to chest pain, shortness of breath, palpitations, and anxiety.[1][3] Complications may include mitral regurgitation, atrial fibrillation, infective endocarditis, stroke, and cardiac arrest.[1]
The cause is unknown.[2] The underlying mechanism may involve myxomatous degeneration of the valve or as the result of other health problems such as Ehler-Danlos Syndrome, Marfan syndrome, and Graves' disease.[1] It is also associated with pectus excavatum, scoliosis, and family history of the condition.[3] Diagnosis is generally suspected based on hearing a mid-systolic click followed by a heart murmur and confirmed by ultrasound of the heart.[1][3] There are two types: classic with leaflet thickening; and non-classic without leaflet thickening.[3]
Most people require no treatment and have no significant problems during long term follow up.[2][3] In those with symptoms and severe mitral regurgitation, mitral valve repair or replacement may be an option.[3] In those without symptoms recommendations for surgery are based on ejection fraction less than 60%, left ventricular end-systolic dimension (LVESD) more than 4 or 4.5 cm, pulmonary artery pressure greater than 50 mmHg, or the presence of atrial fibrillation.[3]
Mitral valve prolapse affects about 2–3% of the population.[4] In 1966 the condition was described by John Brereton Barlow with its current name provided by J. Michael Criley.[5][6][7] The mid-systolic click followed by systolic murmur had been noticed early in 1887 by M. Cuffer and M. Barbillon.[8][9]
Signs and symptoms
Murmur
With auscultation, a mid-systolic click, followed by a late systolic murmur heard best at the apex, is common. The length of the murmur signifies the time period over which blood is leaking back into the left atrium, known as regurgitation. A murmur that lasts throughout the whole of systole is known as a holo-systolic murmur. A murmur that is mid to late systolic, although typically associated with less regurgitation, can still be associated with significant hemodynamic consequences.[10]
In contrast to most other heart murmurs, the murmur of mitral valve prolapse is accentuated by standing and Valsalva maneuver (earlier systolic click and longer murmur) and diminished with squatting (later systolic click and shorter murmur). The only other heart murmur that follows this pattern is the murmur of hypertrophic cardiomyopathy. An MVP murmur can be distinguished from a hypertrophic cardiomyopathy murmur by the presence of a mid-systolic click which is virtually diagnostic of MVP. The handgrip maneuver diminishes the murmur of an MVP and the murmur of hypertrophic cardiomyopathy. The handgrip maneuver also diminishes the duration of the murmur and delays the timing of the mid-systolic click.[11]
Both Valsalva maneuver and standing decrease venous return to the heart thereby decreasing left ventricular diastolic filling (preload) and causing more laxity on the chordae tendineae. This allows the mitral valve to prolapse earlier in systole, leading to an earlier systolic click (i.e. closer to S1), and a longer murmur.[12]
Mitral regurgitation

Mitral valve prolapse is frequently associated with mild mitral regurgitation,[13] where blood aberrantly flows from the left ventricle into the left atrium during systole. In the United States, MVP is the most common cause of severe, non-ischemic mitral regurgitation.[14] This is occasionally due to rupture of the chordae tendineae that support the mitral valve.[11]
The degree of MVP regurgitation severeness is usually estimated in grading system:[15][16][17]
- 0 (none or trivial)
- Grade 1 (mild)
- Grade 2 (moderate)
- Grade 3 (moderate to severe)
- Grade 4 (severe)
Arrhythmia
People with mitral valve prolapse might have arrhythmic mitral valve prolapse which includes higher incidence of ventricular contraction disorders and tachycardia compared to the normal population, although the relationship between both phenomena is not entirely clear.[18] Prolapse of both mitral leaflets and the presence of mitral regurgitation further increases the risk of severe ventricular arrhythmias during exertion, which may not be resolved with surgery. The most common rhythm disorder is ventricular extrasystole, followed by paroxysmal atrial tachycardia.[citation needed]
Sudden cardiac death
Severe mitral valve prolapse and moderate-to-severe mitral regurgitation and reduced left ventricular ejection fraction is associated with arrhythmias and atrial fibrillation that can progress to cardiac arrest and sudden cardiac death (SCD). Because there is no evidence that prolapse has contributed to these arrhythmias, these complications may be due to mitral regurgitation or congestive heart failure.[19] The incidence of life threatening arrhythmias in the general population with MVP remain low.[20] Sudden cardiac death results in 0.2% to 0.4% patients per year.[21]
Mitral valve prolapse syndrome
Historically, the term "mitral valve prolapse syndrome" has been applied to MVP associated with palpitations, atypical precordial pain, dyspnea on exertion, low body mass index, and electrocardiogram abnormalities (ventricular tachycardia), syncope, low blood pressure, headaches, lightheadedness, exercise intolerance, gastrointestinal disturbances, cold extremities and other signs suggestive of autonomic nervous system dysfunction (dysautonomia).[14][22][23]
Common risk factors for severe, arrhythmic mitral valve prolapse include:
- Electrocardiography - Inferior T wave inversions in II, III, aVF leads, ST-segment depression, QT dispersion, QT prolongation, complex ventricular ectopy (PVCs), PVC-triggered ventricular fibrillation[24][25][26]
- Echocardiography - ≥ moderate mitral regurgitation, leaflet thickening, bi-leaflet MVP with redundancy, spiked systolic high-velocity signal (pickelhaube spike) when ≥ 16 cm/s in mitral annular tissue Doppler imaging (TDI), mitral annular disjunction (MAD)[27][28][24][29]
- Cardiac MRI - myocardial/papillary scars (fibrosis), myocardial stretch, systolic curling[24][30]
Cause
Risk factors
MVP occurs with greater frequency in Ehlers-Danlos syndrome, Marfan syndrome[31] or polycystic kidney disease.[32] Other risk factors include Graves disease[33] and chest wall deformities such as pectus excavatum.[34] For unknown reasons, MVP patients tend to have a low body mass index (BMI) and are typically leaner than individuals without MVP.[35][36] Also women tend to have joint hypermobility.[37]
Rheumatic fever is common worldwide and responsible for many cases of damaged heart valves. Chronic rheumatic heart disease is characterized by repeated inflammation with fibrinous resolution. The cardinal anatomic changes of the valve include leaflet thickening, commissural fusion, and shortening and thickening of the tendinous cords.[38] The recurrence of rheumatic fever is relatively common in the absence of maintenance of low dose antibiotics, especially during the first three to five years after the first episode. Heart complications may be long-term and severe, particularly if valves are involved. Rheumatic fever, since the advent of routine penicillin administration for Strep throat, has become less common in developed countries. In the older generation and in much of the less-developed world, valvular disease (including mitral valve prolapse, reinfection in the form of valvular endocarditis, and valve rupture) from undertreated rheumatic fever continues to be a problem.[39]
In an Indian hospital between 2004 and 2005, 4 of 24 endocarditis patients failed to demonstrate classic vegetations. All had rheumatic heart disease (RHD) and presented with prolonged fever. All had severe eccentric mitral regurgitation (MR). (One had severe aortic regurgitation (AR) also.) One had flail posterior mitral leaflet (PML).[40]
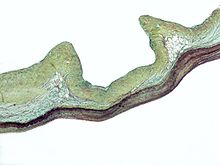
The mitral valve, so named because of its resemblance to a bishop's mitre, is the heart valve that prevents the backflow of blood from the left ventricle into the left atrium of the heart. It is composed of two leaflets, one anterior and one posterior, that close when the left ventricle contracts.[41]
Each leaflet is composed of three layers of tissue: the atrialis, fibrosa, and spongiosa. Patients with classic mitral valve prolapse have excess connective tissue that thickens the spongiosa and separates collagen bundles in the fibrosa. This is due to an excess of dermatan sulfate, a glycosaminoglycan. This weakens the leaflets and adjacent tissue, resulting in increased leaflet area and elongation of the chordae tendineae. Elongation of the chordae tendineae often causes rupture, commonly to the chordae attached to the posterior leaflet. Advanced lesions—also commonly involving the posterior leaflet—lead to leaflet folding, inversion, and displacement toward the left atrium.[35]
Genetics
Mitral valve prolapse is a genetically heterogeneous autosomal dominant trait, which can be passed down from one parent to child, who will have a 50% chance to inherit the mutated gene.
An association with primary cilia defects has been reported.[42] The mutations were found in the Zinc finger protein DZIP1 gene which regulates ciliogenesis; the same problem was found in mice who also developed MVP with this gene. It was found that primary cilia loss during development results in progressive myxomatous degeneration and profound mitral valve pathology.[43][44]
Myxomatous degeneration of mitral valve is a genetic abnormality mapped to gene Xq28.[45][46]
Other genes associated with MVP are:
- 13.q31.3-q32.1[45][47]
- 16p12.1-p11.2[45][48][49]
- Ch16p11.2-p12.1[49][50]
- 11p15.4[45][47]
- DCHS1[51][52]
Diagnosis
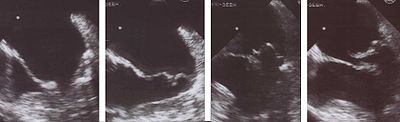
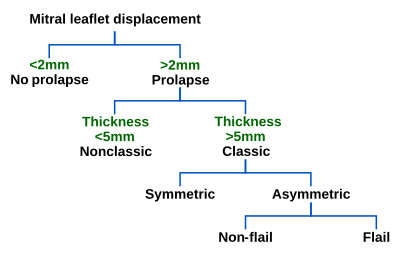
Echocardiography is the most useful method of diagnosing a prolapsed mitral valve. Two- and three-dimensional echocardiography is particularly valuable as they allow visualization of the mitral leaflets relative to the mitral annulus. This allows measurement of the leaflet thickness and their displacement relative to the annulus. Thickening of the mitral leaflets >5 mm and leaflet displacement >2 mm above the annular plane in parasternal long-axis view indicates classic mitral valve prolapse.[35]
Prolapsed mitral valves are classified into several subtypes, based on leaflet thickness, type of connection to the mitral annulus, and concavity. Subtypes can be described as classic, nonclassic, symmetric, asymmetric, flail, or non-flail.[35]
All measurements below refer to adult patients; applying them to children may be misleading.[citation needed]
Classic versus nonclassic
Prolapse occurs when the mitral valve leaflets are displaced more than 2 mm above the mitral annulus high points. The condition can be further divided into classic and nonclassic subtypes based on the thickness of the mitral valve leaflets: up to 5 mm is considered nonclassic, while anything beyond 5 mm is considered classic MVP.[35]
Symmetric versus asymmetric
Classical prolapse may be subdivided into symmetric and asymmetric, referring to the point at which leaflet tips join the mitral annulus. In symmetric coaptation, leaflet tips meet at a common point on the annulus. Asymmetric coaptation is marked by one leaflet displaced toward the atrium with respect to the other. Patients with asymmetric prolapse are susceptible to severe deterioration of the mitral valve, with the possible rupture of the chordae tendineae and the development of a flail leaflet.[35]
Flail versus non-flail

Asymmetric prolapse is further subdivided into flail and non-flail. Flail prolapse occurs when a leaflet tip turns outward, becoming concave toward the left atrium, causing the deterioration of the mitral valve. The severity of flail leaflet varies, ranging from tip eversion to chordal rupture. Dissociation of leaflet and chordae tendineae provides for unrestricted motion of the leaflet (hence "flail leaflet"). Thus patients with flail leaflets have a higher prevalence of mitral regurgitation than those with the non-flail subtype.[35]
Prevention
Mitral valve prolapse is a non-preventable condition, although some of its complications are. Because symptoms rarely appear, the productivity of person's life is not usually affected. The worsening of the disorder can be delayed by avoiding smoking, the use of hormonal birth control (because they have the risk of clotting) and regulating the amount and type of exercise and nutrition.[2] The risk of infective endocarditis is high with prosthetic heart valves, moderate in mitral prolapse concomitant with mitral regurgitation, and low in people with mitral prolapse without other valve disease.[53]
Treatment
Individuals with mitral valve prolapse, particularly those without symptoms, often require no treatment.[54] Those with mitral valve prolapse and symptoms of dysautonomia (palpitations, chest pain) may benefit from beta-blockers (e.g., propranolol, metoprolol, bisoprolol). People with prior stroke or atrial fibrillation may require blood thinners, such as aspirin or warfarin. In rare instances when mitral valve prolapse is associated with severe mitral regurgitation, surgical repair or replacement of the mitral valve may be necessary. Mitral valve repair is generally considered preferable to replacement. Current ACC/AHA guidelines promote repair of mitral valve in people before symptoms of heart failure develop. Symptomatic people, those with evidence of diminished left ventricular function, or those with left ventricular dilatation need urgent attention.[55]
Infective endocarditis
Individuals with MVP are at higher risk of bacterial infection of the heart, called infective endocarditis. This risk is approximately three-to eightfold the risk of infective endocarditis in the general population.[14] Until 2007, the American Heart Association recommended prescribing antibiotics before invasive procedures, including those in dental surgery. Thereafter, they concluded that "prophylaxis for dental procedures should be recommended only for patients with underlying cardiac conditions associated with the highest risk of adverse outcome from infective endocarditis."[56]
Many organisms responsible for endocarditis are slow-growing and may not be easily identified on routine blood cultures (these fastidious organisms require special culture media to grow). These include the HACEK organisms, which are part of the normal oropharyngeal flora and are responsible for perhaps 5 to 10% of infective endocarditis affecting native valves. It is important when considering endocarditis to keep these organisms in mind.[57]
Those with mitral prolapse are at increased risk of infective endocarditis, a bacterial infection of the heart tissue, as a result of certain routine non-sterile procedures, such as brushing the teeth. However, in April 2007, a study by the American Heart Association had determined that the risks of prescribing antibiotics outweigh the prophylactic antibiotics before invasive surgery, such as dental surgery or biopsy by colonoscopy or bronchoscopy.[58]
Prognosis
Generally, MVP is benign. However, those with a murmur, not just an isolated click, have an increased mortality rate of 15-20%.[59] The major predictors of mortality are the severity of mitral regurgitation and reduction in ejection fraction.[60]
Epidemiology
Prior to the strict criteria for the diagnosis of mitral valve prolapse, as described above, the incidence of mitral valve prolapse in the general population varied greatly.[35] Some studies estimated the incidence of mitral valve prolapse at 5 to 15 percent or even higher.[61] One 1985 study suggested MVP in up to 35% of healthy teenagers.[62]
Recent elucidation of mitral valve anatomy and the development of three-dimensional echocardiography have resulted in improved diagnostic criteria, and the true prevalence of MVP based on these criteria is estimated at 2-3%.[14] As a part of the Framingham Heart Study, for example, the prevalence of mitral valve prolapse in Framingham, MA was estimated at 2.4%. There was a near-even split between classic and nonclassic MVP, with no significant age or sex discrimination.[36] MVP is observed in 7% of autopsies in the United States.[59]
In a Taiwanese CHIEF heart study of Asian adult military personnel, it was estimated that out of 2442 people in Hualien aged 18 to 39, mitral valve prolapse occurred in 3.36%. People with MVP had lower body mass index, somatic symptoms related to exercise (chest pain, dyspnea, palpitations during exercise) and systolic click in auscultation. 7 out of 82 participants with MVP had mild pectus excavatum.[63]
History
The term "mitral valve prolapse" was coined by J. Michael Criley in 1966 and gained acceptance over the other descriptor of "billowing" of the mitral valve, as described by John Brereton Barlow.[6]
Research
In a human and mice study of MVP, a relationship was found between MVP and progressive fibrosis effects on left ventricular structure, which suggests the cause of molecular and cellular changes are a response of papillary and inferobasal myocardium to increased chordal tension from prolapsing mitral valve leaflets.[64]
In 2019 an experimental adeno-associated virus (AAV)-based gene therapy method developed by Rejuvenate Bio which was successfully and effectively used on mice that reversed multiple age-related diseases: heart failure, kidney failure, type 2 diabetes and obesity. Mice experienced a 58% increase in heart function and 75% reduction in kidney degeneration.[65] Rejuvenate Bio in collaboration with Tufts University later used the same method on Cavalier King Charles spaniel to stop the progression of mitral valve disease by stopping the accumulation of scar tissue in the heart.[66][67]
References
- ↑ 1.0 1.1 1.2 1.3 1.4 1.5 1.6 1.7 1.8 1.9 Shah, SN; Gangwani, MK; Oliver, TI (January 2022). "Mitral Valve Prolapse". PMID 29262039.
{{cite journal}}: Cite journal requires|journal=(help) - ↑ 2.0 2.1 2.2 2.3 2.4 "Mitral Valve Prolapse | NHLBI, NIH". www.nhlbi.nih.gov. Archived from the original on 2021-11-11. Retrieved 2021-11-11.
- ↑ 3.0 3.1 3.2 3.3 3.4 3.5 3.6 3.7 3.8 Althunayyan, Aeshah; Petersen, Steffen E; Lloyd, Guy; Bhattacharyya, Sanjeev (2 January 2019). "Mitral valve prolapse". Expert Review of Cardiovascular Therapy. 17 (1): 43–51. doi:10.1080/14779072.2019.1553619.
- ↑ 4.0 4.1 Basso, C; Iliceto, S; Thiene, G; Perazzolo Marra, M (10 September 2019). "Mitral Valve Prolapse, Ventricular Arrhythmias, and Sudden Death". Circulation. 140 (11): 952–964. doi:10.1161/CIRCULATIONAHA.118.034075. PMID 31498700.
- ↑ Orsinelli, David A. (17 April 2015). Color Atlas and Synopsis of Echocardiography. McGraw Hill Professional. p. 68. ISBN 978-0-07-182954-0. Archived from the original on 30 July 2022. Retrieved 29 July 2022.
- ↑ 6.0 6.1 Barlow JB, Bosman CK (February 1966). "Aneurysmal protrusion of the posterior leaflet of the mitral valve. An auscultatory-electrocardiographic syndrome". American Heart Journal. 71 (2): 166–178. doi:10.1016/0002-8703(66)90179-7. PMID 4159172.
- ↑ Criley JM, Lewis KB, Humphries JO, Ross RS (July 1966). "Prolapse of the mitral valve: clinical and cine-angiocardiographic findings". British Heart Journal. 28 (4): 488–496. doi:10.1136/hrt.28.4.488. PMC 459076. PMID 5942469.
- ↑ Grau JB, Pirelli L, Yu PJ, Galloway AC, Ostrer H (October 2007). "The genetics of mitral valve prolapse". Clinical Genetics. 72 (4): 288–295. doi:10.1111/j.1399-0004.2007.00865.x. PMID 17850623. S2CID 40874345.
- ↑ Anyanwu AC, Adams DH (2007). "Etiologic classification of degenerative mitral valve disease: Barlow's disease and fibroelastic deficiency". Seminars in Thoracic and Cardiovascular Surgery. 19 (2): 90–96. doi:10.1053/j.semtcvs.2007.04.002. PMID 17870001.
- ↑ Ahmed MI, Sanagala T, Denney T, Inusah S, McGiffin D, Knowlan D, et al. (August 2009). "Mitral valve prolapse with a late-systolic regurgitant murmur may be associated with significant hemodynamic consequences". The American Journal of the Medical Sciences. 338 (2): 113–115. doi:10.1097/MAJ.0b013e31819d5ec6. PMID 19561453. S2CID 44385990.
- ↑ 11.0 11.1 Armstrong, Guy P. (Aug 2021). "Mitral Valve Prolapse (MVP) - Cardiovascular Disorders". Merck Manuals Professional Edition. Archived from the original on 2021-11-21. Retrieved 2021-11-21.
- ↑ "Aortic Regurgitation". The Lecturio Medical Concept Library. Archived from the original on 29 June 2021. Retrieved 30 June 2021.
- ↑ Kolibash AJ (October 1988). "Progression of mitral regurgitation in patients with mitral valve prolapse". Herz. 13 (5): 309–317. PMID 3053383.
- ↑ 14.0 14.1 14.2 14.3 Hayek E, Gring CN, Griffin BP (2005). "Mitral valve prolapse". Lancet. 365 (9458): 507–518. doi:10.1016/S0140-6736(05)17869-6. PMID 15705461.
- ↑ "Mitral regurgitation - WikEM". wikem.org. Archived from the original on 2021-11-07. Retrieved 2021-11-07.
- ↑ Apostolakis EE, Baikoussis NG (July 2009). "Methods of estimation of mitral valve regurgitation for the cardiac surgeon". Journal of Cardiothoracic Surgery. 4: 34. doi:10.1186/1749-8090-4-34. PMC 2723095. PMID 19604402.
- ↑ Chew PG, Bounford K, Plein S, Schlosshan D, Greenwood JP (April 2018). "Multimodality imaging for the quantitative assessment of mitral regurgitation". Quantitative Imaging in Medicine and Surgery. 8 (3): 342–359. doi:10.21037/qims.2018.04.01. PMC 5941213. PMID 29774187.
- ↑ Basso C, Perazzolo Marra M, Rizzo S, De Lazzari M, Giorgi B, Cipriani A, et al. (August 2015). "Arrhythmic Mitral Valve Prolapse and Sudden Cardiac Death". Circulation. 132 (7): 556–566. doi:10.1161/CIRCULATIONAHA.115.016291. PMID 26160859.
- ↑ Hourdain J, Clavel MA, Deharo JC, Asirvatham S, Avierinos JF, Habib G, et al. (September 2018). "Common Phenotype in Patients With Mitral Valve Prolapse Who Experienced Sudden Cardiac Death" (PDF). Circulation. 138 (10): 1067–1069. doi:10.1161/CIRCULATIONAHA.118.033488. PMID 30354542. S2CID 53022372. Archived (PDF) from the original on 2022-03-02. Retrieved 2022-05-17.
- ↑ Nalliah CJ, Mahajan R, Elliott AD, Haqqani H, Lau DH, Vohra JK, et al. (January 2019). "Mitral valve prolapse and sudden cardiac death: a systematic review and meta-analysis". Heart. 105 (2): 144–151. doi:10.1136/heartjnl-2017-312932. PMID 30242141. S2CID 52313443.
- ↑ Basso C, Iliceto S, Thiene G, Perazzolo Marra M (September 2019). "Mitral Valve Prolapse, Ventricular Arrhythmias, and Sudden Death". Circulation. 140 (11): 952–964. doi:10.1161/CIRCULATIONAHA.118.034075. PMID 31498700. S2CID 202405456.
- ↑ Watkins PC (July 1997). "Treatment of Symptomatic Mitral Valve Prolapse Syndrome and Dysautonomia". Cardiology in Review. 5 (4): 208–212. doi:10.1097/00045415-199707000-00011. ISSN 1061-5377. S2CID 71861832. Archived from the original on 2021-09-26. Retrieved 2022-05-17.
- ↑ Styres KS (January 1994). "The phenomenon of dysautonomia and mitral valve prolapse". Journal of the American Academy of Nurse Practitioners. 6 (1): 11–15. doi:10.1111/j.1745-7599.1994.tb00888.x. PMID 8003350. S2CID 31933859.
- ↑ 24.0 24.1 24.2 Chu E (16 December 2020). "Mitral Valve Prolapse Syndrome: Once Benign and Now Malignant". American College of Cardiology. Archived from the original on 2021-10-16. Retrieved 2021-10-15.
- ↑ Muthukumar L, Jahangir A, Jan MF, Perez Moreno AC, Khandheria BK, Tajik AJ (September 2020). "Association Between Malignant Mitral Valve Prolapse and Sudden Cardiac Death: A Review". JAMA Cardiology. 5 (9): 1053–1061. doi:10.1001/jamacardio.2020.1412. PMID 32936277. S2CID 218910727.
- ↑ Essayagh B, Sabbag A, Antoine C, Benfari G, Yang LT, Maalouf J, et al. (August 2020). "Presentation and Outcome of Arrhythmic Mitral Valve Prolapse". Journal of the American College of Cardiology. 76 (6): 637–649. doi:10.1016/j.jacc.2020.06.029. PMID 32762897. S2CID 221076345.
- ↑ Widmer F (2019-11-14). "Arrhythmic mitral valve prolapse". Cardiovascular Medicine (6). doi:10.4414/cvm.2019.02075. S2CID 208973897. Archived from the original on 2022-03-31. Retrieved 2022-05-17.
- ↑ Ignatowski D, Schweitzer M, Pesek K, Jain R, Muthukumar L, Khandheria BK, Tajik AJ (May 2020). "Pickelhaube Spike, a High-Risk Marker for Bileaflet Myxomatous Mitral Valve Prolapse: Sonographer's Quest for the Highest Spike". Journal of the American Society of Echocardiography. 33 (5): 639–640. doi:10.1016/j.echo.2020.02.004. PMID 32199779. S2CID 214617051.
- ↑ Coutsoumbas GV, Di Pasquale G (October 2021). "Mitral valve prolapse with ventricular arrhythmias: does it carries a worse prognosis?". European Heart Journal Supplements. 23 (Suppl E): E77–E82. doi:10.1093/eurheartj/suab096. PMC 8503385. PMID 34650360.
- ↑ Han HC, Ha FJ, Teh AW, Calafiore P, Jones EF, Johns J, et al. (December 2018). "Mitral Valve Prolapse and Sudden Cardiac Death: A Systematic Review". Journal of the American Heart Association. 7 (23): e010584. doi:10.1161/JAHA.118.010584. PMC 6405538. PMID 30486705.
- ↑ "Related Disorders: Mitral Valve Prolapse". National Marfan Foundation. Archived from the original on 2007-02-25. Retrieved 2007-07-11.
- ↑ Lumiaho A, Ikäheimo R, Miettinen R, Niemitukia L, Laitinen T, Rantala A, et al. (December 2001). "Mitral valve prolapse and mitral regurgitation are common in patients with polycystic kidney disease type 1". American Journal of Kidney Diseases. 38 (6): 1208–1216. doi:10.1053/ajkd.2001.29216. PMID 11728952.
- ↑ "Graves Disease". National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK). U.S. Department of Health and Human Services. August 10, 2012. Archived from the original on 30 June 2021. Retrieved 2015-04-02.
- ↑ Hebra A (30 October 2018). Windle ML, Sharma GD (eds.). "Pectus Excavatum: Epidemiology". Medscape. Archived from the original on 2 April 2019. Retrieved 14 April 2016.
- ↑ 35.0 35.1 35.2 35.3 35.4 35.5 35.6 35.7 Playford D, Weyman AE (2001). "Mitral valve prolapse: time for a fresh look". Reviews in Cardiovascular Medicine. 2 (2): 73–81. PMID 12439384. Archived from the original on 2014-09-03. Retrieved 2009-03-24.
- ↑ 36.0 36.1 Freed LA, Levy D, Levine RA, Larson MG, Evans JC, Fuller DL, et al. (July 1999). "Prevalence and clinical outcome of mitral-valve prolapse". The New England Journal of Medicine. 341 (1): 1–7. doi:10.1056/NEJM199907013410101. PMID 10387935.
- ↑ Araújo CG, Chaves CP (October 2005). "Adult women with mitral valve prolapse are more flexible". British Journal of Sports Medicine. 39 (10): 720–724. doi:10.1136/bjsm.2004.014324. PMC 1725042. PMID 16183767.
- ↑ Cotran RS, Kumar V, Fausto N, Robbins SL, Abbas AK (2005). Robbins and Cotran pathologic basis of disease. St. Louis, Mo: Elsevier Saunders. ISBN 978-0-7216-0187-8. Archived from the original on 10 September 2005.
- ↑ "NLM/NIH: Medline Plus Medical Encyclopedia: Rheumatic fever". Archived from the original on 2016-04-07. Retrieved 2022-05-17.
- ↑ Venkatesan S (Sep–Oct 2007). "Can we diagnose Infective endocarditis without vegetation?". Indian Heart Journal. 59 (5). Archived from the original on 2015-07-14. Retrieved 2022-05-17.
- ↑ Standring S (2016). Gray's anatomy : the anatomical basis of clinical practice (Forty-first ed.). [Philadelphia]. ISBN 9780702052309. OCLC 920806541.
- ↑ Toomer KA, Yu M, Fulmer D, Guo L, Moore KS, Moore R, et al. (May 2019). "Primary cilia defects causing mitral valve prolapse". Science Translational Medicine. 11 (493). doi:10.1126/scitranslmed.aax0290. PMC 7331025. PMID 31118289.
- ↑ Toomer KA, Yu M, Fulmer D, Guo L, Moore KS, Moore R, et al. (May 2019). "Primary cilia defects causing mitral valve prolapse". Science Translational Medicine. 11 (493). doi:10.1126/scitranslmed.aax0290. PMC 7331025. PMID 31118289.
- ↑ Norris, Russell. "Genetic trigger discovered for common heart problem, mitral valve prolapse". The Conversation. Archived from the original on 2021-11-14. Retrieved 2021-11-14.
- ↑ 45.0 45.1 45.2 45.3 Delling FN, Vasan RS (May 2014). "Epidemiology and pathophysiology of mitral valve prolapse: new insights into disease progression, genetics, and molecular basis". Circulation. 129 (21): 2158–2170. doi:10.1161/CIRCULATIONAHA.113.006702. PMC 4052751. PMID 24867995.
- ↑ Trochu JN, Kyndt F, Schott JJ, Gueffet JP, Probst V, Bénichou B, Le Marec H (June 2000). "Clinical characteristics of a familial inherited myxomatous valvular dystrophy mapped to Xq28". Journal of the American College of Cardiology. 35 (7): 1890–1897. doi:10.1016/S0735-1097(00)00617-3. PMID 10841240.
- ↑ 47.0 47.1 Nesta F, Leyne M, Yosefy C, Simpson C, Dai D, Marshall JE, et al. (September 2005). "New locus for autosomal dominant mitral valve prolapse on chromosome 13: clinical insights from genetic studies". Circulation. 112 (13): 2022–2030. doi:10.1161/CIRCULATIONAHA.104.516930. PMID 16172273. S2CID 3177975.
- ↑ "OMIM Entry - % 157700 - MITRAL VALVE PROLAPSE 1; MVP1". omim.org. Archived from the original on 2021-01-28. Retrieved 2021-11-07.
- ↑ 49.0 49.1 Gasser S, Reichenspurner H, Girdauskas E (February 2018). "Genomic analysis in patients with myxomatous mitral valve prolapse: current state of knowledge". BMC Cardiovascular Disorders. 18 (1): 41. doi:10.1186/s12872-018-0755-y. PMC 5830049. PMID 29486707.
- ↑ Disse S, Abergel E, Berrebi A, Houot AM, Le Heuzey JY, Diebold B, et al. (November 1999). "Mapping of a first locus for autosomal dominant myxomatous mitral-valve prolapse to chromosome 16p11.2-p12.1". American Journal of Human Genetics. 65 (5): 1242–1251. doi:10.1086/302624. PMC 1288276. PMID 10521289.
- ↑ Durst, Ronen; Sauls, Kimberly; Peal, David S; deVlaming, Annemarieke; Toomer, Katelynn; Leyne, Maire; Salani, Monica; Talkowski, Michael E.; Brand, Harrison; Perrocheau, Maëlle; Simpson, Charles (2015-09-03). "Mutations in DCHS1 Cause Mitral Valve Prolapse". Nature. 525 (7567): 109–113. Bibcode:2015Natur.525..109D. doi:10.1038/nature14670. ISSN 0028-0836. PMC 4720389. PMID 26258302.
- ↑ Moore, Reece; Moore, Kelsey; Stairley, Rebecca; Fulmer, Diana B; Guo, Lilong; Norris, Russell A (2020-07-31). "Abstract MP173: Loss of DCHS1 Promotes Mitral Valve Prolapse Through Cytoskeleton Destabilization". Circulation Research. 127 (Suppl_1): AMP173. doi:10.1161/res.127.suppl_1.MP173. S2CID 229072540. Archived from the original on 2021-11-20. Retrieved 2022-05-17.
- ↑ Urriola B., Patricia (March 2000). "Pericarditis y endocarditis infecciosa". Revista chilena de pediatría. 71 (2): 142–146. doi:10.4067/S0370-41062000000200011. ISSN 0370-4106.
{{cite journal}}: CS1 maint: url-status (link) - ↑ "Mitral valve prolapse". Mayo Clinic. Archived from the original on 3 February 2008.
- ↑ "Mitral Regurgitation". The Lecturio Medical Concept Library. Archived from the original on 11 August 2021. Retrieved 11 August 2021.
- ↑ Wilson W, Taubert KA, Gewitz M, Lockhart PB, Baddour LM, Levison M, et al. (June 2007). "Prevention of infective endocarditis: guidelines from the American Heart Association: a guideline from the American Heart Association Rheumatic Fever, Endocarditis and Kawasaki Disease Committee, Council on Cardiovascular Disease in the Young, and the Council on Clinical Cardiology, Council on Cardiovascular Surgery and Anesthesia, and the Quality of Care and Outcomes Research Interdisciplinary Working Group". Journal of the American Dental Association. 138 (6): 739–45, 747–60. doi:10.14219/jada.archive.2007.0262. PMID 17545263.
- ↑ "Mitral Valve Prolapse". The Lecturio Medical Concept Library. Archived from the original on 11 July 2021. Retrieved 18 July 2021.
- ↑ Wilson, Walter; Taubert, Kathryn A.; Gewitz, Michael; Lockhart, Peter B.; Baddour, Larry M.; Levison, Matthew; Bolger, Ann; Cabell, Christopher H.; Takahashi, Masato; Baltimore, Robert S.; Newburger, Jane W. (June 2007). "Prevention of infective endocarditis: guidelines from the American Heart Association: a guideline from the American Heart Association Rheumatic Fever, Endocarditis and Kawasaki Disease Committee, Council on Cardiovascular Disease in the Young, and the Council on Clinical Cardiology, Council on Cardiovascular Surgery and Anesthesia, and the Quality of Care and Outcomes Research Interdisciplinary Working Group". Journal of the American Dental Association. 138 (6): 739–745, 747–760. doi:10.14219/jada.archive.2007.0262. ISSN 0002-8177. PMID 17545263. Archived from the original on 2022-07-30. Retrieved 2022-05-17.
- ↑ 59.0 59.1 Mitral Valve Prolapse at eMedicine
- ↑ "Mitral Valve Prolapse". The Lecturio Medical Concept Library. Archived from the original on 9 July 2021. Retrieved 3 July 2021.
- ↑ Levy D, Savage D (May 1987). "Prevalence and clinical features of mitral valve prolapse". American Heart Journal. 113 (5): 1281–1290. doi:10.1016/0002-8703(87)90956-2. PMID 3554946.
- ↑ Warth DC, King ME, Cohen JM, Tesoriero VL, Marcus E, Weyman AE (May 1985). "Prevalence of mitral valve prolapse in normal children". Journal of the American College of Cardiology. 5 (5): 1173–1177. doi:10.1016/S0735-1097(85)80021-8. PMID 3989128.
- ↑ Liu, Pang-Yen; Tsai, Kun-Zhe; Lin, Yen-Po; Lin, Chin-Sheng; Zeng, Huan-Chang; Takimoto, Eiki; Lin, Gen-Min (2021-02-01). "Prevalence and characteristics of mitral valve prolapse in military young adults in Taiwan of the CHIEF Heart Study". Scientific Reports. 11 (1): 2719. Bibcode:2021NatSR..11.2719L. doi:10.1038/s41598-021-81648-z. ISSN 2045-2322. PMC 7851121. PMID 33526804.
- ↑ Morningstar, Jordan E.; Gensemer, Cortney; Moore, Reece; Fulmer, Diana; Beck, Tyler C.; Wang, Christina; Moore, Kelsey; Guo, Lilong; Sieg, Franz; Nagata, Yasufumi; Bertrand, Philippe (2021). "Mitral Valve Prolapse Induces Regionalized Myocardial Fibrosis". Journal of the American Heart Association. 10 (24): e022332. doi:10.1161/JAHA.121.022332. PMID 34873924. S2CID 244922478. Archived from the original on 2022-03-21. Retrieved 2022-05-17.
- ↑ Davidsohn, Noah; Pezone, Matthew; Vernet, Andyna; Graveline, Amanda; Oliver, Daniel; Slomovic, Shimyn; Punthambaker, Sukanya; Sun, Xiaoming; Liao, Ronglih; Bonventre, Joseph V.; Church, George M. (2019-11-19). "A single combination gene therapy treats multiple age-related diseases". Proceedings of the National Academy of Sciences. 116 (47): 23505–23511. doi:10.1073/pnas.1910073116. ISSN 0027-8424. PMC 6876218. PMID 31685628.
- ↑ "A New Approach to Gene Therapy—Now In Dogs, Maybe Later In Humans - WSJ". wsj.com. 2019-11-06. Archived from the original on 2019-11-06. Retrieved 2021-11-26.
- ↑ "Rejuvenate Bio launches to help dogs live longer, healthier lives". Harvard Office of Technology Development. Archived from the original on 2021-11-26. Retrieved 2021-11-26.
External links
| Classification | |
|---|---|
| External resources |
- Pages with script errors
- CS1 errors: missing periodical
- CS1 maint: url-status
- Articles with hAudio microformats
- All articles with unsourced statements
- Articles with unsourced statements from November 2021
- Articles with invalid date parameter in template
- Articles with unsourced statements from February 2021
- Articles with Curlie links
- Valvular heart disease
- Syndromes
- RTT
