Miglustat
 | |
| Names | |
|---|---|
| Trade names | Zavesca, Brazaves |
| Other names | OGT 918, 1,5-(butylimino)-1,5-dideoxy-D-glucitol, N-butyl-deoxynojirimycin |
| |
| Clinical data | |
| Drug class | Glucosylceramide synthase inhibitor[1] |
| Main uses | Gaucher disease type 1 (GD1), Niemann–Pick disease type C (NPC)[2] |
| Side effects | Weight loss, shaking, diarrhea, increased intestinal gas, abdominal pain[2] |
| Pregnancy category | |
| Routes of use | By mouth |
| Typical dose | 100 mg TID[1] |
| External links | |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a604015 |
| Legal | |
| License data | |
| Legal status | |
| Pharmacokinetics | |
| Bioavailability | 97% |
| Protein binding | Nil |
| Metabolism | Nil |
| Elimination half-life | 6–7 hours |
| Excretion | Kidney, unchanged |
| Chemical and physical data | |
| Formula | C10H21NO4 |
| Molar mass | 219.281 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Miglustat, sold under the brand name Zavesca, is a medication used to treat Gaucher disease type 1 (GD1) and Niemann–Pick disease type C (NPC).[2] It is taken by mouth.[1]
Common side effects include weight loss, shaking, diarrhea, increased intestinal gas, and abdominal pain.[2] Other side effects may include peripheral neuropathy and low platelets.[1] Safety in pregnancy is unclear.[1] It is a glucosylceramide synthase inhibitor.[1]
Miglustat was approved for medical use Europe in 2002 and the United States in 2003.[2][1] It is available as a generic medication.[6] In the United Kingdom 4 weeks of medication costs the NHS about £3,400 as of 2021.[6] This amount in the United States costs about 23,000 USD.[7]
Medical uses
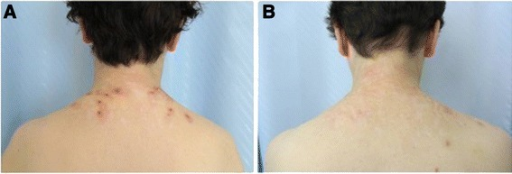
Miglustat is used to treat adults with mild to moderate type I Gaucher disease for whom enzyme replacement therapy is unsuitable.[8]
Dosage
It is typically taken at a dose of 100 mg three times per day; though 100 mg once to twice a day may be used if side effects are too great.[1]
Contraindications
Miglustat is contraindicated for people with neurological conditions, kidney problems, women who are pregnant, and men and women planning to conceive a child.[9]
Side effects
Serious side effects include pain, burning, numbness or tingling in the hands, arms, legs, or feet; shaking hands that cannot be controlled; changes in vision; and easy bruising or bleeding. Common side effects include gastrointestinal effects (including diarrhea, stomach pain or bloating, gas, loss of appetite, weight loss, upset stomach, vomiting, constipation), dry mouth, muscular effects (including weakness, muscle cramps, especially in the legs, feeling of heaviness in the arms or legs, unsteadiness when walking), back pain, dizziness, nervousness, headache, memory problems, and difficult or irregular menstruation (period).[9]
Mechanism of action
Type I Gaucher's disease is an autosomal recessive disorder; parents are generally healthy carriers with one functional and one mutated (nonfunctioning) copy of the Gaucher disease gene, GBA. People with type I Gaucher have a defect in the enzyme called glucocerebrosidase (also known as acid β-glucosidase). Glucocerebrosidase is an enzyme, and its function is to convert glucocerebroside (also known as glucosylceramide) into ceramide and glucose. When this enzyme doesn't work, glucocerebroside accumulates, which in turn causes liver and spleen enlargement, changes in the bone marrow and blood, and bone disease. It functions as a competitive and reversible inhibitor of the enzyme glucosylceramide synthase, the initial enzyme in a series of reactions which results in the synthesis of most glycosphingolipids.[10][11]
Earlier treatments on the market (imiglucerase (approved in 1995),[12] velaglucerase (approved in 2010),[13] taliglucerase alfa (Elelyso) (approved in 2012)[14]) are enzyme replacement therapy - they are functioning versions of the enzyme that doesn't work. Miglustat works differently - it prevents the formation of the substance that builds up when the enzyme doesn't work; this is called substrate reduction therapy.[15]
Chemistry
Miglustat is an iminosugar, a synthetic analogue of D-glucose[16] and a white to off-white crystalline solid that has a bitter taste.[17]
Society and culture
Legal status
Miglustat has been approved in the EU, Canada, and Japan for treating progressive neurological complications in people with Niemann–Pick disease, type C (NPC). Miglustat has not received approval for this indication outside of these countries and use for this disease, including the US, is off-label.[18][19][20][21][22]
Research
In July 2004, Actelion started a clinical trial of miglustat to treat Tay–Sachs disease, particularly late-onset Tay–Sachs with an estimated enrollment of 10 subjects; the trial ended August 2007.[23]
In November 2007, Actelion initiated a clinical trial with miglustat in people with cystic fibrosis (CF) who have the ΔF508 in both copies of the cystic fibrosis transmembrane conductance regulator (CFTR) gene; the study ended in March 2008.[24] The cystic fibrosis trial showed no effect.[25]
See also
- Migalastat, a drug for the treatment of Fabry disease, with a similar structure
- Miglitol, an oral antidiabetic drug with a similar structure
References
- ↑ 1.0 1.1 1.2 1.3 1.4 1.5 1.6 1.7 "Miglustat Monograph for Professionals". Drugs.com. Archived from the original on 4 March 2021. Retrieved 18 November 2021.
- ↑ 2.0 2.1 2.2 2.3 2.4 2.5 "Zavesca EPAR". European Medicines Agency (EMA). Archived from the original on 5 August 2020. Retrieved 7 August 2020.
- ↑ 3.0 3.1 "Miglustat (Zavesca) Use During Pregnancy". Drugs.com. 4 February 2020. Archived from the original on 28 November 2020. Retrieved 7 August 2020.
- ↑ "Summary for ARTG Entry:122957 Zavesca miglustat 100 mg capsules blister pack". Therapeutic Goods Administration.
{{cite web}}: CS1 maint: url-status (link) - ↑ "Zavesca (miglustat) 100 mg hard capsules - Summary of Product Characteristics (SmPC)". (emc). Archived from the original on 25 February 2021. Retrieved 7 August 2020.
- ↑ 6.0 6.1 BNF 81: March-September 2021. BMJ Group and the Pharmaceutical Press. 2021. p. 1117. ISBN 978-0857114105.
- ↑ "Miglustat Prices, Coupons & Patient Assistance Programs". Drugs.com. Archived from the original on 18 April 2021. Retrieved 18 November 2021.
- ↑ Cox TM, Aerts JM, Andria G, Beck M, Belmatoug N, Bembi B, et al. (2003). "The role of the iminosugar N-butyldeoxynojirimycin (miglustat) in the management of type I (non-neuronopathic) Gaucher disease: a position statement". Journal of Inherited Metabolic Disease. 26 (6): 513–26. doi:10.1023/a:1025902113005. PMID 14605497. S2CID 6681399.
- ↑ 9.0 9.1 American Society of Health-System Pharmacists, Inc. for the Public Library of Medicine. Miglustat on MedlinePlus Archived 2016-07-05 at the Wayback Machine Accessed 1 September 2014
- ↑ Grabowski, GA (2012). "Gaucher disease and other storage disorders". Hematology Am Soc Hematol Educ Program. 2012: 13–8. doi:10.1182/asheducation.v2012.1.13.3797921. PMID 23233555.
- ↑ Huddleston, Richard D. (July 1999). "FDA Clinical Investigator Site Inspections: The Sponsor's Role". Drug Information Journal. 33 (3): 965–968. doi:10.1177/009286159903300338. ISSN 0092-8615. S2CID 72975032.
- ↑ Deegan, PB; Cox, TM (2012). "Imiglucerase in the treatment of Gaucher disease: a history and perspective". Drug Design, Development and Therapy. 6: 81–106. doi:10.2147/DDDT.S14395. PMC 3340106. PMID 22563238.
- ↑ "Shire Announces FDA Approval Of VPRIV(TM) (velaglucerase Alfa For Injection) For The Treatment Of Type I Gaucher Disease". Medicalnewstoday.com. Archived from the original on 2011-06-13. Retrieved 2012-08-13.
- ↑ Yukhananov, Anna (1 May 2012). "U.S. FDA approves Pfizer/Protalix drug for Gaucher". Chicago Tribune. Reuters. Archived from the original on 14 September 2022. Retrieved 2 May 2012.[permanent dead link]
- ↑ Actelion. FDA Advisory Briefing Book for Miglustat (Ogt 918, Zavesca) in Niemann-Pick Type C Disease NDA 021-348/S-007 Archived 2017-05-09 at the Wayback Machine Prepared for the Endocrinologic and Metabolic Drugs Advisory Committee meeting, 1 December 2009
- ↑ Abian O, Alfonso P, Velazquez-Campoy A, Giraldo P, Pocovi M, Sancho J (December 2011). "Therapeutic strategies for Gaucher disease: miglustat (NB-DNJ) as a pharmacological chaperone for glucocerebrosidase and the different thermostability of velaglucerase alfa and imiglucerase". Molecular Pharmaceutics. 8 (6): 2390–7. doi:10.1021/mp200313e. PMID 21988669.
- ↑ European Medicines Agency 1 April 2003 Scientific discussion related to approval of Zavesca Archived 2015-09-24 at the Wayback Machine.
- ↑ UK Medicines Information. New Drugs Online Report for miglustat Archived 2016-03-04 at the Wayback Machine
- ↑ Staff, The Pharma Letter. 4 April 2012. Actelion drops setipiprant, gets miglustat approval in Japan Archived 2018-10-31 at the Wayback Machine
- ↑ Kevin Grogan for PharmaTimes. 10 March 2010. FDA rejects Actelion's Zavesca for rare NP-C disease Archived 2014-09-03 at the Wayback Machine
- ↑ Actelion Press Release. 23 March 2010 Zavesca (Miglustat) First Treatment Available in Canada for Rare Progressive Niemann-Pick Type C Disease Archived 2017-05-17 at the Wayback Machine
- ↑ Solomon BI, Smith AC, Sinaii N, Farhat N, King MC, Machielse L, Porter FD. Association of Miglustat With Swallowing Outcomes in Niemann-Pick Disease, Type C1. JAMA Neurol. 2020 Sep 8;77(12):1–6. doi: 10.1001/jamaneurol.2020.3241. Epub ahead of print. PMID: 32897301; PMCID: PMC7489403. [1] Archived 2021-05-27 at the Wayback Machine
- ↑ Clinicaltrials.gov Pharmacokinetics, Safety and Tolerability of Zavesca (Miglustat) in Patients With Infantile Onset Gangliosidosis: Single and Steady State Oral Doses Archived 2012-02-13 at the Wayback Machine Accessed 1 September 2014
- ↑ Clinicaltrials.gov Miglustat / OGT 918 in the Treatment of Cystic Fibrosis Archived 2021-04-06 at the Wayback Machine Accessed 1 September 2014
- ↑ Leonard, A; et al. (May 2012). "A randomized placebo-controlled trial of miglustat in cystic fibrosis based on nasal potential difference". Journal of Cystic Fibrosis. 11 (3): 231–6. doi:10.1016/j.jcf.2011.12.004. PMID 22281182.
External links
| External sites: | |
|---|---|
| Identifiers: |
|
- Pages using duplicate arguments in template calls
- CS1 maint: url-status
- Webarchive template wayback links
- All articles with dead external links
- Articles with dead external links from January 2018
- Articles with invalid date parameter in template
- Articles with permanently dead external links
- Drugs with non-standard legal status
- Chemical articles with unknown parameter in Infobox drug
- Chemical articles without CAS registry number
- Articles without EBI source
- Chemical pages without ChemSpiderID
- Chemical pages without DrugBank identifier
- Articles without KEGG source
- Articles without UNII source
- Drugs missing an ATC code
- Drug has EMA link
- Iminosugars
- Transferase inhibitors
- Orphan drugs
- Piperidines
- RTT